Abstract
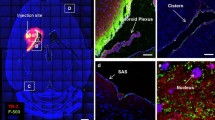
The clearance of brain interstitial fluid (ISF) is important in maintaining brain homeostasis. ISF clearance impairment leads to toxic material accumulation in the brain, and ischemic stroke could impair ISF clearance. The present study investigates ISF clearance under normal and ischemic conditions. The carboxylate-modified FluoSpheres beads (0.04 μm in diameter) were injected into the striatum. Sham or transient middle cerebral artery occlusion surgeries were performed on the mice. The brain sections were immunostained with cell markers, and bead distribution at various time points was examined with a confocal microscope. Primary mouse neuronal cultures were incubated with the beads to explore in vitro endocytosis. Two physiological routes for ISF clearance were identified. The main one was to the lateral ventricle (LV) through the cleft between the striatum and the corpus callosum (CC)/external capsule (EC), where some beads were captured by the ependymal macrophages and choroid plexus. An alternative and minor route was to the subarachnoid space through the CC/EC and the cortex, where some of the beads were endocytosed by neurons. After ischemic stroke, a significant decrease in the main route and an increase in the minor route were observed. Additionally, microglia/macrophages engulfed the beads in the infarction. In conclusion, we report that the physiological clearance of ISF and beads mainly passes through the cleft between the CC/EC and striatum into the LV, or alternatively through the cortex into the subarachnoid space. Stroke delays the main route but enhances the minor route, and microglia/macrophages engulf the beads in the infarction.
Graphical Abstract

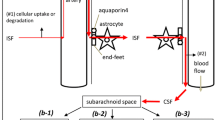
Ischemic stroke impairs the clearance of brain interstitial fluid/beads. Under physiological conditions, the main route ( ① ) of interstitial fluid clearance is to the lateral ventricle, and the minor one ( ② ) is to the subarachnoid space. Ischemic stroke weakens the main route ( ① ), enhances the minor one ( ② ), and leads to microglial/macrophage phagocytosis within the infarction ( ③ )







Similar content being viewed by others
Data Availability
The datasets analyzed and used during the current study are available from the corresponding author on reasonable request.
References
Abe T, Black PM, Foley L (1984) Changes in parenchymal and ventricular pressure with experimental epidural compression. Surg Neurol 22(5):477–480. https://doi.org/10.1016/0090-3019(84)90306-9
Allen DJ (1975) Scanning electron microscopy of epiplexus macrophages (Kolmer cells) in the dog. J Comp Neurol 161(2):197–213. https://doi.org/10.1002/cne.901610205
Arbel-Ornath M, Hudry E, Eikermann-Haerter K, Hou S, Gregory JL, Zhao LZ, Betensky RA, Frosch MP, Greenberg SM, Bacskai BJ (2013) Interstitial fluid drainage is impaired in ischemic stroke and Alzheimer’s disease mouse models. Acta Neuropathol 126(3):353–364. https://doi.org/10.1007/s00401-013-1145-2
Bedussi B, van Lier MG, Bartstra JW, de Vos J, Siebes M, VanBavel E, Bakker EN (2015) Clearance from the mouse brain by convection of interstitial fluid towards the ventricular system. Fluids Barriers Cns 12:23. https://doi.org/10.1186/s12987-015-0019-5
