Abstract
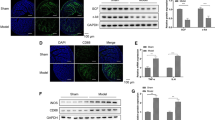
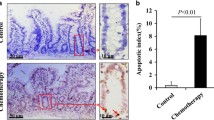
The interstitial cells of Cajal (ICCs) play an important role in maintaining the normal function of gastrointestinal dynamics. In our previous study, we reported that, in advanced gastric cancer, the frequency of bowel movement is always reduced, due in part to the decreased number of ICCs. To investigate the impact of ICCs in gastric cancer, we established a mouse model of gastric cancer peritoneal metastasis using SGC-7901 gastric adenocarcinoma cells and their supernatant. Then, stem cell factor (SCF) and ouabain were used as therapeutic agents to improve gut dynamics. Our data showed that, compared with the normal mice, treatment with SGC-7901 cells and their supernatant led to a significant reduction of the muscle layer thickness, a decreased number of ICCs, broadened gaps between ICCs and surrounding cells, degeneration and necrosis of smooth muscle cells (SMCs), and infiltration of inflammatory cells. In contrast to SGC-7901 cell and supernatant treatment, SCF intervention caused mild submucosal edema and mitochondrial proliferation in the ICCs and SMCs. Additionally, ouabain treatment led to inflammatory cells infiltration into the submucosa and a decreased volume of ICCs. In conclusion, our data illustrated that, under the condition of gastric cancer peritoneal metastasis, the dysfunction of intestinal peristalsis may be related to pathological changes in ICCs. Moreover, we demonstrated that SCF treatment may help to improve intestinal dynamics by regulating the number and function of ICCs.





Similar content being viewed by others
Abbreviations
- ICC:
-
Interstitial cells of Cajal
- SMC:
-
Smooth muscle cells
- SCF:
-
Stem cell factor
- GI:
-
Gastrointestinal
- SID:
-
Stomach–intestine dyskinesis
- VIP:
-
Vasoactive intestinal peptide
- CX43:
-
Connexin 43
- NOS1:
-
NO synthase-1
- NKA:
-
Na+-K+-ATPase
- ENS:
-
Enteric nervous system
References
Siegel R, Naishadham D, Jemal A (2013) Cancer statistics, 2013. CA Cancer J Clin 63(1):11–30
Lu Z et al (2010) Intraperitoneal therapy for peritoneal cancer. Future Oncol 6(10):1625–1641
Zheng H et al (2011) Is gastrointestinal dysfunction induced by gastric cancer peritoneal metastasis relevant to impairment of interstitial cells of Cajal? Clin Exp Metastasis 28(3):291–299
Faussone-Pellegrini MS (1992) Histogenesis, structure and relationships of interstitial cells of Cajal (ICC): from morphology to functional interpretation. Eur J Morphol 30(2):137–148
Lee JC et al (1999) Generation of slow waves in membrane potential is an intrinsic property of interstitial cells of Cajal. Am J Physiol 277(2 Pt 1):G409–G423
Sanders KM et al (2000) A novel pacemaker mechanism drives gastrointestinal rhythmicity. News Physiol Sci 15:291–298
Du P et al (2011) A preliminary model of gastrointestinal electromechanical coupling. IEEE Trans Biomed Eng 58(12):3491–3495
Mostafa RM, Moustafa YM, Hamdy H (2010) Interstitial cells of Cajal, the Maestro in health and disease. World J Gastroenterol 16(26):3239–3248
Ordog T, Ward SM, Sanders KM (1999) Interstitial cells of Cajal generate electrical slow waves in the murine stomach. J Physiol 518(Pt 1):257–269
Tomita T, Hata T (2000) Effects of removal of Na(+) and Cl(−) on spontaneous electrical activity, slow wave, in the circular muscle of the guinea-pig gastric antrum. Jpn J Physiol 50(5):469–477
Habib S, Ali A (2011) Biochemistry of nitric oxide. Indian J Clin Biochem 26(1):3–17
Chen Y et al (2011) Visualization of the interstitial cells of Cajal (ICC) network in mice. J Vis Exp. doi:10.379/2802
Yun HY et al (2010) Regional distribution of interstitial cells of Cajal (ICC) in human stomach. Korean J Physiol Pharmacol 14(5):317–324
Bernex F et al (1996) Spatial and temporal patterns of c-kit-expressing cells in WlacZ/+ and WlacZ/WlacZ mouse embryos. Development 122(10):3023–3033
Wu JJ, Rothman TP, Gershon MD (2000) Development of the interstitial cell of Cajal: origin, kit dependence and neuronal and nonneuronal sources of kit ligand. J Neurosci Res 59(3):384–401
Sanders KM, Ward SM (2006) Interstitial cells of Cajal: a new perspective on smooth muscle function. J Physiol 576(Pt 3):721–726
