Abstract
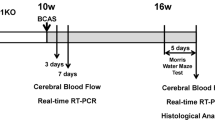
Smoking is a risk factor for dementia. Cognitive function can be partially restored after quitting smoking, but still lower than never smoked group. The underlying mechanisms still remain unclear. The effects of smoking cessation combined with cerebral chronic hypoperfusion (CCH) on cognitive function have never been described. Here, we established a cigarette smoking cessation model, a CCH model, and a cigarette smoking cessation plus CCH model. We investigated cognitive function in these models and the mechanisms of the neuroinflammation, nucleotide-binding oligomerization domain-like receptor family pyrin domain-containing 3(NLRP3)/cysteine aspartate-specific proteinase (caspase-1)/interleukin- 1β (IL-1β) pathway, and eucaryotic initiation factor 2α (eIF2α) /autophagy pathway. We used morris water maze (MWM) and novel object recognition (NOR) test to evaluate cognitive function in rats. Nissl staining was performed to observe cell morphology in the hippocampal CA1 area. A neuroinflammatory marker (glial fibrillary acidic protein, GFAP) was assessed by Western blot analysis and immunohistochemistry staining. IL-1β levels were detected by ELISA. The protein levels of NLRP3/caspase-1/ IL-1β and eIF2α/autophagy pathway were evaluated by Western blot analysis. LC3 was assessed by immunofluorescence staining. CCH can affect cognitive function by influencing neuroinflammation, NLRP3/caspase-1/IL-1β pathway, and eIF2α/autophagy pathway. Past exposure to cigarette smoke can also affect cognitive function by influencing neuroinflammation and NLRP3/caspase-1/IL-1β pathway, which may be induced by smoking and may not be alleviated after smoking cessation. Past exposure to cigarette smoke does not influence autophagy, which may be increased by smoking and then decrease to normal levels after smoking cessation. Past exposure to smoking can further aggravate cognitive impairment and neuroinflammation in VaD animals: cognitive impairment induced by CCH via neuroinflammation, NLRP3/caspase-1/IL-1β, and eIF2α/autophagy pathway and cognitive impairment induced by past exposure to cigarette smoke via neuroinflammation and NLRP3/caspase-1/IL-1β pathway. The combined group had the worst cognitive impairment because of harmful reasons.






Similar content being viewed by others
Availability of Data and Material
The data used in this article are available to researchers subject to confidentiality if necessary.
References
Alrouji M, Manouchehrinia A, Gran B, Constantinescu CS (2019) Effects of cigarette smoke on immunity, neuroinflammation and multiple sclerosis. J Neuroimmunol 329:24–34. https://doi.org/10.1016/j.jneuroim.2018.10.004
Ambrogio F, Martella LA, Odetti P, Monacelli F (2019) Behavioral disturbances in dementia and beyond: time for a new conceptual frame? Int J Mol Sci 20:3647. https://doi.org/10.3390/ijms20153647
Amorim BC, Pereira-Latini AC, Golim MDA, Júnior RLR, Yoo HHB, Arruda MSPD, Tavares AH, Cavalcante RDS, Mendes RP, Pontillo A, Venturini J (2019) Enhanced expression of NLRP3 inflammasome components by monocytes of patients with pulmonary paracoccidioidomycosis is associated with smoking and intracellular hypoxemia. Microbes Infect 22:137–143. https://doi.org/10.1016/j.micinf.2019.11.001
Ardura-Fabregat A, Boddeke E, Boza-Serrano A et al (2017) Targeting neuroinflammation to treat Alzheimer’s disease. CNS Drugs 31:1057–1082. https://doi.org/10.1007/s40263-017-0483-3
Berg CJ, Fong GT, Thrasher JF, Cohen JE, Maziak W, Lando H, Drope J, Mejia R, Barnoya J, Nakkash R, Salloum RG, Parascandola M (2018) The impact and relevance of tobacco control research in low-and middle-income countries globally and to the US. Addict Behav 87:162–168. https://doi.org/10.1016/j.addbeh.2018.07.012
