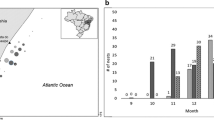
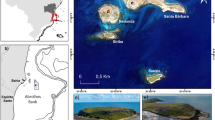
Abstract
Sea turtle hybridization is a common phenomenon in Brazil between loggerheads (Caretta caretta) and hawksbills (Eretmochelys imbricata) as well as between loggerheads and olive ridleys (Lepidochelys olivacea). In a previous study we showed that the reproductive output of loggerhead/hawksbill hybrids is similar to that of parental species, suggesting no negative effect of hybridization at this life stage. In this study, we used pooled amplicon sequencing to assign species identity to dams and their progeny, and to investigate the fitness consequences of hybridization, using hatchling viability as a proxy for fitness. We genotyped 4829 hatchlings from egg clutches laid by 78 loggerheads, 13 hawksbills, seven loggerhead/hawksbill hybrids, and three loggerhead/olive ridley hybrids. The proportion of viable hybrid (heterozygous) hatchlings was similar to that of homozygous hatchlings (based on data at two loci), independent of the dam’s genotype. Multiple species paternity was observed in 35.7% of the nests. Both hybrid males and females were fertile and produced viable offspring, and we found no evidence for hybrid breakdown. We suggest a genome-wide study of the hybrids and parental species to better characterize hybrids, as well as studies on additional demographic and ecological parameters to further assess the effects of hybridization and its consequences for sea turtles and their environment.



Similar content being viewed by others
References
Arnold ML, Hodges SA (1995) Are natural hybrids fit or unfit relative to their parents? TREE 10:67–71
Bass AL, Good DA, Bjorndal KA, Richardson JI, Hillis ZM, Horrocks JA, Bowen BW (1996) Testing models of female reproductive migratory behaviour and population structure in the Caribbean hawksbill turtle, Eretmochelys imbricata, with mtDNA sequences. Mol Ecol 5:321–328. https://doi.org/10.1046/j.1365-294X.1996.00073.x
Bjorndal KA (1997) Foraging ecology and nutrition of sea turtles. In: Lutz PL, Musick JA (eds) The biology of sea turtles. CRC Press, Boca Raton, pp 199–231
Bjorndal KA, Jackson JBC (2003) Roles of sea turtles in marine ecosystems: reconstructing the past. In: Lutz PL, Musick JA, Wyneken J (eds) Biology of sea turtles, vol II. CRC Press, Boca Raton, pp 259–273
Bowen BW, Karl SA (2007) Population genetics and phylogeography of sea turtles. Mol Ecol 16:4886–4907. https://doi.org/10.1111/j.1365-294X.2007.03542.x
Breese MR, Liu Y (2013) NGSUtils: a software suite for analyzing and manipulating next-generation sequencing datasets. Bioinformatics 29:494–496
Butlin R (1987) Speciation by reinforcement. Trends Ecol Evol 2:8–13. https://doi.org/10.1016/0169-5347(87)90193-5
Castilhos JC, Coelho CA, Argolo JF, Santos EAP, Marcovaldi M, Santos AS, Lopez M (2011) Avaliação do estado de conservação da tartaruga marinha Lepidochelys olivacea (Eschscholtz, 1829) no Brasil. [Analysis of the conservation status of the olive ridley turtle (Lepidochelys olivacea, Eschscholtz, 1829) in Brazil]. Biodivers Bras 1:28–36
Conceicao MB, Levy JA, Marins LF, Marcovaldi MA (1990) Electrophoretic characterization of a hybrid between Eretmochelys imbricata and Caretta caretta (Cheloniidae). Comp Biochem Physio 97B:275–278
Garman S (1888) Reptiles and batrachians from the Caymans and the Bahamas. Bull Essex Inst 20:1–13
Giese CLA (2005) The big bad wolf hybrid: how molecular genetics research may undermine protection for gray wolves under the Endangered Species Act. Minn J Law Sci Technol 1532:865–872
Kearse M, Moir R et al (2012) Geneious basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 28:1647–1649
Kelez S, Velez-Zuazo X, Pacheco AS (2016) First record of hybridization between green Chelonia mydas and hawksbill Eretmochelys imbricata sea turtles in the Southeast Pacific. PeerJ 4:e1712
Lara-Ruiz P, Lopez GG, Santos FR, Soares LS (2006) Extensive hybridization in hawksbill turtles (Eretmochelys imbricata) nesting in Brazil revealed by mtDNA analyses. Conserv Genet 7:773–781
Marcovaldi MA, Chaloupka M (2007) Conservation status of the loggerhead sea turtle in Brazil: an encouraging outlook. Endanger Species Res 3:133–143
Marcovaldi MA, Marcovaldi GG (1999) Marine turtles of Brazil: the history and structure of Projeto TAMAR-IBAMA. Biol Conserv 91:35–41. https://doi.org/10.1016/S0006-3207(99)00043-
Marcovaldi MA, Lopez GG, Soares LS, Santos AJB, Bellini C, Barata PCR (2007) Fifteen years of hawksbill sea turtle (Eretmochelys imbricata) nesting in Northern Brazil. Chelonian Conserv Biol 6:223–228
Marcovaldi MA, Lopez GG, Soares LS, López-Mendilaharsu M (2012) Satellite tracking of hawksbill turtles Eretmochelys imbricata nesting in northern Bahia, Brazil: turtle movements and foraging destinations. Endanger Species Res 17:123–132
