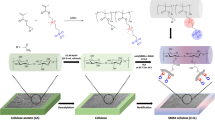
Abstract
Cellulose phenyl carbamates such as cellulose tris(3,5-dimethylphenyl carbamate) (CDMPC) are known to act as chiral selectors when they are coated or immobilized on silica gels that are packed in the columns for high-performance liquid chromatography (HPLC). Previously, we reported a proof of concept of enantioselective membrane filtration using a nonwoven membrane prepared by electrospinning of the CDMPC synthesized from microcrystalline cellulose Avicel®. In the present work, TECHNOCEL® cellulose fiber samples derived from hardwood are functionalized to synthesize CDMPCs having higher and different degrees of polymerization. The length and width of the cellulose fiber samples are characterized using a MorFi image analyzer. The obtained CDMPCs are electrospun to form nanofibrous membranes and their morphology is studied in relation to the electrospinning process parameters, the degrees of polymerization of the CDMPCs, and their solution concentrations. Liquid-liquid permeation experiments of a racemic compound, (R,S)-1-(1-naphthyl)ethanol, through the CDMPC membranes demonstrate preferable permeation of the (S)-enantiomer. This is supported by theoretical simulations using a molecular docking model that indicates stronger hydrogen bonding and π–π interactions with higher binding energy between CDMPC and the (R)-enantiomer than the (S)-enantiomer.
Graphical abstract













Similar content being viewed by others
Data availability
Not applicable.
References
Adhikari S, Bhujbal S, Paik M-J, Lee W (2023) Enantioseparation and molecular modeling study of chiral amines as three naphthaldimine derivatives using amylose or cellulose trisphenylcarbamate chiral. Station Ph Chirality 35:29–39. https://doi.org/10.1002/chir.23513
Ahmadi-Nohadani H, Nono-Tagne S, Barrett CJ, Otsuka I (2022) Electrospun azo-cellulose fabric: a smart polysaccharidic photo-actuator. Macromol Rapid Commun 43:2200063. https://doi.org/10.1002/marc.202200063
Ahn K, Zaccaron S, Rosenau T, Potthast A (2019) How alkaline solvents in viscosity measurements affect data for oxidatively damaged celluloses: cupri-ethylenediamine. Biomacromolecules 20:4117–4125. https://doi.org/10.1021/acs.biomac.9b00956
Brunauer S, Emmett PH, Teller E (1938) Adsorption of gases in multimolecular layers. J Am Chem Soc 60:309–319. https://doi.org/10.1021/ja01269a023
Bui CV, Rosenau T, Hettegger H (2023) Synthesis by carbonate aminolysis and chiral recognition ability of cellulose 2,3-bis(3,5-dimethylphenyl carbamate)-6-(α-phenylethyl carbamate). Sel Cellul 30:153–168. https://doi.org/10.1007/s10570-022-04898-8
Das S, Xu S, Ben T, Qiu S (2018) Chiral recognition and separation by chirality-enriched metal–organic frameworks. Angew Chem Int Ed 57:8629–8633. https://doi.org/10.1002/anie.201804383
Fong H, Chun I, Reneker DH (1999) Beaded nanofibers formed during electrospinning. Polymer 40:4585–4592. https://doi.org/10.1016/S0032-3861(99)00068-3
Fumagalli M, Ouhab D, Boisseau SM, Heux L (2013) Versatile gas-phase reactions for surface to bulk esterification of cellulose. Microfibrils Aerogels Biomacromol 14:3246–3255. https://doi.org/10.1021/bm400864z
Gong W, Chen Z, Dong J, Liu Y, Cui Y (2022) Chiral metal–organic frameworks. Chem Rev 122:9078–9144. https://doi.org/10.1021/acs.chemrev.1c00740
Greiner A, Wendorff JH (2007) Electrospinning: a fascinating method for the preparation of ultrathin fibers. Angew Chem Int Ed 46:5670–5703. https://doi.org/10.1002/anie.200604646
Hwang K, Kwon B, Byun H (2011) Preparation of PVdF nanofiber membranes by electrospinning and their use as secondary battery separators. J Membr Sci 378:111–116. https://doi.org/10.1016/j.memsci.2011.06.005
