Abstract
Polyelectrolyte complexes (PECs) from common papermaking additives were prepared at different cation/anion ratios, resulting in colloidal light scattering particles. The polycations were cationic starches and polyDADMACs, while the polyanions were different carboxymethyl celluloses. The PECs were studied by turbidity measurements, as well as by flow cytometry (FCM). Turbidity maxima were detected close to the theoretical neutralization point of the polycation and polyanion. The turbidity response of the PEC mixtures varied with polyelectrolyte charge density. The PECs were in most cases quite stable over 24 h, but certain combinations resulted in unstable particles over time. Flow cytometry of PECs revealed clear populations of hydrophilic particles. The light scattering properties of PECs in side direction (SSC) and forward direction (FSC) were recorded for the different PEC combinations. The determined FSC and SSC offered information about very different PEC properties, and a new term was suggested for better understanding the mechanisms behind PEC formation; FSC/SSC. It was suggested that the determined FSC/SSC values were connected to the structural density of different particles. The premise was tested by analyses of solid, dense particles as well as swollen, soft particles. In addition to this, the hydrophobicity of PECs was determined by FCM. It was seen that the PECs were quite hydrophilic overall and that the measured hydrophobicities were lowest around the theoretical point of neutralization. Finally, the behavior of a coagulating PEC mixture as a function of contact time was studied with FCM.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Lately, materials research has focused more and more on multi-composites. The potential of combining two or more desirable properties by tailoring the material opens up a vast array of new possibilities (Decher 1997; de Vasconcelos et al. 2006). The use of polyelectrolyte complexes (PECs), or polyelectrolyte multilayers (PEMs) have received quite a lot of attention in the field of research (Philipp et al. 1989; Dautzenberg 1997; Decher 1997; Dautzenberg and Karibyants 1999). Multi-layered structures on surfaces can be formed from a wide variety of components, either from solutions or dispersions.
The most common approach for PEC or PEM formation is the electrostatic attraction between oppositely charged groups. The formation of PECs is driven by the increase in entropy when counter ions are liberated from the oppositely charged polyelectrolytes during associative phase separation into colloidal particles (Piculell and Lindman 1992; Kekkonen et al. 2001). Strong ionic binding between the opposite charges of the polyelectrolytes results in kinetically frozen structures (Dautzenberg and Karibyants 1999). It has been reported that the core of PEC particles consists of a neutralized polyelectrolyte core, while the outer shell consists mainly of excess polyelectrolyte, which stabilizes the colloids against aggregation (Dautzenberg and Karibyants 1999).
The use of PECs and PEMs have also found their way into the field of pulping and papermaking (Petzold and Lunkwitz 1995; Petzold et al. 1996; Gernandt et al. 2003; Hubbe et al. 2003, 2005; Gärdlund et al. 2005; Lofton et al. 2005; Torgnysdotter and Wågberg 2006; Myllytie et al. 2009; Renneckar and Zhou 2009; Lin and Renneckar 2011; Granberg et al. 2012; Ankerfors and Wågberg 2013; Marais et al. 2014). The use of PECs and PEMs in papermaking is somewhat expected, since the properties of paper and retention of fillers commonly have been tailored by additions of polyelectrolytes, in single or dual systems, for a long time (Hubbe 2006). Additionally, surfaces of pulp fibers carry anionic charges that are convenient anchor points for polyelectrolytes and PECs. In papermaking applications, the size of the PECs can be tailored to exceed the average pore radius of pulp fibers, to ensure that the PECs are retained mainly on fiber surfaces. In papermaking, surface-specific strengthening agents are especially interesting, since the additive should be adsorbed within the cross-over region of two fibers in order to enhance paper strength (Stratton 1989).
The properties and behavior of PECs in suspension has previously been studied by measurements of turbidity, static light scattering, dynamic light scattering, polyelectrolyte titration, viscosity, surface charge analysis, FTIR spectroscopy, and electrophoretic mobility (Buchhammer et al. 1995; Dautzenberg 1997; Dautzenberg and Karibyants 1999; Dragan and Cristea 2001; Kekkonen et al. 2001; Hubbe et al. 2005; de Vasconcelos et al. 2006). It has been reported that the formation of PECs is sensitive towards the nature of the cationic polyelectrolyte (Hubbe et al. 2005). It is known that the formation of PECs is path dependent, which means that all aspects of the PEC formation will influence the result (Kötz 1993; Feng et al. 2008). Due to the wide variety of available polyelectrolytes, additional information about their interactions and the complexes they form is still needed.