Bi Y, Yuan K, Yu D, Wang R, Li M, Li Y, Zhai J, Lin W, Tian J (2017) White matter integrity of central executive network correlates with enhanced brain reactivity to smoking cues. Hum Brain Mapp 38:6239–6249. https://doi.org/10.1002/hbm.23830
Bond S, Lopez-Lloreda C, Gannon PJ, Akay-Espinoza C, Jordan-Sciutto KL (2020) The integrated stress response and phosphorylated eukaryotic initiation factor 2α in neurodegeneration. J Neuropathol Exp Neurol 79:123–143. https://doi.org/10.1093/jnen/nlz129
Cai M, Lee JH, Yang EJ (2017) Bee venom ameliorates cognitive dysfunction caused by neuroinflammation in an animal model of vascular dementia. Mol Neurobiol 54:5952–5960. https://doi.org/10.1007/s12035-016-0130-x
Chen W, Wang Q, Xu X, Saxton B, Tessema M, Leng S, Choksi S, Belinsky SA, Liu Z-G, Lin Y (2020) Vasorin/ATIA promotes cigarette smoke–induced transformation of human bronchial epithelial cells by suppressing autophagy-mediated apoptosis. Transl Oncol 13:32–41. https://doi.org/10.1016/j.tranon.2019.09.001
Choi D, Choi S, Park SM (2018) Effect of smoking cessation on the risk of dementia: a longitudinal study. Ann Clin Transl Neurol 5:1192–1199. https://doi.org/10.1002/acn3.633
Deal JA, Power MC, Palta P, Alonso A, Schneider ALC, Perryman K, Bandeen-Roche K, Sharrett AR (2020) Relationship of cigarette smoking and time of quitting with incident dementia and cognitive decline. J Am Geriatr Soc 68:337–345. https://doi.org/10.1111/jgs.16228
Depp CA, Bowie CR, Mausbach BT, Wolyniec P, Thornquist MH, Luke JR, McGrath JA, Pulver AE, Patterson TL, Harvey PD (2015) Current smoking is associated with worse cognitive and adaptive functioning in serious mental illness. Acta Psychiatr Scand 131:333–341. https://doi.org/10.1111/acps.12380
Deretic V, Levine B (2009) Autophagy, immunity, and microbial adaptations. Cell Host Microbe 5:527–549. https://doi.org/10.1016/j.chom.2009.05.016
Deretic V, Levine B (2018) Autophagy balances inflammation in innate immunity. Autophagy 14:243–251. https://doi.org/10.1080/15548627.2017.1402992
Gao C, Cai Y, Zhang X, Huang H, Wang J, Wang Y, Tong X, Wang J, Wu J (2015) Ischemic preconditioning mediates neuroprotection against ischemia in mouse hippocampal CA1 neurons by inducing autophagy. PLoS ONE 10:e0137146. https://doi.org/10.1371/journal.pone.0137146
Gellner CA, Reynaga DD, Leslie FM (2016) Cigarette smoke extract: a preclinical model of tobacco dependence. Curr Protoc Neurosci 77:9.54.1-9.54.10. https://doi.org/10.1002/cpns.14
Gorgun MF, Zhuo M, Dineley KT, Englander EW (2019) Elevated neuroglobin lessens neuroinflammation and alleviates neurobehavioral deficits induced by acute inhalation of combustion smoke in the mouse. Neurochem Res 44:2170–2181. https://doi.org/10.1007/s11064-019-02856-8
Hall FS, Der-Avakian A, Gould TJ, Markou A, Shoaib M, Young JW (2015) Negative affective states and cognitive impairments in nicotine dependence. Neurosci Biobehav Rev 58:168–185. https://doi.org/10.1016/j.neubiorev.2015.06.004
Halle A, Hornung V, Petzold GC, Stewart CR, Monks BG, Reinheckel T, Fitzgerald KA, Latz E, Moore KJ, Golenbock DT (2008) The NALP3 inflammasome is involved in the innate immune response to amyloid-beta. Nat Immunol 9:857–865. https://doi.org/10.1038/ni.1636
He B, Chen Q, Zhou D, Wang L, Liu Z (2019) Role of reciprocal interaction between autophagy and endoplasmic reticulum stress in apoptosis of human bronchial epithelial cells induced by cigarette smoke extract. IUBMB Life 71:66–80. https://doi.org/10.1002/iub.1937
Houtman J, Freitag K, Gimber N, Schmoranzer J, Heppner FL, Jendrach M (2019) Beclin1-driven autophagy modulates the inflammatory response of microglia via NLRP3. EMBO J 38:e99430. https://doi.org/10.15252/embj.201899430
