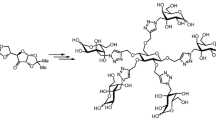
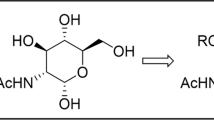
Abstract

Carbohydrate–triazole conjugates proved themselves as valuable enzyme activity-modifying agents. Recent exploration of nontrivial conjugates in which the bonding is formed not at the glycosidic or terminal carbons of the carbohydrates, but at C-3 position showed a potential of this type of structures as the ligands for various glycosidases and galectins. Here, we report synthesis of protected monomeric 3-C-(triazolylmethyl)-substituted gluco- and alloconjugates. Diastereomeric azides are synthesized from common intermediate, 3-deoxy-1,2:5,6-di-O-isopropylidene-3-oxo-α-D-allofuranose, and used in Cu-catalyzed azide–alkyne cycloaddition (CuAAC) reactions with commercial alkynes. The yields of the cycloaddition reactions are good to excellent under different catalytic conditions.



Similar content being viewed by others
References
Gonzaga, D. T. G.; da Rocha, D. R.; da Silva, F. de C.; Ferreira, V. F. Curr. Top. Med. Chem. 2013, 13, 2850.
Sharma, S.; Saquib, M.; Verma, S.; Mishra, N. N.; Shukla, P. K.; Srivastava, R.; Prabhakar, Y. S.; Shaw, A. K. Eur. J. Med. Chem. 2014, 83, 474.
Parida, P. K.; Sau, A.; Ghosh, T.; Jana, K.; Biswas, K.; Raha, S.; Misra, A. K. Bioorg. Med. Chem. Lett. 2014, 24, 3865.
(a) Koester, D. C.; Holkenbrink, A.; Werz, D. B. Synthesis 2010, 3217. (b) da Rocha, D. R.; Santos, W. C.; Lima, E. S.; Ferreira, V. F. Carbohydr. Res. 2012, 350, 14. (c) Ferreira, S.; Sodero, A. C. R.; Cardoso, M. F. C.; Lima, E. S.; Kaiser, C. R.; Silva F. P., Jr.; Ferreira, V. F. J. Med. Chem. 2010, 53, 2364. (d) Seelhorst, K.; Piernitzki, T.; Lunau, N.; Meier, C.; Hahn, U. Bioorg. Med. Chem. 2014, 22, 6430.
Awan, S. I.; Werz, D. B. Bioorg. Med. Chem. 2012, 20, 1846.
Goyard, D.; Docsa, T.; Gergely, P.; Praly, J.-P.; Vidal, S. Carbohydr. Res. 2015, 402, 245.
(a) Tornoe, C. W.; Christensen, C.; Meldal, M. J. Org. Chem. 2002, 67, 3057. (b) Rostovtsev, V. V.; Green, L. G.; Fokin, V. V.; Sharpless, K. B. Angew. Chem., Int. Ed. 2002, 41, 2596.
Uzuleņa, J.; Rjabovs, V.; Moreno Vargas, A. J.; Turks, M. Chem. Heterocycl. Compd. 2015, 51, 664. [Khim. Geterotsikl. Soed. 2015, 51, 664.]
Mackeviča, J.; Ostrovskis, P.; Leffler, H.; Nilsson, U. J.; Rudovica, V.; Viksna, A.; Belyakov, S.; Turks, M. ARKIVOC 2014, (iii), 90.
Rjabova, J.; Rjabovs, V.; Moreno Vargas, A. J.; Moreno Clavijo, E.; Turks, M. Cent. Eur. J. Chem. 2012, 10, 386.
(a) Ostrovskis, P.; Mackeviča, J.; Kumpiņš, V.; López, Ó.; Turks, M. In Carbohydrate Chemistry: Proven Synthetic Methods; van der Marel, G.; Codee, J., Eds.; CRC Press: Boca Raton, London, New York, 2014, vol. 2, p. 275. (b) Turks, M.; Rodins, V.; Rolava, E.; Ostrovskis, P.; Belyakov, S. Carbohydr. Res. 2013, 375, 5. (c) Turks, M.; Rolava, E.; Stepanovs, D.; Mishnev, A.; Marković, D. Tetrahedron: Asymmetry 2015, 26, 952.
(a) Luginina, J.; Rjabovs, V.; Belyakov, S.; Turks, M. Carbohydr. Res. 2012, 350, 86. (b) Albrecht, H. P.; Moffatt, J. G. Tetrahedron Lett. 1970, 13, 1063.
(a) Cavender, C. J.; Shiner, V. J. J. Org. Chem. 1972, 37, 3567. (b) Strakova, I.; Kumpiņa, I.; Rjabovs, V.; Lugiņina, J.; Belyakov, S.; Turks, M. Tetrahedron: Asymmetry 2011, 22,
(c) Rjabovs, V.; Ostrovskis, P.; Posevins, D.; Kiseļovs, G.; Kumpiņš, V.; Mishnev, A.; Turks, M. Eur. J. Org. Chem. 2015, 5572.
(a) Sahabuddin, Sk.; Ghosh, R.; Achari, B.; Mandal, S. B. Org. Biomol. Chem. 2006, 4, 551. (b) Tardy, S.; Lobo Vicente, J.; Tatibouët, A.; Dujardin, G.; Rollin, P. Synthesis 2008, 3108.
Lugiņina, J.; Rjabovs, V.; Belyakov, S.; Turks, M. Tetrahedron Lett. 2013, 54, 5328.
Schmidt, M. S.; Leitner, K.; Welter, M.; Wurmthaler, L. A.; Ringwald, M. Carbohydr. Res. 2014, 387, 42.
This work was supported by the Latvian Council of Science (grant No. 10.0030). The authors thank JSC ''Olainfarm'' for kind donation of diacetone-D-glucose. JSC ''Grindeks'' is acknowledged for kind donation of organic solvents.
Author information
Authors and Affiliations
Corresponding author
Additional information
Pablished in Khimiya Geterotsiklicheskikh Soedinenii, 2015, 51(10), 883–890
Rights and permissions
About this article
Cite this article
Grigorjeva, J., Uzuleņa, J., Rjabovs, V. et al. Synthesis of monomeric methylene-linked 1,2,3-triazole glycoconjugates from allo- and glucofuranoses. Chem Heterocycl Comp 51, 883–890 (2015). https://doi.org/10.1007/s10593-015-1791-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-015-1791-5