Adsorption experiments and modeling have been used to gain information about formed layers of polyelectrolytes (Van de Steeg et al. 1992; Fleer et al. 1993). Adsorption studies using QCM-D and SPR techniques have revealed that polyelectrolytes adsorb differently depending on their chemical properties, such as molar mass and charge density, onto model cellulose or silica surfaces (Tammelin et al. 2004; Kontturi et al. 2008; Strand et al. 2017). A polyelectrolyte with a high charge density will adsorb as a stiffer and more rigid layer, compared to a polyelectrolyte with a low charge density (Kontturi et al. 2008). The charge density of the surface itself in adsorption studies is also of great importance, which was shown by adsorption experiments onto cellulose and silica surfaces. While the QCM-D and SPR methods provide information about adsorption of polyelectrolytes onto surfaces, the behavior of unretained polycations and polyanions is an open question. An analysis method that could bridge the gap between the previously used analyses for PECs and the techniques used in adsorption studies would be helpful for gaining additional insight into the behavior of polyelectrolyte mixtures.
Flow cytometry (FCM) is a relatively new technique in the field of pulping and papermaking. The technique was adapted from medical science, where it is used mainly for the counting of cells (Shapiro 2003). FCM also measures the light scattering properties of particles in suspension in forward and side direction. Further, FCM can be used to measure the fluorescence of particles at different wavelengths, which is very useful when combined with addition of selective dyes to samples. In the field of pulping and papermaking, FCM has so far been used to analyze colloidal wood pitch, bacteria, coated broke, precipitated oxalate, and filler particles (Vähäsalo et al. 2003; Lindberg et al. 2004; Vähäsalo and Holmbom 2005; Strand et al. 2013; Häärä et al. 2014). With FCM techniques, it is possible to analyze particles in suspension directly without complicated pre-treatments. The strengths of the FCM technique are short analysis times, as well as insight into the behavior and interactions of particle populations in aqueous suspension.
The aim of this work was to gain additional knowledge about PECs formed from commonly used papermaking additives, by analysis with flow cytometry. The light scattering properties of these particles might reveal important information about their properties and structure. The additives chosen for the study were different cationic starches, polyDADMAC and carboxymethyl cellulose (CMC). Cationic starches are the most commonly used additive in papermaking (Fornue et al. 2011). Studies have shown that CMC greatly increases both the strength and stiffness of paper, which makes it a very interesting papermaking additive (Beghello et al. 1997; Strand et al. 2017). An additional aim of this work was to determine the hydrophobicity of PECs, and to study the time dependent stability of different PEC mixtures, since contact times between polyelectrolytes in papermaking can vary between fractions of a second to several hours.
Materials and methods
Materials
Three different cationic starches were received from Chemigate as pre-boiled slurries; Raifix 01015 SW, Raifix 25035, and Raisabond 15. The cationic starch slurries were diluted with distilled water to 0.1% solutions under agitation. Sodium carboxymethyl cellulose (CMC), Mw 700 kDa and DS 0.80–0.95, was obtained from Aldrich as dry powder. Additionally, three 250 kDa CMCs of varying DS were obtained from Acros Organics as dry powders. The different CMCs were dissolved in distilled water to a concentration of 0.1% with the aid of agitation and boiling, until clearly dissolved. Two different polydiallyldimethylammonium chloride (PolyDADMAC) solutions were obtained from Aldrich. The polyDADMACs were received as viscous solutions with dry contents of about 26% and molar masses of 100–200 and 200–350 kDa, respectively. The polyDADMAC was diluted with distilled water to a concentration of 0.1% under agitation. Poly(vinyl sulfate) potassium salt (KPVS) was obtained from Wako. The KPVS salt was dissolved in deionized water under agitation. Hexadimethrine bromide (polybrene) was obtained from Sigma. The polybrene was dissolved in deionized water under agitation. Nile red was obtained in dry form from from Sigma-Aldrich, USA. The Nile red was dissolved in methanol to a concentration of 10 ppm.
