Abstract
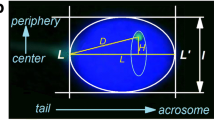
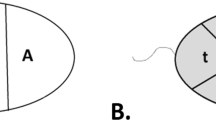
Recently it has been shown that the nucleus of the human spermatozoon appears to possess a specific architecture. The current prevailing view is that spatial organization of the male genome contains information critical for the spermatozoon’s function as well as for early embryonic development. The purpose of this study was to determine whether there are alterations in intranuclear localization of centromeres in spermatozoa of chromosomes associated with particular reciprocal chromosome translocations (RCT). We analyzed the longitudinal and spatial localization of centromeres of selected chromosomes in sperm nuclei of four control males with normal karyotypes as well as in six carriers of reciprocal chromosome translocations: t(1;7), t(7;2), t(7;13), t(7;9), t(9;14), and t(4;13). Our study revealed that chromosomes with translocations may have shifted their intranuclear localization and that these translocations may influence the localization of other chromosomes in sperm nuclei. The chromocenter in sperm nuclei of translocation carriers was widened toward the apical side in comparison with chromocenter sites visible in control males. Our study also revealed interindividual differences in the localization of the Y chromosome centromere in the chromocenter area of sperm from fertile individuals.
Similar content being viewed by others
References
Biggers JD, Whitt WK, Whittingham DG (1971) The culture of mouse embryos in vitro. In: Daniel JC, ed. Methods in Mammalian Embriology. San Francisco: Freeman WH and Co. pp. 86–116.
Celik-Ozenci C, Jakab A, Kovacs T et al. (2004) Sperm selection for ICSI: shape properties do not predict the absence or presence of numerical chromosomal aberrations. Hum Reprod 19: 2052–2059.
Cremer T, Cremer C (2001) Chromosome territories, nuclear architecture and gene regulation in mammalian cells. Nat Rev Genet 2: 292–301.
Cremer T, Kupper K, Dietzel S, Fakan S (2004) Higher order chromatin architecture in the cell nucleus: on the way from structure to function. Biol Cell 96: 555–567.
Claussen U (2005) Chromosomics. Cytogenet Genome Res 111: 101–106.
Dehghani H, Dellaire G, Bazett-Jones DP (2005) Organization of chromatin in the interphase mammalian cell. Micron 36: 95–108.
Foster AH, Bridger JM (2005) The genome and the nucleus: a marriage made by evolution. Genome organization and nuclear architecture. Chromosoma 30: 1–18.
Fraser L (2004) Structural damage to nuclear DNA in mammalian spermatozoa: its evaluation techniques and relationship with male infertility. Pol J Vet Sci 7: 311–321.
Garagna S, Zuccotti M, Thornhill A et al. (2001) Alteration of nuclear architecture in male germ cells of chromosomally derived subfertile mice. J Cell Sci 114: 4429–4434.
Gerlich D, Beaudouin J, Kalbfuss, B, Daigle N, Eils R, Ellenberg J (2003) Global chromosome positions are transmitted through mitosis in mammalian cells. Cell 112: 751–764.
Gineitis AA, Zalenskaya IA, Yau PM, Bradbury EM, Zalensky AO (2000) Human sperm telomere-binding complex involves histone H2B and secures telomere membrane attachment. J Cell Biol 151: 1591–1598.
Greaves IK, Svartman M, Wakefield M et al. (2001) Chromosome painting detects non-random chromosome arrangement in dasyurid marsupial sperm. Chromosome Res 9: 251–259.
Greaves IK, Rens W, Ferguson-Smith MA, Griffin D, Marshall-Graves JA (2003) Conservation of chromosome arrangement and position of the X in mammalian sperm suggest functional significance. Chromosome Res 11: 503–512.
Gurevitch M, Amiel A, Ben-Zion M, Fejgin M, Bartoov B (2001) Acrocentric centromere organization within the chromocenter of the human sperm nucleus. Mol Reprod Dev 60: 507–516.
Haaf T, Ward DC (1995) Higher order nuclear structure in mammalian sperm revealed by in situ hybridization and extended chromatin fibers. Exp Cell Res 219: 604–611.
Hagstrom KA, Meyer BJ (2003) Condensin and cohesin: more than chromosome compactor and glue. Nat Rev Genet 4: 520–534.
Hazzouri M, Rousseaux S, Mongelard F et al. (2000) Genome organization in the human sperm nucleus studies by FISH and confocal microscopy. Mol Reprod Dev 55: 307–315.
Hoyer-Fender S, Singh PB, Motzkus D (2000) The murine heterochromatin protein M31 is associated with the chromocenter in round spermatids and is a component of mature spermatozoa. Exp Cell Res 254: 72–79.
Iwarsson E, Malmgren H, Inzunza J et al. (2000) Highly abnormal cleavage divisions in preimplantation embryos from translocation carriers. Prenat Diagn 20: 1038–1047.
Luetjens CM, Payne C, Schatten G (1999) Non-random chromosome positioning in human sperm and sex chromosome anomalies following intracytoplasmic sperm injection. Lancet 353: 1240.
Martianov I, Brancorsini S, Gansmuuler A, Parvinen M, Davidson I, Sassone-Corsi P (2002) Distinct functions of TBP and TLF/TRF2 during spermatogenesis: requirement of TLF for heterochromatic chromocenter formation in haploid round spermatids. Development 129: 945–955.