Sharma B, Singh N (2013) Pharmacological inhibition of inducible nitric oxide synthase (iNOS) and nicotinamide adenine dinucleotide phosphate (NADPH) oxidase, convalesce behavior and biochemistry of hypertension induced vascular dementia in rats. Pharmacol Biochem Behav 103(4):821–830
Forstermann U, Sessa WC (2012) Nitric oxide synthases: regulation and function. Eur Heart J 33(7): 829–837, 837a–837d
Lowry JL et al (2013) Endothelial nitric-oxide synthase activation generates an inducible nitric-oxide synthase-like output of nitric oxide in inflamed endothelium. J Biol Chem 288(6):4174–4193
Voice JK et al (2003) Roles of vasoactive intestinal peptide (VIP) in the expression of different immune phenotypes by wild-type mice and T cell-targeted type II VIP receptor transgenic mice. J Immunol 170(1):308–314
Li JM et al (2011) Absence of vasoactive intestinal peptide expression in hematopoietic cells enhances Th1 polarization and antiviral immunity in mice. J Immunol 187(2):1057–1065
Pauls S et al (2014) Differential contributions of intra-cellular and inter-cellular mechanisms to the spatial and temporal architecture of the suprachiasmatic nucleus circadian circuitry in wild-type, cryptochrome-null and vasoactive intestinal peptide receptor 2-null mutant mice. Eur J Neurosci 40(3):2528–2540
Zhang MQ et al (2014) Cigarette smoking promotes inflammation in patients with COPD by affecting the polarization and survival of Th/Tregs through up-regulation of muscarinic receptor 3 and 5 expression. PLoS One 9(11):e112350
Santini E, Sepulveda-Orengo M, Porter JT (2012) Muscarinic receptors modulate the intrinsic excitability of infralimbic neurons and consolidation of fear extinction. Neuropsychopharmacology 37(9):2047–2056
Stahlhut M et al (2006) The antiarrhythmic peptide rotigaptide (ZP123) increases connexin 43 protein expression in neonatal rat ventricular cardiomyocytes. Cell Commun Adhes 13(1–2):21–27
Chew SS et al (2010) Role of connexin43 in central nervous system injury. Exp Neurol 225(2):250–261
Cai ZX, Li Y, Qi QH (2010) Morphological changes in interstitial cells of Cajal in the deep muscular plexus and enteric motor neurons of the intestine in rats with multiple organ dysfunction syndrome. Neural Regn Res 5(8):635–636
Qi QH et al (2010) Morphological changes in network of enteric nerve-interstitial cells of Cajal-smooth muscle cells in rats with multiple organ dysfunction syndrome and therapeutic effects of Dachengqi decoction. Chin J Integr Med 16(5):422–429
Li K et al (2013) Connexin43 hemichannel-mediated regulation of connexin43. PLoS One 8(2):e58057
Cousins HM et al (2003) Electrical coupling between the myenteric interstitial cells of Cajal and adjacent muscle layers in the guinea-pig gastric antrum. J Physiol 550(Pt 3):829–844
Haas M, Askari A, Xie Z (2000) Involvement of Src and epidermal growth factor receptor in the signal-transducing function of Na +/K + -ATPase. J Biol Chem 275(36):27832–27837
Liu L et al (2004) Role of caveolae in ouabain-induced proliferation of cultured vascular smooth muscle cells of the synthetic phenotype. Am J Physiol Heart Circ Physiol 287(5):H2173–H2182
Xie Z, Askari A (2002) Na(+)/K(+)-ATPase as a signal transducer. Eur J Biochem 269(10):2434–2439
Dostanic I et al (2004) The alpha 1 isoform of Na, K-ATPase regulates cardiac contractility and functionally interacts and co-localizes with the Na/Ca exchanger in heart. J Biol Chem 279(52):54053–54061
Acknowledgments
We thank Heilongjiang Provincial Science and Technology Innovation Team in Higher Education Institutes for Infection and Immunity, Harbin Medical University, Harbin 150081, China.
Funding
This work was supported by the National Natural Science Fund (No. 81372611), the National Natural Science Youth Fund (No. 81301750) and the key project of Heilongjiang Province Educational Administration (No. 12521z017).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Dan Kong and Jing Li contributed equally to this work.
Rights and permissions
About this article
Cite this article
Kong, D., Li, J., Zhao, B. et al. The effect of SCF and ouabain on small intestinal motility dysfunction induced by gastric cancer peritoneal metastasis. Clin Exp Metastasis 32, 267–277 (2015). https://doi.org/10.1007/s10585-015-9702-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10585-015-9702-9