Abstract
Cancer patients represent a vulnerable cohort during the Sars-CoV-2 pandemic. Oncological societies have generated a plethora of recommendations, but precise instructions about routine oncological procedures remain scarce. Here, we report on local COVID-19 protection measures established in an interdisciplinary approach at a tertiary care center during the first wave of the pandemia in Germany. Following these measures, no additional morbidity or mortality during oncological procedures was observed, and no nosocomial infections were registered. However, Validation of our measures is outstanding and regional SARS-CoV-2 prevalence was low. However, specific oncological measures might be important to ensure optimal oncological results, especially for advanced cancer stages during this and future pandemia. In the future, communication about these measures might be crucial to a cancer patient´s assigned network to reduce the danger of excess mortality within the second wave of the COVID-19 pandemic.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Commentary
SARS-CoV-2 spread from Wuhan all over the world, ultimately causing a global pandemia affecting several aspects of medicine, as well as oncology. Patients and physicians have had to face several new challenges [1]. Although the number of new infections in Germany was low within the first half of 2020, lack of broad immunity towards SARS-CoV-2 within society requires SARS-CoV-2 to be part of everyday clinical practice. Nonetheless, SARS-CoV-2 has again become particularly important during the ongoing second wave in Germany. Oncological diseases and therapies could cause additional morbidity and mortality in the context of the SARS-CoV-2 pandemic, which might selectively affect patients with advanced or metastatic diseases. Both pandemia-related logistical bottlenecks and SARS-CoV-2 need to be considered when counseling patients. Conversely, excessive protective measures, or fears among patients could lead to increased mortality in cancer care [2,3,4]. Finally, it is a matter of debate whether cancer associated immunological incompetence, or immunological effects of cancer therapies are protective or are accelerating factors towards SARS-CoV-2 associated lung failure, most likely based on a hyperinflammatory mechanism [5].
Preliminary data suggest that infection rates with SARS-CoV-2 are comparable between cancer and non-cancer patients [6,7,8]. At present, 20% of SARS-CoV-2 cases are assumed to be severe, and at least 4% demand intensive care support [9, 10]. Age, as well as concomintant cardiovascular diseases, are major risk factors for a serious course of this infection [11]. Experience from comparable constellations with community-acquired respiratory viruses suggests that cancer patients are at considerable risk of SARS-CoV-2 related mortality of up to 25% [12,13,14]. Preliminary data of SARS-CoV-2 are suggestive of an increased risk in cancer patients with high rates of severe courses (up to 39%) and mortality (up to 29%). Further on, oncological therapies might have an impact on both parameters [6, 8]. These concerns are supported by prospective data from the CCC-19-, Teravolt-, as well as U.K. registries, recently presented at ASCO and ESMO. However, the majority of current publications is limited in impact, due to methodological restrictions, special aspects of a pandemia, selection criteria, regional differences, or missing peer review as well as small numbers of cases [15,16,17]. It is currently elusive to what extent the symptoms of SARS-CoV-2 disease are similar among cancer and non-cancer patients. Also, an overlap of symptoms of SARS-CoV-2 and cancer treatment related adverse events can be assumed. Both considerations, rises concerns, that differential diagnosis might cause additional confusions during the SARS-CoV-2 pandemic [18]. Ultimately it needs to be clarified, that the oncological care intensity and measures need to be adapted to the extent of regional outbreak. on the pandemic regional level; considerations in regions with a high SARS-CoV-2 prevalence, possibly accompanied by medical and personal resource bottlenecks, seems to differ from regions with a low prevalence The needs and requirements obviously differ from regions with low SARS-CoV-2 prevalence to regions with a high prevalence that possibly face medical and personal resource bottlenecks [19].