Huang CY, Kuo WW, Liao HE, Lin YM, Kuo CH, Tsai FJ, Tsai CH, Chen JL, Lin JY (2013) Lumbrokinase attenuates side-stream-smoke-induced apoptosis and autophagy in young hamster hippocampus: correlated with eNOS induction and NFkappaB/iNOS/COX-2 signaling suppression. Chem Res Toxicol 26:654–661. https://doi.org/10.1021/tx300429s
Huo T, Jia Y, Yin C, Luo X, Zhao J, Wang Z, Lv P (2019) Iron dysregulation in vascular dementia: focused on the AMPK/autophagy pathway. Brain Res Bull 153:305–313. https://doi.org/10.1016/j.brainresbull.2019.09.006
Jiang X, Niu X, Guo Q, Dong Y, Xu J, Yin N, Qi Q, Jia Y, Gao L, He Q, Lv P (2019) FoxO1-mediated autophagy plays an important role in the neuroprotective effects of hydrogen in a rat model of vascular dementia. Behav Brain Res 356:98–106. https://doi.org/10.1016/j.bbr.2018.05.023
Kaplan HM, Kuyucu Y, Polat S, Pazarci P, Yegani AA, Singirik E, Ertug P (2018) Molecular basis of vascular damage caused by cigarette smoke exposure and a new approach to the treatment: α-linolenic acid. Biomed Pharmacother 102:458–463. https://doi.org/10.1016/j.biopha.2018.03.112
Khanna A, Guo M, Mehra M, Royal W 3rd (2013) Inflammation and oxidative stress induced by cigarette smoke in Lewis rat brains. J Neuroimmunol 254:69–75. https://doi.org/10.1016/j.jneuroim.2012.09.006
Lamark T, Svenning S, Johansen T (2017) Regulation of selective autophagy: the p62/SQSTM1 paradigm. Essays Biochem 61:609–624. https://doi.org/10.1042/EBC20170035
Li H, Li X, Gao S, Wang D, Gao X, Li Y, Wang X, Cui Z, Ma H, Liu Q, Li M (2019) Exposure to cigarette smoke augments post-ischemic brain injury and inflammation via mobilization of neutrophils and monocytes. Front Immunol 10:2576. https://doi.org/10.3389/fimmu.2019.02576
Liu B, Gao JM, Li F, Gong QH, Shi JS (2018) Gastrodin attenuates bilateral common carotid artery occlusion-induced cognitive deficits via regulating abeta-related proteins and reducing autophagy and apoptosis in rats. Front Pharmacol 9:405. https://doi.org/10.3389/fphar.2018.00405
Messner B, Bernhard D (2014) Smoking and cardiovascular disease: mechanisms of endothelial dysfunction and early atherogenesis. Arterioscler Thromb Vasc Biol 34:509–515. https://doi.org/10.1161/ATVBAHA.113.300156
Mo Y, Sun YY, Liu KY (2020) Autophagy and inflammation in ischemic stroke. Neural Regen Res 15:1388–1396. https://doi.org/10.4103/1673-5374.274331
Moon SL, Sonenberg N, Parker R (2018) Neuronal regulation of eIF2α function in health and neurological disorders. Trends Mol Med 24:575–589. https://doi.org/10.1016/j.molmed.2018.04.001
Morsch A, Wisniewski E, Luciano TF, Comin VH, Silveira GB, Marques SO, Thirupathi A, Lock PCS, De Souza CT (2019) Cigarette smoke exposure induces ROS-mediated autophagy by regulating sestrin, AMPK, and mTOR level in mice. Redox Rep 24:27–33. https://doi.org/10.1080/13510002.2019.1601448
Niu XL, Jiang X, Xu GD, Zheng GM, Tang ZP, Yin N, Li XQ, Yang YY, Lv PY (2019) DL-3-n-butylphthalide alleviates vascular cognitive impairment by regulating endoplasmic reticulum stress and the Shh/Ptch1 signaling-pathway in rats. J Cell Physiol 234:12604–12614. https://doi.org/10.1002/jcp.27332
Pirzada RH, Javaid N, Choi S (2020) The roles of the NLRP3 inflammasome in neurodegenerative and metabolic diseases and in relevant advanced therapeutic interventions. Genes 11:131. https://doi.org/10.3390/genes11020131
Quinteros DA, Hansen AW, Bellaver B, Bobermin LD, Pulcinelli R, Bandiera S, Caletti G, Bitencourt PER, Quincozes-Santos A, Gomez R (2019) Combined exposure to alcohol and tobacco smoke changes oxidative, inflammatory, and neurotrophic parameters in different areas of the brains of rats. ACS Chem Neurosci 10:1336–1346. https://doi.org/10.1021/acschemneuro.8b00412