A variety of commercially available mineral particles was obtained. Prismatic precipitated calcium carbonate (PCC p) was obtained from Specialty Minerals, New York, USA, as a slurry, which was diluted with pH-adjusted distilled water (pH 8). Scalenohedral precipitated calcium carbonate (PCC s) was obtained from Specialty Minerals Nordic, Anjalankoski, Finland as a slurry, which was diluted with pH-adjusted distilled water (pH 8). Commercially available ground calcium carbonate (GCC) was obtained as a slurry, and was diluted with distilled water. Kaolin was obtained from Imerys Minerals, Par Cornwall, UK, in powder form and was dispersed in distilled water. Three different grades of commercially available bentonites were obtained from Clariant Produkte, Germany, and dispersed in distilled water. Cellulose nanocrystals (CNC) were prepared by H2SO4 hydrolysis of birch kraft pulp, with subsequent purification steps (Beck-Candanedo et al. 2005). Cellulose nanofibrils (CNF) were produced by TEMPO oxidation of birch kraft pulp followed by mechanical defibration (Shibata and Isogai 2003).
Methods
Charge titration
The anionic or cationic charge of the different polyelectrolytes was analyzed by polyelectrolyte titration with a particle charge detector (Mütek PCD 03, Mütek Analytic GmbH, Germany; BTG, Switzerland) coupled to a titration device (736 GP Titrino, Metrohm Ltd., Switzerland). The cationic polyelectrolytes were titrated with KPVS, and the anionic polyelectrolytes were titrated with polybrene with an Mw of approximately 8 kDa (Sundberg et al. 2000). The amount of titrant needed for charge neutralization was used to calculate the total charge of the analyzed polyelectrolyte (Tables 1 and 2).
Formation of polyelectrolyte complexes
Different volumes of 0.1% polycation solution were pipetted into glass bottles. A fixed volume of 0.1% polyanion solution was pipetted into the polycation solution under vigorous magnetic stirring. To simplify and standardize the procedure, the polyanion was always added to the polycation. Polyelectrolyte complexes (PECs) started forming immediately when the polyanion came in contact with the polycation, which was evident as increasing opacity of the mixture upon visual inspection. The PEC mixtures were diluted to a total volume of 30 mL with distilled water within a few seconds of contact time, prior to analyses. All experiments were performed at room temperature.
Turbidity measurements
The initial turbidity of the PEC mixture was analyzed within 1 min after PEC formation using a turbimeter (Hach 2100AN IS, ISO 7027) equipped with a LED light source (870 ± 30 nm). In order to assess the long-term stability of the PECs, the mixture was stored in closed glass bottles at room temperature for 24 h before the turbidity was measured again using the same device. The mixtures were stirred for a short time prior to the turbidity measurements.
Flow cytometry
Flow cytometry (FCM) was used to analyze the PEC mixtures, using a Partec CyFlow Blue apparatus, equipped with a blue Argon laser (488 nm). The light scattered by the PEC particles were recorded in forward direction (FSC) and side direction (SSC, 90°). The apparatus was also equipped with three different fluorescence channels, which recorded the scattering intensity in the green (512–542 nm), orange (575–605 nm), and red (615–645 nm) spectra. Filtered distilled water was used as sheath fluid in the analyses. Nile red (Tamro, Vantaa, Finland) was used as a fluorescent dye to stain the particles prior to FCM analysis, as in previously published studies (Vähäsalo et al. 2003; Vähäsalo and Holmbom 2005; Strand et al. 2013). Nile red is an environment-sensitive fluorophore that exhibits a blue-shift proportional to the hydrophobicity of its environment (Greenspan and Fowler 1985). It was expected that the added Nile red mainly stained the surface of the PEC particles, since the migration of Nile red into the PECs, and the solubility of Nile red in water, are both negligible effects.
PEC samples were diluted 3–100 times with filtered distilled water prior to FCM analysis, depending on the concentration of particles in the sample. Methanol solution of Nile red (10 ppm) was added to stain the PEC particles, and the fluorescence intensity in the red spectrum was used as an indicator of particle hydrophobicity. FCM analysis was used to count the number of particles in the sample, and group the particles together as populations based on their recorded light scattering properties. Particle populations were gated using Partec FloMax software, in order to calculate the average scattering of particles in side and forward direction, as well as particle hydrophobicity (Vähäsalo et al. 2003).