Bowen S, Ateh DD, Deinhardt K, Bird MM, Price KM, Baker CS, Robson JC, Swash M, Shamsuddin W, Kawar S, El-Tawil T, Roos J, Hoyle A, Nickols CD, Knowles CH, Pullen AH, Luthert PJ, Weller RO, Hafezparast M, Franklin RJ, Revesz T, King RH, Berninghausen O, Fisher EM, Schiavo G, Martin JE (2007) The phagocytic capacity of neurones. Eur J Neurosci 25(10):2947–2955. https://doi.org/10.1111/j.1460-9568.2007.05554.x
Cai H, Wang XP, Yang GY (2021) Sleep disorders in stroke: an update on management. Aging Dis 12(2):570–585. https://doi.org/10.14336/AD.2020.0707
Cao G, Pei W, Lan J, Stetler RA, Luo Y, Nagayama T, Graham SH, Yin XM, Simon RP, Chen J (2001) Caspase-activated DNase/DNA fragmentation factor 40 mediates apoptotic DNA fragmentation in transient cerebral ischemia and in neuronal cultures. J Neurosci 21(13):4678–4690. https://doi.org/10.1523/JNEUROSCI.21-13-04678.2001
Carpenter SJ, McCarthy LE, Borison HL (1970) Electron microscopic study of the epiplexus (Kolmer) cells of the cat choroid plexus. Z Zellforsch Mikrosk Anat 110(4):471–486. https://doi.org/10.1007/BF00330099
Coates PW (1973) Supraependymal cells in recesses of the monkey third ventricle. Am J Anat 136(4):533–539. https://doi.org/10.1002/aja.1001360410
Cserr HF, Cooper DN, Milhorat TH (1977) Flow of cerebral interstitial fluid as indicated by the removal of extracellular markers from rat caudate nucleus. Exp Eye Res 25 Suppl:461–473. https://doi.org/10.1016/s0014-4835(77)80041-9
del Zoppo GJ, Sharp FR, Heiss WD, Albers GW (2011) Heterogeneity in the penumbra. J Cereb Blood Flow Metab 31(9):1836–1851. https://doi.org/10.1038/jcbfm.2011.93
Dubnovitsky A, Sandberg A, Rahman MM, Benilova I, Lendel C, Hard T (2013) Amyloid-beta protofibrils: size, morphology and synaptotoxicity of an engineered mimic. PLoS ONE 8(7):e66101. https://doi.org/10.1371/journal.pone.0066101
Faul F, Erdfelder E, Lang AG, Buchner A (2007) G*Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods 39(2):175–191. https://doi.org/10.3758/bf03193146
Gaberel T, Gakuba C, Goulay R, De Lizarrondo SM, Hanouz JL, Emery E, Touze E, Vivien D, Gauberti M (2014) Impaired glymphatic perfusion after strokes revealed by contrast-enhanced MRI a new target for fibrinolysis? Stroke 45(10):3092–. https://doi.org/10.1161/Strokeaha.114.006617
Garcia-Alloza M, Gregory J, Kuchibhotla KV, Fine S, Wei Y, Ayata C, Frosch MP, Greenberg SM, Bacskai BJ (2011) Cerebrovascular lesions induce transient beta-amyloid deposition. Brain 134:3694–3704. https://doi.org/10.1093/brain/awr300
Goldmann T, Wieghofer P, Jordao MJ, Prutek F, Hagemeyer N, Frenzel K, Amann L, Staszewski O, Kierdorf K, Krueger M, Locatelli G, Hochgerner H, Zeiser R, Epelman S, Geissmann F, Priller J, Rossi FM, Bechmann I, Kerschensteiner M, Linnarsson S, Jung S, Prinz M (2016) Origin, fate and dynamics of macrophages at central nervous system interfaces. Nat Immunol 17(7):797–805. https://doi.org/10.1038/ni.3423
Han X, Ren H, Nandi A, Fan X, Koehler RC (2021) Analysis of glucose metabolism by (18)F-FDG-PET imaging and glucose transporter expression in a mouse model of intracerebral hemorrhage. Sci Rep 11(1):10885. https://doi.org/10.1038/s41598-021-90216-4
Hladky SB, Barrand MA (2022) The glymphatic hypothesis: the theory and the evidence. Fluids Barriers Cns. https://doi.org/10.1186/s12987-021-00282-z
Hollander H, Egensperger R, Dirlich G (1989) Size distribution of rhodamine-labeled microspheres retrogradely transported in cultured neurons. J Neurosci Meth 29(1):1–4 doi:Doi 10.1016/0165–0270(89)90102-7
