Abstract
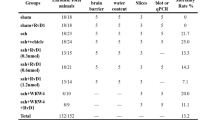
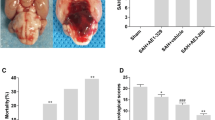
Early brain injury (EBI) is the early phase of secondary complications arising from subarachnoid hemorrhage (SAH). G protein-coupled receptor 18 (GPR18) can exert neuroprotective effects during ischemia. In this study, we investigated the roles of GPR18 in different brain regions during EBI using a GPR18 agonist, resolvin D2 (RvD2). Location and dynamics of GPR18 expression were assessed by immunohistochemistry and western blotting in a rat model of SAH based on endovascular perforation. RvD2 was given intranasally at 1 h after SAH, and SAH grade, brain water content and behavior were assayed before sacrifice. TUNEL and dihydroethidium staining of the cortex were performed at 24 h after SAH. Selected brain regions were also examined for pathway related proteins using immunofluorescence and Western blotting. We found that GPR18 was expressed in meninges, hypothalamus, cortex and white matter before EBI. After SAH, GPR18 expression was increased in meninges and hypothalamus but decreased in cortex and white matter. RvD2 improved neurological scores and brain edema after SAH. RvD2 attenuated mast cell degranulation and reduced expression of chymase and tryptase expression in the meninges. In the hypothalamus, RvD2 attenuated inflammation, increased expression of proopiomelanocortin and interleukin-10, as well as decreased expression of nerve peptide Y and tumor necrosis factor-α. In cortex, RvD2 alleviated oxidative stress and apoptosis, and protected the blood–brain barrier. RvD2 also ameliorated white matter injury by elevating myelin basic protein and suppressing amyloid precursor protein. Our results suggest that GPR18 may help protect multiple brain regions during EBI, particularly in the cortex and hypothalamus. Upregulating GPR18 by RvD2 may improve neurological functions in different brain regions via multiple mechanisms.










Similar content being viewed by others
Data Availability
All data are fully available without restriction. All relevant data can be found in the paper.
Change history
24 April 2024
A Correction to this paper has been published: https://doi.org/10.1007/s10571-024-01475-4
References
Arac A, Grimbaldeston MA, Galli SJ, Bliss TM, Steinberg GK (2019) Meningeal mast cells as key effectors of stroke pathology. Front Cell Neurosci 13:126
Cai J, Xu D, Bai X, Pan R, Wang B, Sun S, Chen R, Sun J et al (2017) Curcumin mitigates cerebral vasospasm and early brain injury following subarachnoid hemorrhage via inhibiting cerebral inflammation. Brain Behav 7(9):e00790
Chen S, Feng H, Sherchan P, Klebe D, Zhao G, Sun X, Zhang J, Tang J et al (2014) Controversies and evolving new mechanisms in subarachnoid hemorrhage. Prog Neurobiol 115:64–91
Chen S, Wu H, Tang J, Zhang J, Zhang JH (2015) Neurovascular events after subarachnoid hemorrhage: focusing on subcellular organelles. Acta Neurochir Suppl 120:39–46
Chen S, Luo J, Reis C, Manaenko A, Zhang J (2017) Hydrocephalus after subarachnoid hemorrhage: pathophysiology, diagnosis, and treatment. Biomed Res Int 2017:8584753
Chiang N, Dalli J, Colas RA, Serhan CN (2015) Identification of resolvin D2 receptor mediating resolution of infections and organ protection. J Exp Med 212:1203–1217
Ciurea AV, Palade C, Voinescu D, Nica DA (2013) Subarachnoid hemorrhage and cerebral vasospasm—literature review. J Med Life 6:120–125
Dong X, Gao J, Zhang CY, Hayworth C, Frank M, Wang Z (2019) Neutrophil membrane-derived nanovesicles alleviate inflammation to protect mouse brain injury from ischemic stroke. ACS Nano 13:1272–1283
Dunn SL, Wilkinson JM, Crawford A, Bunning RAD, Le Maitre CL (2016) Expression of cannabinoid receptors in human osteoarthritic cartilage: implications for future therapies. Cannabis Cannabinoid Res 1:3–15
Echigo R, Shimohata N, Karatsu K, Yano F, Kayasuga-Kariya Y, Fujisawa A, Ohto T, Kita Y et al (2012) Trehalose treatment suppresses inflammation, oxidative stress, and vasospasm induced by experimental subarachnoid hemorrhage. J Transl Med 10:80