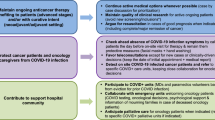
Oncologic practitioners are challenged to manage under- and over-treatment in the everyday world, and even more so within a pandemic Further on, cancer patients are at particular risk of excess mortality, as well as increased SARS-CoV2-related mortality [2]. The likelihood of SARS-CoV-2-related death is usually lower than the risk arising from metastatic disease; however, a severe SARS-CoV-2 course can lead to the premature death of these patients [5]. Several recommendations exist about treatment during the current pandemic [20,21,22]. However, from the view of evidence-based medicine, these guidelines present rather pragmatic perspectives of risk/benefit evaluation. So far, there are hardly any procedural suggestions available. Here, we present an interdisciplinary consensus as a result of communication with practitioners at the Comprehensive Cancer Center Lower Saxony (CCC-N) and the local university hospital SARS-CoV-2 task-force. Therein, based on weekly phone conferences, local pandemic assumptions, local experience and published data, a plethora of measures were discussed (structured panel discussion) and considered to be integrated into daily clinical routine. Discussed measures considered several aspects: administration, cancer care plans, patient-directed, hospital preparedness, employee directed, as listed in Table 1. Although the majority of measures addressed risk reduction for SARS-CoV-2 exposure, either to patients or to staff, adjusting cancer care plans is challenging and needs to be individualized. In our experience, reflecting routine oncological decision steps in an interdisciplinary manner is key to achieving optimized pandemic adapted oncological treatment pathways. (Supp. Fig. S1, Fig. S2, Supplemental Fig. S3, S4).
In general, procedural questions arise at several oncological decision steps, requiring interdisciplinary exchange (Fig. 1, Supp. Fig. S1-3). With regard to oncological therapy in the metastatic setting, neoadjuvant, adjuvant, or palliative, primarily the patient´s risk of acquiring or becoming positive for SARS-CoV-2 needs to be assessed (Fig. 1). In addition to a sufficient medical history and examination with regard to SARS-CoV-2 exposure and symptoms, potentially a methodology-adequate a SARS-CoV-2 test must be considered in accordance with local regulations. SARS-CoV-2 testing is of particular importance when considering in hospital-procedures, rather than out-patient procedures, since nosocomial SARS-CoV-2-infections were described in China’s cancer cohorts [6, 20]. A positive SARS-CoV-2 test result justifies in most cases a postponement of treatment initiation for up to four weeks, although oncological remission pressure, critical anatomical aspects of metastasis and underlying tumor biology need to be considered (Fig. 1). In case of a negative SARS-CoV-2 test, and prior to therapy, risk factors for a severe course of a potential SARS-CoV-2 infection (e.g. age, chronic heart diseases, lymphopenia, D-dimer elevation, immunosuppression) should be evaluated and balanced with the planned anti-cancer treatment (Fig. 1) (23). operability, local control rate, the individualization of post-neoadjuvant therapy and overall survival. (23); in individual cases, neoadjuvant or adjuvant therapy might be omitted (see Supplement Figs. S1, S2, and S3).
Beyond SARS-CoV-2 evaluation, once the decision is made in favor of anti-cancer therapy, the question of treatment urgency needs to be addressed. Apart from objective remission pressure, there are little data on the benefit of immediate versus delayed treatment initiation, in particular in the metastatic setting. Therefore, depending on local infection rates, availability of resources and an individual patient´s risk for a severe course of a potential SARS-CoV-2 infection must be weighed against the benefits of immediate treatment initiation. Drug holidays or watchful waiting might be considered as alternatives to active treatment, especially in the advanced disease cases, during a high SARS-CoV-2 prevalence, and in high risk patients (Fig. 1) [23]. At the start of palliative therapy due to advanced or metastatic disease status, the possibility of a less intensive therapy should be discussed in the first-line setting, as well as throughout sequencial palliative therapy, in particular once remission pressure is considered to be low. Here, the ESMO prioritization between high, medium and low priority therapies appears adequate for everyday use, when addressing the question of which regimen to select [20]. For example, gemcitabine/abraxane instead of FOLFIRINOX in advanced pancreatic cancer, or de-escalation of doxorubicin/ifosfamide to doxorubicin in soft-tissue sarcoma, or early de-escalation of inductive immuno-oncological combination (CTLA-4/PD-1) to PD-1 monotherapy could be considered. Response rates, as well as overall survival, main drivers of standard of care definitions, should be critically weighed against rates of adverse events and hospitalizations during a pandemic.
