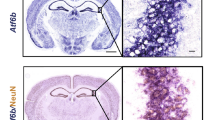
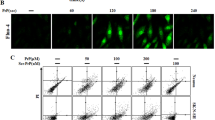
Abstract
Excessive activation of α-amino-3-hydroxy-5-methyl-4-isoxazole propoinic acid (AMPA) receptors instigates excitotoxicity via enhanced calcium influx in the neurons thus inciting deleterious consequences. Additionally, Endoplasmic Reticulum (ER) is pivotal in maintaining the intracellular calcium balance. Considering this, studying the aftermath of enhanced calcium uptake by neurons and its effect on ER environment can assist in delineating the pathophysiological events incurred by excitotoxicty. The current study was premeditated to decipher the role of ER pertaining to calcium homeostasis in AMPA-induced excitotoxicity. The findings showed, increased intracellular calcium levels (measured by flowcytometry and spectroflourimeter using Fura 2AM) in AMPA excitotoxic animals (male Sprague dawely rats) (intra-hippocampal injection of 10 mM AMPA). Further, ER resident proteins like calnexin, PDI and ERp72 were found to be upregulated, which further modulated the functioning of ER membrane calcium channels viz. IP3R, RyR, and SERCA pump. Altered calcium homeostasis further led to ER stress and deranged the protein folding capacity of ER post AMPA toxicity, which was ascertained by unfolded protein response (UPR) pathway markers such as IRE1α, eIF2α, and ATF6α. Chemical chaperone, 4-phenybutric acid (4-PBA), ameliorated the protein folding capacity and subsequent UPR markers. In addition, modulation of calcium channels and calcium regulating machinery of ER post 4-PBA administration restored the calcium homeostasis. Therefore the study reinforces the significance of ER stress, a debilitating outcome of impaired calcium homeostasis, under AMPA-induced excitotoxicity. Also, employing chaperone-based therapeutic approach to curb ER stress can restore the calcium imbalance in the neuropathological diseases.
Graphical Abstract









Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this manuscript. Primary datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- AMPA:
-
α-Amino-3-hydroxy-5-methyl-4-isoxazole propoinic acid
- ATF-6:
-
Activating transcription factor-6
- ER:
-
Endoplasmic reticulum
- GRP75:
-
Glucose regulated protein75
- IP3R:
-
Inositol 1,4,5-triphosphate receptors
- KA:
-
Kianic acid
- MAM:
-
Mitochondrial-associated membrane
- NMDA:
-
N-methyl-D-aspartate
- 4-PBA:
-
4-Phenylbutyric acid
- RyR:
-
Ryanodine receptors
- SERCA:
-
Sacroendoplasmic reticulum Ca ATPase
References
Ambrosi G, Cerri S, Blandini F (2014) A further update on the role of excitotoxicity in the pathogenesis of Parkinson’s disease. J Neural Transm 121:849–859. https://doi.org/10.1007/s00702-013-1149-z
Baek HA, Kim DS, Park HS et al (2012) Involvement of endoplasmic reticulum stress in myofibroblastic differentiation of lung fibroblasts. Am J Respir Cell Mol Biol 46:731–739. https://doi.org/10.1165/rcmb.2011-0121OC
Bernal F, Petegnief V, Rodríguez MJ et al (2009) Nimodipine inhibits TMB-8 potentiation of AMPA-induced hippocampal neurodegeneration. J Neurosci Res 87:1240–1249. https://doi.org/10.1002/jnr.21930
Berridge MJ (2002) The endoplasmic reticulum: a multifunctional signaling organelle. Cell Calcium 32:235–249. https://doi.org/10.1016/S0143416002001823
Bhardwaj A, Bhardwaj R, Dhawan DK, Kaur T (2019) Exploring the effect of endoplasmic reticulum stress inhibition by 4-phenylbutyric acid on AMPA-induced hippocampal excitotoxicity in rat brain. Neurotox Res 35:83–91. https://doi.org/10.1007/s12640-018-9932-0
Bhardwaj A, Bhardwaj R, Sharma S et al (2021) AMPA induced cognitive impairment in rats: establishing the role of endoplasmic reticulum stress inhibitor, 4-PBA. J Neurosci Res 99:2573–2591. https://doi.org/10.1002/JNR.24859
Bravo R, Vicencio JM, Parra V et al (2011) Increased ER-mitochondrial coupling promotes mitochondrial respiration and bioenergetics during early phases of ER stress. J Cell Sci 124:2143–2152. https://doi.org/10.1242/jcs.080762