Eisenhut M (2014) Vasospasm in cerebral inflammation. Int J Inflam 2014:1–14
Finlay DB, Joseph WR, Grimsey NL, Glass M (2016) GPR18 undergoes a high degree of constitutive trafficking but is unresponsive to N-arachidonoyl glycine. Peer J 4:e1835
Guerrero-Alba R, Barragan-Iglesias P, Gonzalez-Hernandez A, Valdez-Morales EE, Granados-Soto V, Condes-Lara M, Rodriguez MG, Marichal-Cancino BA (2018) Some prospective alternatives for treating pain: the endocannabinoid system and its putative receptors GPR18 and GPR55. Front Pharmacol 9:1496
Ishikawa Y, Deyama S, Shimoda K, Yoshikawa K, Ide S, Satoh M, Minami M (2017) Rapid and sustained antidepressant effects of resolvin D1 and D2 in a chronic unpredictable stress model. Behav Brain Res 332:233–236
Khey KMW, Huard A, Mahmoud SH (2020) Inflammatory pathways following subarachnoid hemorrhage. Cell Mol Neurobiol 40:675–693
Leitner H (2010) Influence of neurosteroids on the pathogenesis of multiple sclerosis. Med Hypotheses 75:229–234
Liu Y, Cai H, Wang Z, Li J, Wang K, Yu Z, Chen G (2013) Induction of autophagy by cystatin C: a potential mechanism for prevention of cerebral vasospasm after experimental subarachnoid hemorrhage. Eur J Med Res 18:21
Ma L, Jiang Y, Dong Y, Gao J, Du B, Liu D (2018) Anti-TNF-alpha antibody attenuates subarachnoid hemorrhage-induced apoptosis in the hypothalamus by inhibiting the activation of Erk. Neuropsychiatr Dis Treat 14:525–536
Macdonald RL, Schweizer TA (2017) Spontaneous subarachnoid haemorrhage. Lancet 389:655–666
Orru E, Roccatagliata L, Cester G, Causin F, Castellan L (2013) Complications of endovascular treatment of cerebral aneurysms. Eur J Radiol 82:1653–1658
Pang J, Peng J, Matei N, Yang P, Kuai L, Wu Y, Chen L, Vitek MP et al (2018) Apolipoprotein E exerts a whole-brain protective property by promoting M1? Microglia quiescence after experimental subarachnoid hemorrhage in mice. Transl Stroke Res 9:654–668
Pang J, Peng J, Yang P, Kuai L, Chen L, Zhang JH, Jiang Y (2019) White matter injury in early brain injury after subarachnoid hemorrhage. Cell Transpl 28:26–35
Parrella E, Porrini V, Benarese M, Pizzi M (2019) The Role of mast cells in stroke. Cells. https://doi.org/10.3390/cells8050437
Pascoal LB, Bombassaro B, Ramalho AF, Coope A, Moura RF, Correa-da-Silva F, Ignacio-Souza L, Razolli D et al (2017) Resolvin RvD2 reduces hypothalamic inflammation and rescues mice from diet-induced obesity. J Neuroinflammation 14:5
Rass V, Helbok R (2019) Early brain injury after poor-grade subarachnoid hemorrhage. Curr Neurol Neurosci Rep 19:78
Shetty AK, Zanirati G (2020) The interstitial system of the brain in health and disease. Aging Dis 11:200–211
Siddiqui YD, Omori K, Ito T, Yamashiro K, Nakamura S, Okamoto K, Ono M, Yamamoto T et al (2019) Resolvin D2 induces resolution of periapical inflammation and promotes healing of periapical lesions in rat periapical periodontitis. Front Immunol 10:307
Strbian D, Kovanen PT, Karjalainen-Lindsberg ML, Tatlisumak T, Lindsberg PJ (2009) An emerging role of mast cells in cerebral ischemia and hemorrhage. Ann Med 41:438–450
Topkoru B, Egemen E, Solaroglu I, Zhang JH (2017) Early brain injury or vasospasm? An overview of common mechanisms. Curr Drug Targets 18:1424–1429
Tulowiecka N, Kotlega D, Prowans P, Szczuko M (2020) The role of resolvins: EPA and DHA derivatives can be useful in the prevention and treatment of ischemic stroke. Int J Mol Sci. https://doi.org/10.3390/ijms21207628
Yin J, Li H, Meng C, Chen D, Chen Z, Wang Y, Wang Z, Chen G (2016) Inhibitory effects of omega-3 fatty acids on early brain injury after subarachnoid hemorrhage in rats: possible involvement of G protein-coupled receptor 120/beta-arrestin2/TGF-beta activated kinase-1 binding protein-1 signaling pathway. Int J Biochem Cell Biol 75:11–22