Results and discussions
Turbidity measurements
Solutions of polycations were mixed together with solutions of polyanions, forming nearly clear solutions or turbid suspensions depending on the cation/anion ratio (Fig. 1). The increase in turbidity was caused by associative phase separation of the oppositely charged polyelectrolytes into colloidal particles (Piculell and Lindman 1992; Kekkonen et al. 2001). The turbidity increased up to cation/anion ratios close to the theoretical neutralization point of the polyelectrolytes, and decreased after this ratio was exceeded. The calculated theoretical neutralization points between the different polyelectrolytes are presented in Table 3. In order to illustrate that the turbidity maxima were connected to charge neutralization, the polycations were mixed together with CMCs of different charge density, or degree of substitution (DS). Using a CMC with higher charge density shifted theoretical point of neutralization towards higher cation/anion ratios, since a larger amount of polycation was needed in order to neutralize the anionic charge. The turbidity curves in these experiments were similar to previously published results using other polyelectrolytes (Dragan and Cristea 2001; Hubbe et al. 2005; Lofton et al. 2005).
It has been shown that PECs coexist with dissolved polyelectrolyte species at cation/anion ratios above and below the direct vicinity of the neutralization point (Kekkonen et al. 2001). However, PEC mixtures close to the neutralization point only contain low residual amounts of dissolved polyelectrolytes. Excess of one of either anionic or cation polyelectrolyte results in a stabilizing shell around a neutralized polyelectrolyte core (Dautzenberg and Karibyants 1999). The PEC particles carry either negative or positive net charge due to the cation/anion ratio of the mixture. Measurements have shown that the sign of the PEC particles charge changes sharply within a quite narrow cation/anion ratio (Kekkonen et al. 2001).
The maximum turbidity for these mixtures appear close to the theoretical neutralization point, but not necessarily precisely on the neutralization point. Similar observations have been reported earlier in several studies (Chen et al. 2003). It was reported that a slight excess of polyDADMAC was needed to completely neutralize the polyanion used (Kekkonen et al. 2001). Similarly, it was reported that the near-zero electrophoretic mobility of PECs from polyDADMAC and CMC was achieved at a ratio of 8:10 of cationic to anionic groups (Hubbe et al. 2005). Deviations from a 1:1 when using irregularly branched polyelectrolytes are quite common (Kötz 1993). This was noticeable in the determined turbidity values when cationic starches Raifix 01015 SW and Raisabond 15, which both contain a high amount of branched amylopectin, were mixed together with the different CMC grades (Fig. 1). Steric factors, differences in chain lengths, and differences charge densities have previously been used to explain deviations from 1:1 stoichiometric neutralization points between polyelectrolytes of opposite charge (Philipp et al. 1989; Buchhammer et al. 1995; Kekkonen et al. 2001). It has previously been reported that the maximum turbidity decreases with increasing molar mass of the polyelectrolyte (Hubbe et al. 2005). This correlation was not apparent in these experiments, but instead it was noted that the maximum turbidity depended on the charge density of the cationic polyelectrolyte, when the amount of anionic charges were kept constant in all trial points.
Long-term stability of PECs
The long-term stability of PEC mixtures was evaluated by turbidity measurements 24 h after PEC formation. The measured turbidity values after 24 h were lower than the previously measured values for almost all of the tested PEC combinations (Fig. 2). Only small changes in turbidity were expected for PEC mixtures that contained an excess of either polycation or polyanion, due to electrostatic stabilization of the particles. It is known that the PEC particles consist of a core of neutralized polyelectrolytes, and an outer shell is formed from the polyelectrolyte in excess (Dautzenberg and Karibyants 1999). It was clear that some changes occurred in the PEC particles over time, and that these changes were most likely an effect of secondary aggregation of particles. Decreases and increases in turbidity over time have previously been reported for other PEC mixtures (Hubbe et al. 2005). Others have reported colloidally stable PECs over time periods of 48 h, and in some cases even 2 months (Gernandt et al. 2003).
However, some very noticeable decreases in turbidity took place at certain cation/anion ratios. Combinations of Raifix 01015 SW with the different CMCs resulted in unstable PEC mixtures at cation/anion ratios close to the theoretical point of neutralization. In these experiments, Raifix 01015 SW was the only tested cationic starch produced from waxy maize, i.e. it consisted approximately of 98% branched amylopectin, unlike the other cationic starches from potato (Fredriksson et al. 1998). This low-molar mass and branched structure seemed to form unstable aggregates effectively, given enough time. Also a 5.5:1 ratio of Raisabond 15 and DS 1.2 CMC also resulted in unstable aggregates over time, i.e. once again close to the theoretical point of neutralization. An incomplete, and less noticeable turbidity decrease was seen for Raisabond 15 with DS 0.7 CMC at the ratio of 4:1, which also was close to the theoretical neutralization point at 3.8.