SARS-CoV-2 represents a new relevant differential diagnosis for infectious complications during therapy. The management of febrile neutropenia according to the S3 guideline and the local Sars-CoV-2 standard appears appropriate (Supp. Fig. S1). A shift in chemotherapy e.g. to four weeks and continuation of systemic therapy after recovery of lymphopenia or granulopenia seem justifiable (see Supp. Fig. S4). In principle, an earlier preventive use of hematolymphopoietic growth factors is another possibly useful supportive measure, considering potential bottlenecks in blood supply [23]. In the case of follow up, IT-based applications may help to reduce the possibility of SARS-CoV-2 exposure and should be considered [20, 24].
Radiotherapy usually consists of a longer serial therapy period without interruption (Supp. Fig. S2). This type of therapy needs to remain feasible during a pandemic, in particular in the curative setting, or once tumor burden qualifies a patient for radiotherapy. Nevertheless, depending on the risk posed by the pandemic situation, the need for therapy must be checked critically in an interdisciplinary manner. Also dosing has to be critically evaluated. In order to keep the total treatment time as short as possible, hypofractionated radiation could be used preferentially during a pandemic, in accordance with the current recommendations and level of evidence (see Supp. Fig. S3) [25].
Although considerations were made mainly for the advanced or metastatic setting, application to the limited diesease setting is also feasible, as illustrated in Supplemental Figs. S3 and S4.
In summary, the Sars-CoV-2 pandemia poses a major challenge to interdisciplinary oncology and a strong interdisciplinary discussion – always essential in oncology—is necessary for now and in the near future to ensure a more individualized approach to treatment decisions. Due to our experience, oncology in its best form according to interdisciplinary consensus is essential more than ever. due to our experience. The procedural considerations presented here, resulting from an ongoing interdisciplinary structured discussion, enabled our center to continue oncological therapies in the CCC-N without relevant additional Sars-CoV-2-related morbidity or mortality. No nosocomial infections were so far observed. Doubtless, this has to be critically reflected by the fact that local SARS-CoV-2 infections rates were low during the first wave of the pandemic and resource bottlenecks did not define our daily work. However, our measures have not yet been stress tested.
Under or over-treatment, as well as increased excess mortality, must also be avoided in future pandemic phases. The cumulative oncological network knowledge and data expertise, both in the context of interdisciplinary tumor conferences and in national network structures such as CCCs and NCTs, appear to be required and useful to optimally adjust and communicate the individual therapy algorithm during the pandemic. This is essential for the foreseeable future. Ultimately, the current SARS-CoV-2 pandemc is an opportunity to generate evidence to better define needs of oncological patients for future pandemia, which hopefully won’t appear to soon.
References
Hanna TP, Evans GA, Booth CM (2020) Cancer, COVID-19 and the precautionary principle: prioritizing treatment during a global pandemic. Nat Rev Clin Oncol 17:268–270. https://doi.org/10.1038/s41571-020-0362-6
Harky A, Chiu CM, Yau THL, Lai SHD (2020) Cancer Patient Care during COVID-19. Cancer Cell 37:749–750
Lai A, Pasea L, Banerjee A, et al (2020) Estimating excess mortality in people with cancer and multimorbidity in the COVID-19 emergency. Doi: https://doi.org/10.1101/2020.05.27.20083287
Jones D, Neal RD, Duffy SRG et al (2020) Comment Impact of the COVID-19 pandemic on the symptomatic diagnosis of cancer : the view from primary care. Lancet Oncol 2045:19–20. https://doi.org/10.1016/S1470-2045(20)30242-4
Sidaway P (2020) COVID-19 and cancer: what we know so far. Nat Rev Clin Oncol 17:336
Zhang L, Zhu F, Xie L et al (2020) Clinical characteristics of COVID-19-infected cancer patients: a retrospective case study in three hospitals within Wuhan, China. Ann Oncol 31:894–901. https://doi.org/10.1016/j.annonc.2020.03.296
von Lilienfeld-Toal M, Berger A, Christopeit M et al (2016) Community acquired respiratory virus infections in cancer patients—Guideline on diagnosis and management by the Infectious Diseases Working Party of the German Society for haematology and Medical Oncology. Eur J Cancer 67:200–212
Liang W, Guan W, Chen R et al (2020) Cancer patients in SARS-CoV-2 infection: a nationwide analysis in China. Lancet Oncol 21:335–337. https://doi.org/10.1016/S1470-2045(20)30096-6
Wu Z, McGoogan JM (2020) Characteristics of and Important Lessons from the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72314 Cases from the Chinese Center for Disease Control and Prevention. JAMA - J Am Med Assoc 323:1239–1242
Guan W, Ni Z, Hu Y et al (2020) Clinical Characteristics of Coronavirus Disease 2019 in China. N Engl J Med 382:1708–1720. https://doi.org/10.1056/nejmoa2002032
RKI (2020) SARS-CoV-2 Steckbrief zur Coronavirus-Krankheit-2019 (COVID-19).