Ishihara K, Suzuki N, Matsui K (1987) Optical resolution of amino acids by a polymer membrane having cyclodextrin moieties. Nippon Kagaku Kaishi 446–451:446. https://doi.org/10.1246/nikkashi.1987.446
Kasat RB, Wang N-HL, Franses EI (2007) Effects of backbone and side chain on the molecular environments of chiral cavities in polysaccharide-based. Biopolym Biomacromol 8:1676–1685. https://doi.org/10.1021/bm070006h
Kawasaki T, Yoshikawa M (2013) Nanofiber membranes from cellulose triacetate for chiral separation. Desalin Water Treat 51:5080–5088. https://doi.org/10.1080/19443994.2013.768832
Kaya C, Birgül K, Bülbül B (2023) Fundamentals of chirality, resolution, and enantiopure molecule. Synth Methods Chirality 35:4–28. https://doi.org/10.1002/chir.23512
Ke J, Yang K, Bai X, Luo H, Ji Y, Chen J (2021) A novel chiral polyester composite membrane: preparation, enantioseparation of chiral drugs and molecular modeling evaluation. Sep Purif Technol 255:117717. https://doi.org/10.1016/j.seppur.2020.117717
Kim JH, Kim JH, Jegal J, Lee K-H (2003) Optical resolution of α-amino acids through enantioselective polymeric membranes based on polysaccharides. J Membr Sci 213:273–283. https://doi.org/10.1016/S0376-7388(02)00534-3
Lee H, Nishino M, Sohn D, Lee JS, Kim IS (2018) Control of the morphology of cellulose acetate nanofibers via electrospinning. Cellulose 25:2829–2837. https://doi.org/10.1007/s10570-018-1744-0
Li Y, Liu D, Wang P, Zhou Z (2010) Computational study of enantioseparation by amylose tris(3,5-dimethylphenylcarbamate)-based chiral stationary phase. J Sep Sci 33:3245–3255. https://doi.org/10.1002/jssc.201000266
Liu T, Li Z, Wang J, Chen J, Guan M, Qiu H (2021) Solid membranes for chiral separation: a review. Chem Eng J 410:128247. https://doi.org/10.1016/j.cej.2020.128247
Luo H, Bai X, Liu H, Qiu X, Chen J, Ji Y (2022) β-Cyclodextrin covalent organic framework modified-cellulose acetate membranes for enantioseparation of chiral drugs separation. Purif Technol 285:120336. https://doi.org/10.1016/j.seppur.2021.120336
Maida J (2021) Chiral chemicals market by application and geography-forecast and analysis 2021–2025. Technavio company web page. https://www.technavio.com/report/chiral-chemicals-market-industry-analysis. Accessed 16 Feb 2023
Missoum K, Belgacem MN, Barnes J-P, Brochier-Salon M-C, Bras J (2012) Nanofibrillated cellulose surface grafting in ionic liquid. Soft Matter 8:8338–8349. https://doi.org/10.1039/C2SM25691F
Mizushima H, Yoshikawa M, Li N, Robertson GP, Guiver MD (2012) Electrospun nanofiber membranes from polysulfones with chiral selector aimed for optical resolution. Eur Polym J 48:1717–1725. https://doi.org/10.1016/j.eurpolymj.2012.07.003
Motamedi AS, Mirzadeh H, Hajiesmaeilbaigi F, Bagheri-Khoulenjani S, Shokrgozar M (2017) Effect of electrospinning parameters on morphological properties of PVDF nanofibrous scaffolds. Prog Biomater 6:113–123. https://doi.org/10.1007/s40204-017-0071-0
Okada Y, Yamamoto C, Kamigaito M, Gao Y, Shen J, Okamoto Y (2016) Enantioseparation using cellulose tris(3,5-dimethylphenylcarbamate) as chiral stationary phase for HPLC. Influ Mol Weight Cellul Mol 21:1484
Okamoto Y, Ikai T (2008) Chiral HPLC for efficient resolution of enantiomers. Chem Soc Rev 37:2593–2608. https://doi.org/10.1039/B808881K
Okamoto Y, Kawashima M, Hatada K (1986) Chromatographic resolution: XI. controlled chiral recognition of cellulose triphenylcarbamate derivatives supported on silica gel. J Chromatogr A 363:173–186. https://doi.org/10.1016/S0021-9673(01)83736-5
Okamoto Y, Yashima E (1998) Polysaccharide derivatives for chromatographic separation of enantiomers Angew Chem Int Ed 37:1020–1043.