Hoshi M, Sato M, Matsumoto S, Noguchi A, Yasutake K, Yoshida N, Sato K (2003) Spherical aggregates of beta-amyloid (amylospheroid) show high neurotoxicity and activate tau protein kinase I/glycogen synthase kinase-3beta. Proc Natl Acad Sci USA 100(11):6370–6375. https://doi.org/10.1073/pnas.1237107100
Hughes JL, Beech JS, Jones PS, Wang D, Menon DK, Baron JC (2010) Mapping selective neuronal loss and microglial activation in the salvaged neocortical penumbra in the rat. NeuroImage 49(1):19–31. https://doi.org/10.1016/j.neuroimage.2009.08.047
Iliff JJ, Wang MH, Liao YH, Plogg BA, Peng WG, Gundersen GA, Benveniste H, Vates GE, Deane R, Goldman SA, Nagelhus EA, Nedergaard M (2012) A paravascular pathway facilitates CSF flow through the brain parenchyma and the clearance of interstitial solutes, including amyloid beta. Sci Transl Med. https://doi.org/10.1126/scitranslmed.3003748
Jessen NA, Munk AS, Lundgaard I, Nedergaard M (2015) The glymphatic system: a beginner’s guide. Neurochem Res 40(12):2583–2599. https://doi.org/10.1007/s11064-015-1581-6
Ji CH, Yu X, Xu WL, Lenahan C, Tu S, Shao AW (2021) The role of glymphatic system in the cerebral edema formation after ischemic stroke. Exp Neurol. https://doi.org/10.1016/j.expneurol.2021.113685
Kato J, Murata Y, Takashima I, Higo N (2021) Time- and area-dependent macrophage/microglial responses after focal infarction of the macaque internal capsule. Neurosci Res 170:350–359. https://doi.org/10.1016/j.neures.2020.12.001
Katz LC, Iarovici DM (1990) Green fluorescent latex microspheres—a new retrograde tracer. Neuroscience 34(2):511–520.
Katz LC, Burkhalter A, Dreyer WJ (1984) Fluorescent latex microspheres as a retrograde neuronal marker for in vivo and in vitro studies of visual cortex. Nature 310(5977):498–500. https://doi.org/10.1038/310498a0
Kaur J, Fahmy LM, Davoodi-Bojd E, Zhang L, Ding G, Hu J, Zhang Z, Chopp M, Jiang Q (2021) Waste clearance in the brain. Front Neuroanat 15:665803. https://doi.org/10.3389/fnana.2021.665803
Klostranec JM, Vucevic D, Bhatia KD, Kortman HGJ, Krings T, Murphy KP, terBrugge KG, Mikulis DJ (2021) Current concepts in intracranial interstitial fluid transport and the glymphatic system: part I-anatomy and physiology. Radiology 301(3):502–514. https://doi.org/10.1148/radiol.2021202043
Komlosh ME, Benjamini D, Williamson NW, Horkay F, Hutchinson EB, Basser PJ (2019) A novel MRI phantom to study interstitial fluid transport in the glymphatic system. Magn Reson Imaging 56:181–186. https://doi.org/10.1016/j.mri.2018.10.007
Kress BT, Iliff JJ, Xia M, Wang M, Wei HS, Zeppenfeld D, Xie L, Kang H, Xu Q, Liew JA, Plog BA, Ding F, Deane R, Nedergaard M (2014) Impairment of paravascular clearance pathways in the aging brain. Ann Neurol 76(6):845–861. https://doi.org/10.1002/ana.24271
Lei Y, Han H, Yuan F, Javeed A, Zhao Y (2017) The brain interstitial system: anatomy, modeling, in vivo measurement, and applications. Prog Neurobiol 157:230–246. https://doi.org/10.1016/j.pneurobio.2015.12.007
Li Q, Lou J, Yang T, Wei Z, Li S, Zhang F (2021) Ischemic preconditioning induces oligodendrogenesis in mouse brain: effects of Nrf2 deficiency. Cell Mol Neurobiol. https://doi.org/10.1007/s10571-021-01068-5
Li Q, Fadoul G, Ikonomovic M, Yang T, Zhang F (2022) Sulforaphane promotes white matter plasticity and improves long-term neurological outcomes after ischemic stroke via the Nrf2 pathway. Free Radic Biol Med 193(Pt 1):292–303. https://doi.org/10.1016/j.freeradbiomed.2022.10.001
