Abstract
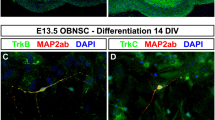
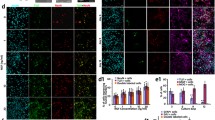
Olfactory ensheathing cells (OECs), the major glia cells in the olfactory system, have been extensively studied because of their ability to promote axonal growth and regeneration. Whether it could facilitate synaptogenesis is an important, but remains as yet an unanswered question. We have identified a subgroup of Wnt signaling-activated OECs, spatiotemporal distribution of which in the olfactory bulb suggests a role for these cells in both axonal growth and synaptogenesis. In the present study, we explored this possibility in vitro. OECs were primarily cultured, in which Wnt signaling was activated by overexpressing β-catenin, and inhibited by dominant negative TCF4. Neurite growth and synaptogenesis were assessed by co-culturing neurons with conditioned medium from control OECs (cOECs CM), Wnt/β-catenin signaling-activated OECs (wOECs CM), or Wnt signaling-inhibited OECs (wiOECs). The results showed that although cOECs CM enhances axonal growth, wOECs CM exhibited a stronger axonal growth-promoting effect, than cOECs CM. More importantly, wOECs CM stimulates synatpogenesis, demonstrated by the expression of Synaptophysin and whole-cell patch clamp recording. In contrast, both cOECs CM and wiOECs CM do not affect synaptogenesis. Our data, for the first time, demonstrated that, in comparison with regularly cultured OECs, wOECs CM are more effective in enhancing axonal growth, and can promote synaptogenesis, probably by secreting factors. These results suggest a potential application of wOECs for treating spinal cord injury.




Similar content being viewed by others
References
Au E, Roskams AJ (2003) Olfactory ensheathing cells of the lamina propria in vivo and in vitro. Glia 41(3):224–236. doi:10.1002/glia.10160
Au WW, Treloar HB, Greer CA (2002) Sublaminar organization of the mouse olfactory bulb nerve layer. J Comp Neurol 446(1):68–80
Booker-Dwyer T, Hirsh S, Zhao H (2008) A unique cell population in the mouse olfactory bulb displays nuclear beta-catenin signaling during development and olfactory sensory neuron regeneration. Dev Neurobiol 68(7):859–869. doi:10.1002/dneu.20606
Budnik V, Salinas PC (2011) Wnt signaling during synaptic development and plasticity. Curr Opin Neurobiol 21(1):151–159. doi:10.1016/j.conb.2010.12.002
Chung RS, Woodhouse A, Fung S, Dickson TC, West AK, Vickers JC, Chuah MI (2004) Olfactory ensheathing cells promote neurite sprouting of injured axons in vitro by direct cellular contact and secretion of soluble factors. Cell Mol Life Sci 61(10):1238–1245. doi:10.1007/s00018-004-4026-y
Faissner A, Pyka M, Geissler M, Sobik T, Frischknecht R, Gundelfinger ED, Seidenbecher C (2010) Contributions of astrocytes to synapse formation and maturation—potential functions of the perisynaptic extracellular matrix. Brain Res Rev 63(1–2):26–38. doi:10.1016/j.brainresrev.2010.01.001
Franssen EH, de Bree FM, Verhaagen J (2007) Olfactory ensheathing glia: their contribution to primary olfactory nervous system regeneration and their regenerative potential following transplantation into the injured spinal cord. Brain Res Rev 56(1):236–258. doi:10.1016/j.brainresrev.2007.07.013
Fuerer C, Nusse R (2010) Lentiviral vectors to probe and manipulate the Wnt signaling pathway. PLoS ONE 5(2):e9370. doi:10.1371/journal.pone.0009370
Grigoryan T, Wend P, Klaus A, Birchmeier W (2008) Deciphering the function of canonical Wnt signals in development and disease: conditional loss- and gain-of-function mutations of beta-catenin in mice. Genes Dev 22(17):2308–2341. doi:10.1101/gad.1686208
Li Y, Field PM, Raisman G (1997) Repair of adult rat corticospinal tract by transplants of olfactory ensheathing cells. Science 277(5334):2000–2002