Flow cytometry
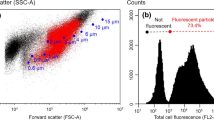
Polyanion was mixed together with polycations under agitation. The PECs were diluted, a small amount of staining agent for hydrophobic components (Nile red) was added, and the mixture was analyzed by flow cytometry. The Argon laser in the FCM instrument scanned the PECs. The light scattered by each colloidal particle was recorded in the forward direction (FSC), side direction (SSC), and in the red spectra. The different light scattering properties of the PEC particles were plotted against each other, in order to create particle populations in FCM density plots. The PEC particle populations were quite visible when FSC was plotted versus SSC, as illustrated in Fig. 3a of a 0.5:1 mixture of polyDADMAC (100–200 kDa) with CMC (250 kDa, DS 0.9).
Two logarithmic flow cytometry dot plots of PECs in aqueous media, using a mixture of polyDADMAC (100–200 kDa) + CMC (250 kDa, DS 0.9) with a cation/anion ratio of 0.5:1. The properties of the measured particles are plotted as a their side scattering (SSC) versus forward scattering (FSC), and b their FSC versus intensity in the red spectrum (hydrophobicity)
The colloidal PEC particles interacted with the added hydrophobic dye, which was seen in the recorded intensities of the particles in the red spectra (Fig. 3b). The added Nile red was most likely adsorbed on the surface of the PECs. Nile red is an environment-sensitive fluorophore, which is almost non-fluorescent in hydrophilic environments, while it undergoes a fluorescent enhancement and large absorption and blue shifts in hydrophobic environments. The fluorescence in the red spectrum has commonly been used as a measurement of particle hydrophobicity (Vähäsalo et al. 2003; Strand et al. 2013). The PECs were quite hydrophilic; the low intensities of the PEC particles in the red spectra indicated that the particles were hydrophilic, even though the chemical environment could activate the hydrophobic dye to some extent. A hydrophilic nature of the PECs was very much suspected, since these consist of kinetically frozen structures of polyelectrolytes that were previously soluble in water (Dautzenberg and Karibyants 1999).
Particle count
PECs of polyDADMAC or three different commercially available cationic starches with CMC of 250 and 700 kDa were prepared at different cation/anion ratios. Noticeable variations in the number of particles per µL was seen when 250 kDa CMC was used as polyanion, while the particle count remained more stable when 700 kDa CMC was used (Fig. 4a, b). Adsorption experiments have previously shown that increasing the molar mass of cationic starch results in a larger hydrodynamic thickness of the adsorbed layer on quartz surfaces (Tammelin et al. 2004). The Mw of PEC particles have previously been shown to increase with increasing molar mass of the used polyDADMAC (Dautzenberg 2000). It would be expected that increasing the molar mass of CMC in these experiments also would lead to thicker, more compact particles in suspension.
It is important to remember that particles smaller than 0.1 µm were not analyzed in the FCM measurements, and are not included in the results. This phenomenon is highlighted when combining Raifix 01015 SW with 250 kDa CMC at a cation/anion ratio of 5:1, which resulted in a coagulating combination of polyelectrolytes (Fig. 4a). Close to the neutralization point, the large number of particles below the detection limit were suddenly elevated into the detection range of the FCM, which resulted in a large spike in particles per µL.
Light scattering properties
PEC mixtures were analyzed by flow cytometry at different cation/anion ratios. The scattering of light in side direction (SSC), and scattering of light in forward direction (FSC) was recorded for the particles. Average values were calculated for the different PEC populations, and these values were plotted as a function of cation/anion ratio. Mixing together polyDADMAC with CMC resulted in PEC particles with quite high SSC values (Fig. 5a). The SSC of particles consisting of cationic starches and CMC were considerably lower at all measured cation/anion ratios. Overall, the changes in the determined SSC values were quite small as function of cation/anion ratio for all of these PEC mixtures.