Calì T, Ottolini D, Brini M (2013) Calcium and endoplasmic reticulum-mitochondria tethering in neurodegeneration. DNA Cell Biol 32:140–146. https://doi.org/10.1089/dna.2013.2011
Chen C-M, Wu C-T, Chiang C-K et al (2012) C/EBP Homologous Protein (CHOP) deficiency aggravates hippocampal cell apoptosis and impairs memory performance. PLoS ONE 7:e40801. https://doi.org/10.1371/journal.pone.0040801
Chen T, Zhu J, Wang YH, Hang CH (2020) Arc silence aggravates traumatic neuronal injury via mGluR1-mediated ER stress and necroptosis. Cell Death Dis 11:1–11. https://doi.org/10.1038/s41419-019-2198-5
Crabbé M, Dirkx N, Casteels C (2019) Laere K Van (2019) Excitotoxic neurodegeneration is associated with a focal decrease in metabotropic glutamate receptor type 5 availability: an in vivo PET imaging study. Sci Reports 91(9):1–10. https://doi.org/10.1038/s41598-019-49356-x
Cullinan SB, Diehl JA (2004) PERK-dependent activation of Nrf2 contributes to redox homeostasis and cell survival following endoplasmic reticulum stress. J Biol Chem 279:20108–20117. https://doi.org/10.1074/jbc.M314219200
Dhanda S, Sunkaria A, Halder A, Sandhir R (2018) Mitochondrial dysfunctions contribute to energy deficits in rodent model of hepatic encephalopathy. Metab Brain Dis 33(1):209–223. https://doi.org/10.1007/s11011-017-0136-8
Dong XX, Wang Y, Qin ZH (2009) Molecular mechanisms of excitotoxicity and their relevance to pathogenesis of neurodegenerative diseases. Acta Pharmacol Sin 30:379–387. https://doi.org/10.1038/aps.2009.24
Ghemrawi R, Khair M (2020) Endoplasmic reticulum stress and unfolded protein response in neurodegenerative diseases. Int J Mol Sci 21:1–25. https://doi.org/10.3390/ijms21176127
Graham JM (2002) Preparation of crude subcellular fractions by differential centrifugation. Scientific World J 2:1638–1642. https://doi.org/10.1100/tsw.2002.851
Hetz C, Saxena S (2017) ER stress and the unfolded protein response in neurodegeneration. Nat Rev Neurol 13:477–491. https://doi.org/10.1038/nrneurol.2017.99
Hsa E, Ya K (2014) Glutamate excitotoxicity and neurodegeneration. J Mol Genet Med 8:1–4. https://doi.org/10.4172/1747-0862.1000141
Itoh T, Kouzu H, Miki T et al (2012) Cytoprotective regulation of the mitochondrial permeability transition pore is impaired in type 2 diabetic Goto-Kakizaki rat hearts. J Mol Cell Cardiol 53:870–879. https://doi.org/10.1016/j.yjmcc.2012.10.001
Joseph SK, Boehning D, Bokkala S et al (1999) Biosynthesis of inositol trisphosphate receptors: selective association with the molecular chaperone calnexin. Biochem J 342:153–161. https://doi.org/10.1042/BJ3420153
King TE, Ohnishi T, Winter DB, Wu JT (1976) Biochemical and EPR probes for structure-function studies of iron sulfur centers of succinate dehydrogenase. Adv Exp Med Biol 74:182–227. https://doi.org/10.1007/978-1-4684-3270-1_15
Kiviluoto S, Vervliet T, Ivanova H et al (2013) Regulation of inositol 1,4,5-trisphosphate receptors during endoplasmic reticulum stress. Biochim Biophys Acta 1833:1612–1624. https://doi.org/10.1016/J.BBAMCR.2013.01.026
Lai S, Li Y, Kuang Y et al (2017) PKCδ silencing alleviates saturated fatty acid induced ER stress by enhancing SERCA activity. Biosci Rep. https://doi.org/10.1042/BSR20170869
Lewerenz J, Maher P (2015) Chronic glutamate toxicity in neurodegenerative diseases-What is the evidence? Front Neurosci 9:1–20. https://doi.org/10.3389/fnins.2015.00469
Li R, Zuo Z, Chen D et al (2011) Inhibition by polycyclic aromatic hydrocarbons of ATPase activities in Sebastiscus marmoratus larvae in relationship with the development of early life stages. Mar Environ Res 71:86–90. https://doi.org/10.1016/j.marenvres.2010.11.002
Lim D, Dematteis G, Tapella L et al (2021) Ca2+ handling at the mitochondria-ER contact sites in neurodegeneration. Cell Calcium 98:102453. https://doi.org/10.1016/J.CECA.2021.102453
Luciani DS, Gwiazda KS, Yang TLB et al (2009) Roles of IP3R and RyR Ca2+ channels in endoplasmic reticulum stress and β-cell death. Diabetes 58:422–432. https://doi.org/10.2337/db07-1762