Lu P, Yang H, Culbertson M, Graham L, Roskams AJ, Tuszynski MH (2006) Olfactory ensheathing cells do not exhibit unique migratory or axonal growth-promoting properties after spinal cord injury. J Neurosci 26(43):11120–11130. doi:10.1523/JNEUROSCI.3264-06.2006
Michaelidis TM, Lie DC (2008) Wnt signaling and neural stem cells: caught in the Wnt web. Cell Tissue Res 331(1):193–210. doi:10.1007/s00441-007-0476-5
Murdoch B, Roskams AJ (2008) A novel embryonic nestin-expressing radial glia-like progenitor gives rise to zonally restricted olfactory and vomeronasal neurons. J Neurosci 28(16):4271–4282. doi:10.1523/JNEUROSCI.5566-07.2008
Pellitteri R, Spatuzza M, Russo A, Stanzani S (2007) Olfactory ensheathing cells exert a trophic effect on the hypothalamic neurons in vitro. Neurosci Lett 417(1):24–29. doi:10.1016/j.neulet.2007.02.065
Pellitteri R, Spatuzza M, Stanzani S, Zaccheo D (2010) Biomarkers expression in rat olfactory ensheathing cells. Front Biosci (Sch Ed) 2:289–298
Ramon-Cueto A, Avila J (1998) Olfactory ensheathing glia: properties and function. Brain Res Bull 46(3):175–187
Ramon-Cueto A, Cordero MI, Santos-Benito FF, Avila J (2000) Functional recovery of paraplegic rats and motor axon regeneration in their spinal cords by olfactory ensheathing glia. Neuron 25(2):425–435
Salinas PC, Zou Y (2008) Wnt signaling in neural circuit assembly. Annu Rev Neurosci 31:339–358. doi:10.1146/annurev.neuro.31.060407.125649
Su Z, He C (2010) Olfactory ensheathing cells: biology in neural development and regeneration. Prog Neurobiol 92(4):517–532. doi:10.1016/j.pneurobio.2010.08.008
Wang YZ, Meng JH, Yang H, Luo N, Jiao XY, Ju G (2003) Differentiation-inducing and protective effects of adult rat olfactory ensheathing cell conditioned medium on PC12 cells. Neurosci Lett 346(1–2):9–12
Wang YZ, Molotkov A, Song L, Li Y, Pleasure DE, Zhou CJ (2008) Activation of the Wnt/beta-catenin signaling reporter in developing mouse olfactory nerve layer marks a specialized subgroup of olfactory ensheathing cells. Dev Dyn 237(11):3157–3168. doi:10.1002/dvdy.21712
Wang T, Cong R, Yang H, Wu MM, Luo N, Kuang F, You SW (2011) Neutralization of BDNF attenuates the in vitro protective effects of olfactory ensheathing cell-conditioned medium on scratch-insulted retinal ganglion cells. Cell Mol Neurobiol 31(3):357–364. doi:10.1007/s10571-010-9626-5
Windus LC, Lineburg KE, Scott SE, Claxton C, Mackay-Sim A, Key B, St John JA (2010) Lamellipodia mediate the heterogeneity of central olfactory ensheathing cell interactions. Cell Mol Life Sci 67(10):1735–1750. doi:10.1007/s00018-010-0280-3
Yi H, Hu J, Qian J, Hackam AS (2012) Expression of brain-derived neurotrophic factor is regulated by the Wnt signaling pathway. NeuroReport 23(3):189–194. doi:10.1097/WNR.0b013e32834fab06
Zaghetto AA, Paina S, Mantero S, Platonova N, Peretto P, Bovetti S, Puche A, Piccolo S, Merlo GR (2007) Activation of the Wnt-beta catenin pathway in a cell population on the surface of the forebrain is essential for the establishment of olfactory axon connections. J Neurosci 27(36):9757–9768. doi:10.1523/JNEUROSCI.0763-07.2007
Acknowledgments
This study was mainly supported by the Natural Science Foundation of China (# 30900712 to Dr. Yin Wu, and # 31271583 to Dr. Yazhou Wang) and partially by projects for the science development of Shaanxi Province (2012KW-34 to Dr. Lianhe Zheng).
Conflict of interest
The authors claim no conflict of interests.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Zhenyu Yang, Yin Wu and Lianhe Zheng contributed equally to this work.
Rights and permissions
About this article
Cite this article
Yang, Z., Wu, Y., Zheng, L. et al. Conditioned Medium of Wnt/β-Catenin Signaling-Activated Olfactory Ensheathing Cells Promotes Synaptogenesis and Neurite Growth In Vitro. Cell Mol Neurobiol 33, 983–990 (2013). https://doi.org/10.1007/s10571-013-9966-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-013-9966-z