Martinez-Pasarell O, Marquez C, Coll MD, Egozcue J, Templado C (1997) Analysis of human sperm-derived pronuclei by three-colour fluorescent in-situ hybridization. Hum Reprod 12: 641–645.
Meyer-Ficca M, Muller-Navia J, Scherthan H (1998) Clustering of pericentromeres initiates in step 9 of spermiogenesis of the rat (Rattus norvegicus) and contributes to a well defined genome architecture in the sperm nucleus. J Cell Sci 111: 1363–1370.
Midro AT, Wiland E, Panasiuk B, Leśniewicz R, Kurpisz M (2006) Risk evaluation of carriers with chromosome reciprocal translocation t(7;13)(q34;q13) and concomitant meiotic segregation analyzed by FISH on ejaculated spermatozoa. Am J Med Genet 140A: 245–256.
Mudrak O, Tomilin N, Zalensky AO (2005) Chromosome architecture in the decondensing human sperm nucleus. J Cell Sci 118: 41–45.
Munne S (2005) Analysis of chromosome segregation during preimplantation genetic diagnosis in both male and female translocation heterozygotes. Cytogenet Genome Res 111: 305–309.
Pujol A, Benet J, Staessen C et al. (2006) The importance of aneuploidy screening in reciprocal translocation carriers. Reproduction 131: 1025–1035.
Sbracia M, Baldi M, Cao D et al. (2002) Preferential location of sex chromosomes, their aneuploidy in human sperm, and their role in determining sex chromosome aneuploidy in embryos after ICSI. Hum Reprod 17: 320–324.
Solovei IV, Joffe BJ, Hori T, Thomson P, Mizuno S, Macgregor HC (1998) Unordered arrangement of chromosomes in the nuclei of chicken spermatozoa. Chromosoma 107: 184–188.
Solov’eva L, Svetlova M, Bodinski D, Zalensky AO (2004) Nature of telomere dimmers and chromosome looping in human spermatozoa. Chromosome Res 12: 817–823.
Sun F, Ko E, Martin RH (2006) Is there a relationship between sperm chromosome abnormalities and sperm morphology? Reprod Biol Endocrinol 4:1 www.rbej.com./content/4/1/1.
Terada Y, Luetjens CM, Sutovsky P, Schatten G (2000) Atypical decondensation of the sperm nucleus, delayed replication of the male genome, and sex chromosome positioning following intracytoplasmic human sperm injection (ICSI) into golden hamster eggs: does ICSI itself introduce chromosomal anomalies? Fertil Steril 74: 454–460.
Thomson I, Gilchrist S, Bickmore WA, Chubb JR (2004) The radial positioning of chromatin is not inherited through mitosis but is established de novo in early G1. Curr Biol 14: 166–172.
Tilgen N, Guttenbach M, Schmid M (2001) Heterochromatin is not an adequate explanation for close proximity of interphase chromosomes 1–9, 9–Y, and 16–Y in human spermatozoa. Exp Cell Res 265: 283–287.
Walter J, Schermelleh L, Cremer M, Tashiro S, Cremer T (2003) Chromosome order in HeLa cells changes during mitosis and early G1, but is stable maintained during subsequent interphase stages. J Cell Biol 160: 685–697.
Ward MA, Ward WS (2004) A model for the function of sperm DNA degradation. Reprod Fertil Dev 16: 547–554.
Williams RR, Fisher AB (2003) Chromosomes, position please! Nat Cell Biol 5: 388–390.
Wiland E, Hobel CJ, Hill D, Kurpisz M (2008) Successful pregnancy after preimplantational genetic diagnosis for carrier of t(2;7)(11.2:q22) with high rates of unbalanced sperm and embryos. Prenatal Diagnosis 78: 36–41.
Zalenskaya IA, Zalensky AO (2004) Non-random positioning of chromosomes in human sperm nuclei. Chromosome Res 12: 163–173.
Zalenskaya I, Bradbury E, Zalensky AO (2000) Chromatin structure of telomere domain in human sperm. Biochem Biophys Res Commun 279: 213–218.
Zalensky AO, Allen MJ, Kobayashi A, Zalenskaya IA, Balhorn R, Bradbury EM (1995) Well-defined genome architecture in the human sperm nucleus. Chromosoma 103: 577–590.
Zalensky AO, Tomilin NV, Zalenskaya IA, Teplitz RL, Bradbury EM (1997) Telomere-telomere interactions and candidate telomere binding protein(s) in mammalian sperm cells. Exp Cell Res 232: 29–41.
Zalensky AO, Siino JS, Gineitis AA et al. (2002) Human testis/sperm –specific histone H2B(hTSH2B). Molecular cloning and characterization. J Biol Chem 277: 43474–43480.
Zini A, Libman J (2006) Sperm DNA damage: clinical significance in the era of assisted reproduction. CMAJ 175: 495–500.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wiland, E., Żegało, M. & Kurpisz, M. Interindividual differences and alterations in the topology of chromosomes in human sperm nuclei of fertile donors and carriers of reciprocal translocations. Chromosome Res 16, 291–305 (2008). https://doi.org/10.1007/s10577-007-1194-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10577-007-1194-2