Lopez-Atalaya JP, Askew KE, Sierra A, Gomez-Nicola D (2018) Development and maintenance of the brain’s immune toolkit: microglia and non-parenchymal brain macrophages. Dev Neurobiol 78(6):561–579. https://doi.org/10.1002/dneu.22545
Maher F, Davies-Hill TM, Lysko PG, Henneberry RC, Simpson IA (1991) Expression of two glucose transporters, GLUT1 and GLUT3, in cultured cerebellar neurons: evidence for neuron-specific expression of GLUT3. Mol Cell Neurosci 2(4):351–360. https://doi.org/10.1016/1044-7431(91)90066-w
Manoonkitiwongsa PS, Jackson-Friedman C, McMillan PJ, Schultz RL, Lyden PD (2001) Angiogenesis after stroke is correlated with increased numbers of macrophages: the clean-up hypothesis. J Cereb Blood Flow Metab 21(10):1223–1231. https://doi.org/10.1097/00004647-200110000-00011
Mijajlovic MD, Pavlovic A, Brainin M, Heiss WD, Quinn TJ, Ihle-Hansen HB, Hermann DM, Assayag EB, Richard E, Thiel A, Kliper E, Shin YI, Kim YH, Choi S, Jung S, Lee YB, Sinanovic O, Levine DA, Schlesinger I, Mead G, Milosevic V, Leys D, Hagberg G, Ursin MH, Teuschl Y, Prokopenko S, Mozheyko E, Bezdenezhnykh A, Matz K, Aleksic V, Muresanu D, Korczyn AD, Bornstein NM (2017) Post-stroke dementia—a comprehensive review. BMC Med 15(1):11. https://doi.org/10.1186/s12916-017-0779-7
Munro DAD, Movahedi K, Priller J (2022) Macrophage compartmentalization in the brain and cerebrospinal fluid system. Sci Immunol 7(69):eabk0391. https://doi.org/10.1126/sciimmunol.abk0391
Paletzki RF, Gerfen CR (2019) Basic neuroanatomical methods. Curr Protoc Neurosci 90(1):e84. https://doi.org/10.1002/cpns.84
Pasic Z, Smajlovic D, Dostovic Z, Kojic B, Selmanovic S (2011) Incidence and types of sleep disorders in patients with stroke. Med Arh 65(4):225–227. https://doi.org/10.5455/medarh.2011.65.225-227
Pu H, Wang Y, Yang T, Leak RK, Stetler RA, Yu F, Zhang W, Shi Y, Hu X, Yin KJ, Hitchens TK, Dixon CE, Bennett MVL, Chen J (2023) Interleukin-4 mitigates anxiety-like behavior and loss of neurons and fiber tracts in limbic structures in a microglial PPARgamma-dependent manner after traumatic brain injury. Neurobiol Dis 180:106078. https://doi.org/10.1016/j.nbd.2023.106078
Rambaran RN, Serpell LC (2008) Amyloid fibrils: abnormal protein assembly. Prion 2(3):112–117
Saber R, Sarkar S, Gill P, Nazari B, Faridani F (2011) High resolution imaging of IgG and IgM molecules by scanning tunneling microscopy in air condition. Scientia Iranica 18:1643–1646
Schilling M, Besselmann M, Leonhard C, Mueller M, Ringelstein EB, Kiefer R (2003) Microglial activation precedes and predominates over macrophage infiltration in transient focal cerebral ischemia: a study in green fluorescent protein transgenic bone marrow chimeric mice. Exp Neurol 183(1):25–33. https://doi.org/10.1016/s0014-4886(03)00082-7
Shetty AK, Zanirati G (2020) The interstitial system of the brain in health and disease. Aging Dis 11(1):200–211. https://doi.org/10.14336/Ad.2020.0103
Steffen U, Koeleman CA, Sokolova MV, Bang H, Kleyer A, Rech J, Unterweger H, Schicht M, Garreis F, Hahn J, Andes FT, Hartmann F, Hahn M, Mahajan A, Paulsen F, Hoffmann M, Lochnit G, Munoz LE, Wuhrer M, Falck D, Herrmann M, Schett G (2020) IgA subclasses have different effector functions associated with distinct glycosylation profiles. Nat Commun 11(1):120. https://doi.org/10.1038/s41467-019-13992-8
Sturrock RR (1978) A developmental study of epiplexus cells and supraependymal cells and their possible relationship to microglia. Neuropathol Appl Neurobiol 4(5):307–322. https://doi.org/10.1111/j.1365-2990.1978.tb01345.x