Chemaly RF, Vigil KJ, Saad M et al (2012) A multicenter study of pandemic influenza A (H1N1) infection in patients with solid tumors in 3 countries: Early therapy improves outcomes. Cancer 118:4627–4633. https://doi.org/10.1002/cncr.27447
Schnell D, Mayaux J, de Bazelaire C et al (2010) Risk factors for pneumonia in immunocompromised patients with influenza. Respir Med 104:1050–1056. https://doi.org/10.1016/j.rmed.2010.01.021
Ljungman P, de la Camara R, Perez-Bercoff L et al (2011) Outcome of pandemic H1N1 infections in hematopoietic stem cell transplant recipients. Haematologica 96:1231–1235. https://doi.org/10.3324/haematol.2011.041913
Desai A, Sachdeva S, Parekh T, Desai R (2020) COVID-19 and Cancer: Lessons From a Pooled Meta-Analysis. JCO Glob Oncol. https://doi.org/10.1200/go.20.00097
Xia Y, Jin R, Zhao J et al (2020) Risk of COVID-19 for patients with cancer. Lancet Oncol 21:e180
Oh WK (2020) COVID-19 Infection in Cancer Patients: Early Observations and Unanswered Questions. Ann Oncol Off J Eur Soc Med Oncol. https://doi.org/10.1016/j.annonc.2020.03.297
Sahu KK, Jindal V, Siddiqui AD (2020) Managing COVID-19 in Patients With Cancer: A Double Blow for Oncologists. JCO Oncol Pract 16:223–225. https://doi.org/10.1200/op.20.00167
Pino L, Perez C, Cardona A, Triana I (2020) Cancer Center Recommendations to Mitigate COVID-19 Impact in Patients With Cancer: Low-Resource Settings Version. JCO Glob Oncol. https://doi.org/10.1200/go.20.00093
Aapro M, Addeo A, Acierto P, Berg T (2020) Cancer Patient Management During the COVID-19 Pandemic | ESMO. https://www.esmo.org/guidelines/cancer-patient-management-during-the-covid-19-pandemic. Accessed 1 May 2020
COVID-19 Clinical Oncology Frequently Asked Questions (FAQs)
Von Lilienfeld-Toal M, Greinix H, Hein A, et al (2020) Coronavirus-Infektion (COVID-19) bei Patienten mit Blut- und Krebserkrankungen. Onkopedia Leitlinien
NCCN (2020) NCCN Hematopoietic Growth Factors Short-Term Recommendations Specific to Issues with COVID-19 (SARS-CoV-2). In: NCCN
Al-Shamsi HO, Alhazzani W, Alhuraiji A et al (2020) A Practical Approach to the Management of Cancer Patients During the Novel Coronavirus Disease 2019 ( COVID -19) Pandemic: An International Collaborative Group. Oncologist. https://doi.org/10.1634/theoncologist.2020-0213
Combs SE, Belka C, Niyazi M et al (2020) First statement on preparation for the COVID-19 pandemic in large German Speaking University-based radiation oncology departments. Radiat Oncol 15:74
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.



Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ivanyi, P., Park-Simon, T., Christiansen, H. et al. Protective measures for patients with advanced cancer during the Sars-CoV-2 pandemic: Quo vadis?. Clin Exp Metastasis 38, 257–261 (2021). https://doi.org/10.1007/s10585-021-10083-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10585-021-10083-1