Luo J, Shi R (2005) Acrolein induces oxidative stress in brain mitochondria. Neurochem Int 46:243–252. https://doi.org/10.1016/j.neuint.2004.09.001
Malhotra JD, Kaufman RJ (2011) ER stress and Its functional link to mitochondria: role in cell survival and death. Cold Spring Harb Perspect Biol 3:1–13. https://doi.org/10.1101/cshperspect.a004424
Marchi S, Patergnani S, Pinton P (2014) The endoplasmic reticulum–mitochondria connection: one touch, multiple functions. Biochim Biophys Acta - Bioenerg 1837:461–469. https://doi.org/10.1016/j.bbabio.2013.10.015
Mattson MP, LaFerla FM, Chan SL et al (2000) Calcium signaling in the ER: Its role in neuronal plasticity and neurodegenerative disorders. Trends Neurosci 23:222–229. https://doi.org/10.1016/S0166-2236(00)01548-4
Mekahli D, Bultynck G, Parys JB et al (2011) Endoplasmic-reticulum calcium depletion and disease. Cold Spring Harb Perspect Biol 3:1–30. https://doi.org/10.1101/cshperspect.a004317
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J Immunol Methods 65(1–2):55–63. https://doi.org/10.1016/0022-1759(83)90303-4
Myhill N, Lynes EM, Nanji JA et al (2008) The subcellular distribution of calnexin is mediated by PACS-2. Mol Biol Cell 19:2777–2788. https://doi.org/10.1091/MBC.E07-10-0995/ASSET/IMAGES/LARGE/ZMK0070885860006.JPEG
Nagakannan P, Islam I et al (2019) Inhibition of VDAC1 protects against glutamate-induced oxytosis and mitochondrial fragmentation in hippocampal HT22 cells. Cell Mol Neurobiol 39:73–85. https://doi.org/10.1007/s10571-018-0634-1
Nakatsu Y, Kotake Y, Komasaka K et al (2006) Glutamate excitotoxicity is involved in cell death caused by tributyltin in cultured rat cortical neurons. Toxicol Sci 89:235–242. https://doi.org/10.1093/TOXSCI/KFJ007
Nishitoh H (2012) CHOP is a multifunctional transcription factor in the ER stress response. J Biochem 151:217–219. https://doi.org/10.1093/jb/mvr143
Ota T, Gayet C, Ginsberg HN (2008) Inhibition of apolipoprotein B100 secretion by lipid-induced hepatic endoplasmic reticulum stress in rodents. J Clin Invest 118:316–332. https://doi.org/10.1172/JCI32752
Pan C, Prentice H, Price AL, Wu JY (2012) Beneficial effect of taurine on hypoxia-and glutamate-induced endoplasmic reticulum stress pathways in primary neuronal culture. Amino Acids 43:845–855. https://doi.org/10.1007/s00726-011-1141-6
Papp S, Dziak E, Michalak M, Opas M (2003) Is all of the endoplasmic reticulum created equal? The effects of the heterogeneous distribution of endoplasmic reticulum Ca2+-handling proteins. J Cell Biol 160:475. https://doi.org/10.1083/JCB.200207136
Petegnief V, Saura J, Dewar D et al (1999) Long-term effects of alpha-amino-3-hydroxy-5-methyl-4-isoxazole propionate and 6-nitro-7-sulphamoylbenzo(f)quinoxaline-2,3-dione in the rat basal ganglia: calcification, changes in glutamate receptors and glial reactions. Neuroscience 94:105–115. https://doi.org/10.1016/S0306-4522(99)00299-7
Phillips MJ (2015) Voeltz GK (2015) Structure and function of ER membrane contact sites with other organelles. Nat Rev Mol Cell Biol 172(17):69–82. https://doi.org/10.1038/nrm.2015.8
Prentice H, Modi JP, Wu J-Y (2015) Mechanisms of neuronal protection against excitotoxicity, endoplasmic reticulum stress, and mitochondrial dysfunction in stroke and neurodegenerative diseases. Oxid Med Cell Longev 2015:7. https://doi.org/10.1155/2015/964518
Rebai O, Amri M (2018) Chlorogenic acid prevents AMPA-mediated excitotoxicity in optic nerve oligodendrocytes through a PKC and caspase-dependent pathways. Neurotox Res 34:559–573. https://doi.org/10.1007/s12640-018-9911-5
Rego AC, Oliveira CR (2003) Mitochondrial dysfunction and reactive oxygen species in excitotoxicity and apoptosis: implications for the pathogenesis of neurodegenerative diseases. Neurochem Res 28:1563–1574
Rizzuto R, Marchi S, Bonora M et al (2009) Ca2+ transfer from the ER to mitochondria: When, how and why. Biochim Biophys Acta - Bioenerg 1787:1342–1351. https://doi.org/10.1016/j.bbabio.2009.03.015
Rizzuto R, Pinton P, Carrington W et al (1998) Close contacts with the endoplasmic reticulum as determinants of mitochondrial Ca2+ responses. Science 280:1763–1766. https://doi.org/10.1126/science.280.5370.1763