Larger and more noticeable changes took place in the average light scattered in forward direction (FSC) of the PEC mixtures. The FSC of PECs formed from polyDADMAC and CMC decreased dramatically as the cation/anion ratio increased between 0.5 and 2, even though this type of behavior was not reflected in the determined SSC values (Fig. 5b). The FSC for PECs formed from Raifix 25035 and CMC also decreased noticeably after the theoretical neutralization point. The FSC of PECs from Raisabond 15 and Raifix 01015 SW were overall significantly higher than for the other mixtures. Large differences in the FSC values as a function of cation/anion ratio, that were not reflected in the SSC values, indicated that FSC and SSC were connected to very different properties of the PEC particles. Available explanations from medical science about FCM of cells may be useful for understanding the scattered light in different directions of these PEC particles.
In medical science, SSC is commonly used as an indicator of the granularity of cells, i.e. the amount of granular components within a cell that are able to scatter light in the side direction (Shapiro 2003). Therefore, SSC is not a direct measurement of particle size, but can be used to distinguish cell types from each other. Degranulation of cells and membrane ruffling following activation are known to decrease the SSC of cells. On the other hand, FSC is commonly used as an indicator of cell size, but is not a direct measurement of cell size (Shapiro 2003). It is known that many factors influence the FSC of a cell, such as the wavelength of the incoming light, the precise angle that the light is collected, the internal structure of the cells, the presence of strongly absorbing materials within the cell, and refractive index of the cell. It has been shown that osmotic swelling of cells, as well as damages from freezing and thawing of cells will lower their measured FSC. Especially interesting is the fact that changes in lipid packing within a cell can affect the FSC, without changes in cell volumes. It has also been reported that highly textured surfaces or internal structures lower the FSC of a cell.
With the explanations of FCM data from medical science, it is clear that SSC and FSC give information about very different properties of the PEC particles. The SSC of a particle gives information about the amount of dense and light scattering surfaces. The FSC of a particle gives information about the transparency of a particle, as well as the complexity of its external or internal texture. The light scattering in both side and forward direction are still connected to particle size, even though the dependence is indirect. An attempt to diminish the influence of particle size by combining the SSC and FSC of PEC particles into one single term, in the form of FSC/SSC values, gave new correlations.
The FSC/SSC values of PECs formed from polyDADMAC and CMC at low cation/anion ratio was 2.4, but decreased to about 1.1 with increasing cation/anion ratio (Fig. 6). The size estimation of these PEC particles from SSC and FSC were almost the same at the highest examined cation/anion ratio. The FSC/SSC of PECs from Raifix 25035 and CMC were slightly higher at low cation/anion ratio, but also decreased after the theoretical neutralization point was exceeded. The FSC/SSC values of PECs formed from Raifix 01015 SW or Raisabond 15 with CMC were higher throughout the studied cation/anion interval, yet similar to each other. PolyDADMAC, with a charge density of about 4 meq/g, gave lower FSC/SSC values than Raifix 25035 (1.9 meq/g), which in its turn gave lower values than Raifix 01015 SW and Raisabond 15 (0.8 and 0.9 meq/g, respectively). The results indicated that the FSC/SSC values decreased with increasing charge density of the polycation, while the properties of the polyanion was kept constant. Furthermore, the results indicated that for most combinations the FSC/SSC values seemed to decrease after the theoretical point of neutralization.
In order to understand these results, available literature about the adsorption behavior of polyelectrolytes onto model surfaces need to be considered, since the interactions between two dissolved polyelectrolytes may exhibit similar mechanisms. Adsorption experiments with QCM-D have previously shown that polyelectrolytes adsorb differently onto model surfaces depending on their charge density and molar mass (Tammelin et al. 2004; Kontturi et al. 2008). Adsorbed polyelectrolyte layers consist of the polymer, electrolyte (if present), and solvent that is imbibed inside the layer (Kontturi et al. 2008). The amount of water moving with the polyelectrolyte can be quite substantial, if the polyelectrolyte forms extended loops, trains, or tails. It was previously shown that adsorbed layers of CMC and these cationic starches onto nanocellulose surfaces contained between 52 and 66% of coupled water by weight at adsorption equilibrium (Strand et al. 2017). Cationic starches with high DS onto cellulose surfaces results in a significantly more rigid layer, compared to cationic starches with a low DS, because polyelectrolytes with high DS are more strongly attracted to surfaces of opposite charge.