Reddy AT, Lakshmi SP, Banno A, Jadhav SK, Kadamberi IP, Kim SC, Reddy RC (2020) Cigarette smoke downregulates Nur77 to exacerbate inflammation in chronic obstructive pulmonary disease (COPD). PLoS ONE 15:e0229256. https://doi.org/10.1371/journal.pone.0229256
Saitoh T, Akira S (2016) Regulation of inflammasomes by autophagy. J Allergy Clin Immunol 138:28–36. https://doi.org/10.1016/j.jaci.2016.05.009
Saravia R, Ten-Blanco M, Grande MT, Maldonado R, Berrendero F (2019) Anti-inflammatory agents for smoking cessation? Focus on cognitive deficits associated with nicotine withdrawal in male mice. Brain Behav Immun 75:228–239. https://doi.org/10.1016/j.bbi.2018.11.003
Shimada K, Crother TR, Karlin J, Chen S, Chiba N, Ramanujan VK, Vergnes L, Ojcius DM, Arditi M (2011) Caspase-1 dependent IL-1beta secretion is critical for host defense in a mouse model of Chlamydia pneumoniae lung infection. PLoS ONE 6:e21477. https://doi.org/10.1371/journal.pone.0021477
Sprenkle NT, Sims SG, Sanchez CL, Meares GP (2017) Endoplasmic reticulum stress and inflammation in the central nervous system. Mol Neurodegener 12:42. https://doi.org/10.1186/s13024-017-0183-y
Su SH, Wu YF, Lin Q, Wang DP, Hai J (2019) URB597 protects against NLRP3 inflammasome activation by inhibiting autophagy dysfunction in a rat model of chronic cerebral hypoperfusion. J Neuroinflamm 16:260. https://doi.org/10.1186/s12974-019-1668-0
Wang D, Zhang J, Jiang W, Cao Z, Zhao F, Cai T, Aschner M, Luo W (2017) The role of NLRP3-CASP1 in inflammasome-mediated neuroinflammation and autophagy dysfunction in manganese-induced, hippocampal-dependent impairment of learning and memory ability. Autophagy 13:914–927. https://doi.org/10.1080/15548627.2017.1293766
Wek RC (2018) Role of eIF2α kinases in translational control and adaptation to cellular stress. Cold Spring Harb Perspect Biol 10:a032870. https://doi.org/10.1101/cshperspect.a032870
Xu H, Boychuk JA, Boychuk CR, Uteshev VV, Smith BN (2015) Nicotine enhances inhibition of mouse vagal motor neurons by modulating excitability of premotor GABAergic neurons in the nucleus tractus solitarii. J Neurophysiol 113:1165–1174. https://doi.org/10.1152/jn.00614.2014
Zhang X, Zhu LB, He JH, Zhang HQ, Ji SY, Zhang CN, Hou NN, Huang CP, Zhu JH (2020) Paroxetine suppresses reactive microglia-mediated but not lipopolysaccharide-induced inflammatory responses in primary astrocytes. J Neuroinflammation 17:50. https://doi.org/10.1186/s12974-020-1712-0
Zhao J, Zhang X, Dong L, Wen Y, Zheng X, Zhang C, Chen R, Zhang Y, Li Y, He T, Zhu X, Li L (2015) Cinnamaldehyde inhibits inflammation and brain damage in a mouse model of permanent cerebral ischaemia. Br J Pharmacol 172:5009–5023. https://doi.org/10.1111/bph.13270
Acknowledgements
Thanks to the Department of Pathophysiology of Hebei Medical university for providing some experimental sites.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
NM and PYL conceived and designed this work. NM performed the main experiments. MYS worked on the cigarette smoke exposure. YHD, TTH, XJ, and YNX analyzed the data. NM wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethics Approval
All animal care and experimental procedures were approved by the Animal Care and Management Committee of Hebei General Hospital (Permit Number SCXK20160006).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Meng, N., Dong, Y., Huo, T. et al. Past Exposure to Cigarette Smoke Aggravates Cognitive Impairment in a Rat Model of Vascular Dementia via Neuroinflammation. Cell Mol Neurobiol 42, 1021–1034 (2022). https://doi.org/10.1007/s10571-020-00992-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-020-00992-2