Otsuka I, Pandey K, Ahmadi-Nohadani H, Nono-Tagne S (2021) Electrospun cellulosic membranes toward efficient chiral resolutions via enantioselective permeation. ACS Macro Lett 10:921–925. https://doi.org/10.1021/acsmacrolett.1c00349
Peluso P, Dessì A, Dallocchio R, Mamane V, Cossu S (2019) Recent studies of docking and molecular dynamics simulation for liquid-phase enantioseparations. Electrophoresis 40:1881–1896. https://doi.org/10.1002/elps.201800493
Peluso P, Mamane V, Dallocchio R, Dessì A, Cossu S (2020) Noncovalent interactions in high-performance liquid chromatography enantioseparations on polysaccharide-based chiral selectors. J Chromatogr A 1623:461202. https://doi.org/10.1016/j.chroma.2020.461202
Rajendran A, Paredes G, Mazzotti M (2009) Simulated moving bed chromatography for the separation of enantiomers. J Chromatogr A 1216:709–738. https://doi.org/10.1016/j.chroma.2008.10.075
Scriba GKE (2016) Chiral recognition in separation science–an update. J Chromatogr A 1467:56–78. https://doi.org/10.1016/j.chroma.2016.05.061
Shen J, Okamoto Y (2016) Efficient separation of enantiomers usingstereoregular chiral polymers. Chem Rev 116:1094–1138. https://doi.org/10.1021/acs.chemrev.5b00317
Shiomi K, Yoshikawa M (2013) Multi-stage chiral separation with electrospun chitin nanofiber membranes separation and. Purif Technol 118:300–304. https://doi.org/10.1016/j.seppur.2013.07.004
Shiomi K, Yoshikawa M (2016) Molecularly imprinted chitin nanofiber membranes: multi-stage cascade membrane separation within the membrane. J Membr Sep Technol 5:103–114
Sueyoshi Y, Utsunomiya A, Yoshikawa M, Robertson GP, Guiver MD (2012) Chiral separation with molecularly imprinted polysulfone-aldehyde derivatized nanofiber membranes. J Membr Sci 401–402:89–96. https://doi.org/10.1016/j.memsci.2012.01.033
Thompson CJ, Chase GG, Yarin AL, Reneker DH (2007) Effects of parameters on nanofiber diameter determined from electrospinning model. Polymer 48:6913–6922. https://doi.org/10.1016/j.polymer.2007.09.017
Vedovello P, Marcio Paranhos C, Fernandes C, Elizabeth Tiritan M (2022) Chiral polymeric membranes: recent applications and trends. Sep Purif Technol 280:119800. https://doi.org/10.1016/j.seppur.2021.119800
Wang Z, Zhang S, Chen Y, Zhang Z, Ma S (2020) Covalent organic frameworks for separation applications. Chem Soc Rev 49:708–735. https://doi.org/10.1039/C9CS00827F
Weng X et al (2013) Synthesis and characterization of cellulose 3,5-dimethylphenylcarbamate silica hybrid spheres for enantioseparation of chiral β-blockers. J Chromatogr A 1321:38–47. https://doi.org/10.1016/j.chroma.2013.10.048
Yao Y, Song P, Wen X, Deng M, Wang J, Guo X (2017) Chiral separation of 12 pairs of enantiomers by capillary electrophoresis using heptakis-(2,3-diacetyl-6-sulfato)-β-cyclodextrin as the chiral selector and the elucidation of the chiral recognition mechanism by computational methods. J Sep Sci 40:2999–3007. https://doi.org/10.1002/jssc.201700137
Yoshikawa M, Nakai K, Matsumoto H, Tanioka A, Guiver MD, Robertson GP (2007) Molecularly imprinted nanofiber membranes from carboxylated polysulfone by electrospray deposition. Macromol Rapid Commun 28:2100–2105. https://doi.org/10.1002/marc.200700359
Zhang X, Liu J, Ray AK, Li Y (2023) Research progress on the typical variants of simulated moving bed: from the established processes to the advanced technologies. Processes 11:508
Zhao Y, Zhu X, Jiang W, Liu H, Sun B (2021) Chiral recognition for chromatography and membrane-based separations: recent developments and future prospects. Molecules 26:1145
Zhuo S, Zhang X, Luo H, Wang X, Ji Y (2020) The application of covalent organic frameworks for chiral chemistry. Macromol Rapid Commun 41:2000404. https://doi.org/10.1002/marc.202000404
Acknowledgements
The authors thank Ms. P. Chaud for the SSA measurements, Ms. S. Ortega Murillo for the WCA measurements, Dr. C. Lancelon-Pin for the SEM observation at the Electronic Microscopy Platform (PMIEL) of the Institut de Chimie Moléculaire de Grenoble (ICMG), and Ms. L. Buon and Mr. E. Bayma for the HPLC analysis at the Chromatography and Sugar Analysis Platform (PCANS) of the CERMAV. The authors are thankful to Drs. K. Mazeau, Y. Nishiyama, and J.-L. Putaux (CERMAV) for fruitful discussions and to Prof. R. Pecora (Stanford University) for helpful suggestions during the writing of this manuscript.
Funding
This study was financially supported by the Institut Carnot PolyNat (ANR-16-CARN-0025-0), the Centre de Recherches sur les Macromolécules Végétales (CERMAV, CNRS), Université Grenoble Alpes ARCANE Bio-driven chemistry and CBH-EUR-GS (ANR-17-EURE-0003).
Author information
Authors and Affiliations
Contributions
The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of a collection of articles in honor of Dr. Henri Chanzy on the occasion of his 90th birthday. The article was not finished in time to be bound with the rest of the papers in the Special Issue #13, September, 2023.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nono-Tagne, S., Navon, Y., Ogawa, Y. et al. Enantioselective membranes prepared by electrospinning of cellulose tris(3,5-dimethylphenyl carbamate) having various degrees of polymerization: effect of the DP on the morphology. Cellulose 31, 2765–2782 (2024). https://doi.org/10.1007/s10570-023-05644-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-023-05644-4