Zhang YL, Zhang PB, Qiu SD, Liu Y, Tian YF, Wang Y (2006) Effects of ketamine-midazolam anesthesia on the expression of NMDA and AMPA receptor subunit in the peri-infarction of rat brain. Chin Med J 119:1555–1562
Zhang T, Wu P, Zhang JH, Li Y, Xu S, Wang C, Wang L, Zhang G et al (2018) Docosahexaenoic acid alleviates oxidative stress-based apoptosis via improving mitochondrial dynamics in early brain injury after subarachnoid hemorrhage. Cell Mol Neurobiol 38:1413–1423
Zhang LY, Liu ZH, Zhu Q, Wen S, Yang CX, Fu ZJ, Sun T (2018) Resolvin D2 relieving radicular pain is associated with regulation of inflammatory mediators, Akt/GSK-3beta signal pathway and GPR18. Neurochem Res 43:2384–2392
Zhang T, Xu S, Wu P, Zhou K, Wu L, Xie Z, Xu W, Luo X et al (2019) Mitoquinone attenuates blood-brain barrier disruption through Nrf2/PHB2/OPA1 pathway after subarachnoid hemorrhage in rats. Exp Neurol 317:1–9
Zhang T, Wu P, Budbazar E, Zhu Q, Sun C, Mo J, Peng J, Gospodarev V et al (2019) Mitophagy reduces oxidative stress via keap1 (kelch-like epichlorohydrin-associated protein 1)/Nrf2 (nuclear factor-E2-related factor 2)/PHB2 (prohibitin 2) pathway after subarachnoid hemorrhage in rats. Stroke 50:978–988
Zuo G, Zhang D, Mu R, Shen H, Li X, Wang Z, Li H, Chen G (2018) Resolvin D2 protects against cerebral ischemia/reperfusion injury in rats. Mol Brain 11:9
Zuo G, Zhang T, Huang L, Araujo C, Peng J, Travis Z, Okada T, Ocak U, Zhang G, Tang J, Lu X, Zhang JH (2019) Activation of TGR5 with INT-777 attenuates oxidative stress and neuronal apoptosis via cAMP/PKCε/ALDH2 pathway after subarachnoid hemorrhage in rats. Free Radic Biol Med 143:441–453. https://doi.org/10.1016/j.freeradbiomed.2019.09.002
Funding
This research was supported by grants from the China Postdoctoral Science Foundation to TY Zhang (2019TQ0216 and 2019M660716), the Beijing Postdoctoral Science Foundation to TY Zhang (2020-ZZ-020), the National Natural Science Foundation of China to Dr. TY Zhang (82001324). And the Jiangsu Provincial Health and Family Planning Commission of China to G Zuo (QNRC2016263, H201654, and GSWS2019080).
Author information
Authors and Affiliations
Contributions
TZh designed the study, performed the experiments and wrote the manuscript. GZ contributed to data analysis. HZh contributed to research design. All authors analyzed the results and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no potential conflicts of interest with respect to the research, authorship, or publication of this article.
Research Involving Human and Animal Participants
All animal procedures were conducted in accordance with the ethical standards of our institution and with national regulations.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: Figure 8 has been corrected.
Supplementary Information
Below is the link to the electronic supplementary material.
10571_2021_1114_MOESM1_ESM.tif
Supplementary file1 Fig. SI Photographs of representative preparations of four brain regions, for subsequent analysis by Western blot (TIF 1232 kb)
10571_2021_1114_MOESM2_ESM.tif
Supplementary file2 Fig. SII Colocalization of GPR18 (green) with markers (red) of (A-B) mast cells (chymase, tryptase) or (C) microglia (Iba1). Colocalization was not observed with microglia. The results shown are representative of those obtained with three animals from each group. Scale bars = 50 µm (TIF 2437 kb)
10571_2021_1114_MOESM3_ESM.tif
Supplementary file3 Fig. SIII RvD2 improved neurological scores of SAH rats. (A) Modified Garcia and (B) beam balance scores. Bars represent mean ± SD (n = 6 from each group). # P < 0.05 vs. Sham + vehicle group; * P < 0.05 vs. SAH + vehicle group (TIF 937 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, T., Zuo, G. & Zhang, H. GPR18 Agonist Resolvin D2 Reduces Early Brain Injury in a Rat Model of Subarachnoid Hemorrhage by Multiple Protective Mechanisms. Cell Mol Neurobiol 42, 2379–2392 (2022). https://doi.org/10.1007/s10571-021-01114-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-021-01114-2