Mir K, Neuhaus K, Bossert M, Schober S (2013) Short barcodes for next generation sequencing. PLoS ONE 8(12):e82933. https://doi.org/10.1371/journal.pone.0082933
Mortimer JA (1982) Feeding ecology of sea turtles. In: Bjorndal KA (ed) Biology and conservation of sea turtles. Smithsonian Institute Press, Washington, DC, pp 45–51
Musick JA, Limpus CJ (1997) Habitat utilization and migration in juvenile sea turtles. Biol Sea Turt 1:137–163
Plotkin P (2003) Adult migrations and habitat use. Biol Sea Turt 2:225–241
Proietti MC, Reisser J, Marins LF, Marcovaldi MA, Soares LS, Monteiro DS, Wijeratne S, Pattiaratchi C, Secchi ER (2014) Hawksbill × loggerhead sea turtle hybrids at Bahia, Brazil: where do their offspring go? PeerJ 2:e255
Prosdocimi L, Bruno I, Diaz L, Carman VG, Albareda DA, Remis MI (2014) Southernmost reports of the hawksbill sea turtle, Eretmochelys imbricata, in temperate waters of Argentina and evidence of a hybrid origin supported by mitochondrial DNA analysis. Herp Rev 45:1–5
Reis EC, Soares LS, Lôbo-Hajdu G (2010) Evidence of olive ridley mitochondrial genome introgression into loggerhead turtle rookeries of Sergipe, Brazil. Conserv Genet 11:1587–1591
Silva ACCD, Castilhos JC, Lopez GG, Barata PCR (2007) Nesting biology and conservation of the olive ridley sea turtle (Lepidochelys olivacea) in Brazil, 1991/1992 to 2002/2003. J Mar Biol Assoc UK 87:1047–1056. https://doi.org/10.1017/S0025315407056378
Soares LS, Bolten AB, Wayne ML, Vilaça ST, Santos FR, dei Marcovaldi MAG, Bjorndal KA (2017) Comparison of reproductive output of hybrid sea turtles and parental species. Mar Biol 164:9. https://doi.org/10.1007/s00227-016-3035-3
Stronen AV, Paquet PC (2013) Perspectives on the conservation of wild hybrids. Biol Conserv 167:390–395. https://doi.org/10.1016/j.biocon.2013.09.004
Todesco M, Pascual MA et al (2016) Hybridization and extinction. Evol Appl 9(7):892–908. https://doi.org/10.1111/eva.12367
Vilaça ST, Vargas SM, Lara-Ruiz P, Molfetti E, Reis EC, Lobo-Hadju G, Soares LS, Santos FR (2012) Nuclear markers reveal a complex introgression pattern among marine turtle species on the Brazilian coast. Mol Ecol 21:4300–4312. https://doi.org/10.1111/j.1365-294X.2012.05685.x
Vilaça ST, Lara-Ruiz P, Marcovaldi MA, Soares LS, Santos FR (2013) Population origin and historical demography in hawksbill (Eretmochelys imbricata) feeding and nesting aggregates from Brazil. J Exp Mar Biol Ecol 446:334–344
Acknowledgements
We are very grateful to Projeto TAMAR biologists and trainees for their help collecting the data, especially L. Verissímo, B. Canal, and D. Mora. We thank G. Maurutto for his help with Fig. 1. We are thankful to J. Wellehan for his contribution and discussions on the design of the genetic assessment on this study. We are also grateful to M. Schlig for the indispensable help processing the genetic samples, and P. Thompson for his help with processing the samples in the NGS Illumina sequencer. Our studies were supported by the following funding sources: Archie Carr Center for Sea Turtle Research general funds, Tropical Conservation and Development Grant, PADI Foundation, Maturo Excellence Fund, Lerner-Gray Memorial Fund, Beckman Foundation, the Michael L. May Research Grants, Lalita Shastry, the Cynthia A. Melnick Endowment, and an NSF Grant (DEB 1541005) to SFM.
Author information
Authors and Affiliations
Contributions
LSS, KAB, ABB and MLW designed the study with input from MAM. Field work and sampling were conducted by LSS, PBL and RM. Laboratory and data analyses were conducted by LSS, RL and ACP with input from MLW, SFM, TBW, KAB, and ABB. LSS drafted the first version of the manuscript, and all authors contributed to the writing of the final version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving animals were in accordance with the ethical standards of the University of Florida and Projeto TAMAR-ICMBio at which the studies were conducted. This research was approved by the Institutional Animal Care and Use Committees at the University of Florida (201101985) and conducted under SISBIO permit 28938-3 from the Brazilian Ministry of the Environment. Samples were exported under CITES permit 13BR010456/DF, and were imported into the United States under CITES permits 13US724540/9 (Archie Carr Center for Sea Turtle Research).
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Soares, L.S., Bjorndal, K.A., Bolten, A.B. et al. Effects of hybridization on sea turtle fitness. Conserv Genet 19, 1311–1322 (2018). https://doi.org/10.1007/s10592-018-1101-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10592-018-1101-8