Sun BL, Wang LH, Yang T, Sun JY, Mao LL, Yang MF, Yuan H, Colvin RA, Yang XY (2018) Lymphatic drainage system of the brain: a novel target for intervention of neurological diseases. Prog Neurobiol 163–164:118–143. https://doi.org/10.1016/j.pneurobio.2017.08.007
Szentistvanyi I, Patlak CS, Ellis RA, Cserr HF (1984) Drainage of interstitial fluid from different regions of rat-brain. Am J Physiol 246(6):F835–F844. https://doi.org/10.1152/ajprenal.1984.246.6.F835
Tarasoff-Conway JM, Carare RO, Osorio RS, Glodzik L, Butler T, Fieremans E, Axel L, Rusinek H, Nicholson C, Zlokovic BV, Frangione B, Blennow K, Menard J, Zetterberg H, Wisniewski T, de Leon MJ (2015) Clearance systems in the brain-implications for Alzheimer disease. Nat Rev Neurol 11(8):457–470. https://doi.org/10.1038/nrneurol.2015.119
Thrane AS, Rangroo Thrane V, Nedergaard M (2014) Drowning stars: reassessing the role of astrocytes in brain edema. Trends Neurosci 37(11):620–628. https://doi.org/10.1016/j.tins.2014.08.010
Tian Y, Zhao M, Chen Y, Yang M, Wang Y (2022) The underlying role of the glymphatic system and meningeal lymphatic vessels in cerebral small vessel disease. Biomolecules. https://doi.org/10.3390/biom12060748
Ting SM, Zhao X, Sun G, Obertas L, Ricote M, Aronowski J (2020) Brain cleanup as a potential target for poststroke recovery: the role of RXR (retinoic X receptor) in phagocytes. Stroke 51(3):958–966. https://doi.org/10.1161/STROKEAHA.119.027315
Toh CH, Siow TY (2021) Glymphatic dysfunction in patients with ischemic stroke. Front Aging Neurosci. https://doi.org/10.3389/fnagi.2021.756249
Tonnesen J, Hrabetova S, Soria FN (2023) Local diffusion in the extracellular space of the brain. Neurobiol Dis 177:105981. https://doi.org/10.1016/j.nbd.2022.105981
Ueno M, Chiba Y, Murakami R, Matsumoto K, Fujihara R, Uemura N, Yanase K, Kamada M (2019) Disturbance of intracerebral fluid clearance and blood-brain barrier in vascular cognitive impairment. Int J Mol Sci. https://doi.org/10.3390/ijms20102600
Wallace DM, Ramos AR, Rundek T (2012) Sleep disorders and stroke. Int J Stroke 7(3):231–242. https://doi.org/10.1111/j.1747-4949.2011.00760.x
Wallace VJ, Cimbro R, Rubio FJ, Fortuno LV, Necarsulmer JC, Koivula PP, Henderson MJ, DeBiase LM, Warren BL, Harvey BK, Hope BT (2017) Neurons internalize functionalized micron-sized silicon dioxide microspheres. Cell Mol Neurobiol 37(8):1487–1499. https://doi.org/10.1007/s10571-017-0479-z
Xie L, Kang H, Xu Q, Chen MJ, Liao Y, Thiyagarajan M, O’Donnell J, Christensen DJ, Nicholson C, Iliff JJ, Takano T, Deane R, Nedergaard M (2013) Sleep drives metabolite clearance from the adult brain. Science 342(6156):373–377. https://doi.org/10.1126/science.1241224
Yang T, Zhang F (2021) Targeting transcription factor Nrf2 (nuclear factor erythroid 2-Related factor 2) for the intervention of vascular cognitive impairment and dementia. Arterioscler Thromb Vasc Biol 41(1):97–116. https://doi.org/10.1161/ATVBAHA.120.314804
Yang G, Chan PH, Chen J, Carlson E, Chen SF, Weinstein P, Epstein CJ, Kamii H (1994) Human copper-zinc superoxide dismutase transgenic mice are highly resistant to reperfusion injury after focal cerebral ischemia. Stroke 25(1):165–170. https://doi.org/10.1161/01.str.25.1.165