Roderick HL, Lechleiter JD, Camacho P (2000) Cytosolic phosphorylation of calnexin controls intracellular Ca2+ oscillations via an interaction with Serca2b. J Cell Biol 149:1235–1248. https://doi.org/10.1083/JCB.149.6.1235
Ruiz A, Matute C, Alberdi E (2010) Intracellular Ca2+ release through ryanodine receptors contributes to AMPA receptor-mediated mitochondrial dysfunction and ER stress in oligodendrocytes. Cell Death Dis 1:54–62. https://doi.org/10.1038/cddis.2010.31
Segal M, Korkotian E (2014) Endoplasmic reticulum calcium stores in dendritic spines. Front Neuroanat 8:1–7. https://doi.org/10.3389/fnana.2014.00064
Seo MD, Enomoto M, Ishiyama N et al (2014) Structural insights into endoplasmic reticulum stored calcium regulation by inositol 1,4,5-trisphosphate and ryanodine receptors. Biochim Biophys Acta - Mol Cell Res 1853:1980–1991. https://doi.org/10.1016/j.bbamcr.2014.11.023
Sokka AL, Putkonen N, Mudo G et al (2007) Endoplasmic reticulum stress inhibition protects against excitotoxic neuronal injury in the rat brain. J Neurosci 27:901–908. https://doi.org/10.1523/JNEUROSCI.4289-06.2007
Srinivasan K, Sharma SS (2011) Sodium phenylbutyrate ameliorates focal cerebral ischemic/reperfusion injury associated with comorbid type 2 diabetes by reducing endoplasmic reticulum stress and DNA fragmentation. Behav Brain Res 225:110–116. https://doi.org/10.1016/J.BBR.2011.07.004
Vannuvel K, Renard P, Raes M, Arnould T (2013) Functional and morphological impact of ER stress on mitochondria. J Cell Physiol 228:1802–1818. https://doi.org/10.1002/jcp.24360
Wang FM, Chen YJ, Ouyang HJ (2011) Regulation of unfolded protein response modulator XBP1s by acetylation and deacetylation. Biochem J 433:245–252. https://doi.org/10.1042/BJ20101293
Wieckowski MRMR, Giorgi C, Lebiedzinska M et al (2009) Isolation of mitochondria-associated membranes and mitochondria from animal tissues and cells. Nat Protoc 4:1582–1590. https://doi.org/10.1038/nprot.2009.151
Xu H, Guan N, Ren YL et al (2018) IP3R-Grp75-VDAC1-MCU calcium regulation axis antagonists protect podocytes from apoptosis and decrease proteinuria in an Adriamycin nephropathy rat model. BMC Nephrol. https://doi.org/10.1186/s12882-018-0940-3
Zhang Y, Tan F, Xu P, Qu S (2016) Recent advance in the relationship between excitatory amino acid transporters and parkinson’s disease. Neural Plast. https://doi.org/10.1155/2016/8941327
Zündorf G, Reiser G (2011) Calcium dysregulation and homeostasis of neural calcium in the molecular mechanisms of neurodegenerative diseases provide multiple targets for neuroprotection. Antioxidants Redox Signal 14:1275–1288. https://doi.org/10.1089/ars.2010.3359
Acknowledgements
We would like to thank center facilities instrumentation laboratory of Centre for Stem Cell and Tissue Engineering and Excellence in Biomedical Sciences Panjab University (PU) Chandigarh for providing facilities for stereotactic surgeries.
Funding
This work was supported by the University Grants Commission-Basic Scientific Research (UGC-BSR) (F.25–1/2013–14(BSR)/7–209/2009(BSR), Department of Science and Technology (FIST) (SR/FST/LS1-645) and University Grant Commission (SAP) (F.4–1/2015/DSA-1 (Sap-4).
Author information
Authors and Affiliations
Contributions
Study concept and design were elaborated by AB and TK. Material preparations and data collection were performed by AB. Formal analysis was accomplished by AB, RB, and TK. The first draft of the manuscript was written by AB and TK supervised the study. All the authors AB, RB, AS, DKD and TK reviewed and commented on the previous version of manuscript. All the authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bhardwaj, A., Bhardwaj, R., Saini, A. et al. Impact of Calcium Influx on Endoplasmic Reticulum in Excitotoxic Neurons: Role of Chemical Chaperone 4-PBA. Cell Mol Neurobiol 43, 1619–1635 (2023). https://doi.org/10.1007/s10571-022-01271-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-022-01271-y