Yang T, Sun Y, Mao L, Zhang M, Li Q, Zhang L, Shi Y, Leak RK, Chen J, Zhang F (2018) Brain ischemic preconditioning protects against ischemic injury and preserves the blood-brain barrier via oxidative signaling and Nrf2 activation. Redox Biol 17:323–337. https://doi.org/10.1016/j.redox.2018.05.001
Yang T, Guo R, Zhang F (2019) Brain perivascular macrophages: recent advances and implications in health and diseases. CNS Neurosci Ther 25(12):1318–1328. https://doi.org/10.1111/cns.13263
Yang T, Li Q, Fadoul G, Alraqmany N, Ikonomovic M, Zhang F (2023) Aldo-Keto reductase 1C15 characterization and protection in ischemic brain injury. Antioxidants (Basel) 12(4):909. https://doi.org/10.3390/antiox12040909
Yang T, Sun Y, Li Q, Li S, Shi Y, Leak RK, Chen J, Zhang F (2020) Ischemic preconditioning provides long-lasting neuroprotection against ischemic stroke: the role of Nrf2. Exp Neurol 325:113142. https://doi.org/10.1016/j.expneurol.2019.113142
Zhang ET, Richards HK, Kida S, Weller RO (1992) Directional and compartmentalised drainage of interstitial fluid and cerebrospinal fluid from the rat brain. Acta Neuropathol 83(3):233–239. https://doi.org/10.1007/BF00296784
Zhang F, Vannucci SJ, Philp NJ, Simpson IA (2005) Monocarboxylate transporter expression in the spontaneous hypertensive rat: effect of stroke. J Neurosci Res 79(1–2):139–145. https://doi.org/10.1002/jnr.20312
Zhang F, Wang S, Signore AP, Chen J (2007) Neuroprotective effects of leptin against ischemic injury induced by oxygen-glucose deprivation and transient cerebral ischemia. Stroke 38(8):2329–2336. https://doi.org/10.1161/STROKEAHA.107.482786
Zhang F, Wang S, Zhang M, Weng Z, Li P, Gan Y, Zhang L, Cao G, Gao Y, Leak RK, Sporn MB, Chen J (2012) Pharmacological induction of heme oxygenase-1 by a triterpenoid protects neurons against ischemic injury. Stroke 43(5):1390–1397. https://doi.org/10.1161/STROKEAHA.111.647420
Zhang L, Johnson D, Johnson JA (2013) Deletion of Nrf2 impairs functional recovery, reduces clearance of myelin debris and decreases axonal remyelination after peripheral nerve injury. Neurobiol Dis 54:329–338. https://doi.org/10.1016/j.nbd.2013.01.003
Acknowledgements
This work was supported in part by the National Institutes of Health (NS092810), and start-up funds from the Pittsburgh Institute of Brain Disorders and Recovery and the Department of Neurology of the University of Pittsburgh. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. We thank Pat Strickler for her administrative support.
Funding
National Institutes of Health (NS092810).
Author information
Authors and Affiliations
Contributions
TY and FZ designed the research. YS, TY, QL, NA, and FZ performed the experiments. YS, TY, and FZ analyzed and/or interpreted the data. TY and FZ drafted and revised the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interest.
Consent for Publication
All authors agree for publication.
Research Involving Human and Animal Participants
University of Pittsburgh Institutional Animal Care and Use Committee (IACUC) and the National Institutes of Health Guide for the Care and Use of Laboratory Animals. All the animals used in the study were purchased.
Additional information
Publisher’s Note
Springer nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yang, T., Sun, Y., Li, Q. et al. Effects of Ischemic Stroke on Interstitial Fluid Clearance in Mouse Brain: a Bead Study. Cell Mol Neurobiol 43, 4141–4156 (2023). https://doi.org/10.1007/s10571-023-01400-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-023-01400-1