There are some similarities between adsorption of polyelectrolytes onto model surfaces and the creation of PECs, since both are driven by attraction of oppositely charged groups. Instead of adsorbing the polyelectrolyte onto an existing surface, it is adsorbed onto another polyelectrolyte in order to create a completely new colloidal phase. The FCM results indicate that similar rules apply in the formation of PECs as in the adsorption studies; A polyelectrolyte with higher charge density will have higher affinity to the oppositely charged polyelectrolyte, creating a more dense structure. In these experiments, both polyDADAMAC and CMC consisted of linear and highly charged polyelectrolyte chains, that resulted in FSC/SSC values as low as 1.1. The dense PEC structure was able to scatter light more effectively in SSC, and the increasing complexity of the particle structure caused less effective light scattering in FSC. A determined FSC/SSC value approaching 1.0 indicated that quite dense PEC particles were formed. It has previously been reported that higher structure density of PECs was obtained when CMC of DS 1.3 was mixed with amine-epichlorhydrin resin (PAE), compared to when CMC of DS 0.7 was used (Gernandt et al. 2003). Different models for explaining the degree of PEC particle swelling/density as a function of polyelectrolyte chain length and charge density have been theorized upon, and it was shown that the density of the PEC core is influenced by polyelectrolyte charge density (Dautzenberg 1997; Mende et al. 2002).
In mixtures of cationic starches and CMC, the FSC/SSC values indicated that the PECs were less dense than the PECs formed with polyDADMAC (Fig. 6). The determined FSC/SSC values increased with decreasing charge density of the polycations, indicating that less dense PECs were formed with the cationic starches of lower charge density. It has been reported that large differences in charge density between the polycation and polyanion may lead to less dense, and more swollen PEC structures (Dautzenberg 1997). The cationic starches originated either from potato or waxy maize, that contain about 77 and 98% of branched amylopectin respectively (Fredriksson et al. 1998). Less well-ordered packing of the cationic starches in the PEC particles would be expected, compared with the linear and highly charged polyDADMAC.
FCM analysis of various particle types was performed in order to gain more insight about FSC/SSC values. The available particles were kaolin, scalenohedral and prismatic precipitated calcium carbonates (PCC s, PCC p), ground calcium carbonate (GCC), three different bentonites, cellulose nanocrystals (CNC), and cellulose nanofibrils (CNF). The FCM analyses were performed in similar fashion, and the calculations were performed in the same way as for the PECs. Theoretically, solid particles should have FSC/SSC values close to 1.0 in the FCM setup. This was seen for both of the PCC particle types, GCC, and kaolin (Table 4). The bentonites, which are known to swell in water, had FSC/SSC values between 2.8 and 4.6. The FSC/SSC of cellulose nanocrystals, which are known to be dense and crystalline cellulose particles, was low. The FSC/SSC of cellulose nanofibrils, which are known to be very swollen in water, was 6.6. The obtained results indicated that the determined FSC/SSC values could in fact be connected to particle density or swelling. By mixing together different types of polyelectrolytes into PECs, FSC/SSC values of 1.1–8.7 have so far been obtained, indicating that the choice of polyelectrolytes and the ratio between them will results in very different populations of PEC particles.
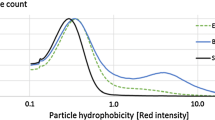
The hydrophobicity of PEC particles was determined by FCM analyses with the added hydrophobic staining agent Nile red. A small volume of Nile red dissolved in methanol was added to the different water samples. It was expected that the added Nile red mainly adsorbed onto the surface of the different PEC particles, since the solubility of Nile red in water is negligible and the migration of Nile red into the PECs is unlikely (Greenspan and Fowler 1985; Jose and Burgess 2006). Once activated, Nile red emitted fluorescent light that was detected in the red spectra of the FCM apparatus. The PEC particles interacted and activated the added Nile red, but the detected red intensities were quite low, which indicated that the PECs were ultimately hydrophilic in nature (Fig. 7). The average hydrophobicity of the PEC particles were of the same size range for all of the measured polyelectrolyte combinations at different cation/anion ratios. The average hydrophobicity of the PECs was lowest around the theoretical point of neutralization for all of the PEC combinations, and increased once this cation/anion ratio was exceeded. The results showed that the added Nile red was activated to a lesser extent close to the neutralization points, which indicated that the particles were slightly less hydrophobic.
It has been reported that the hydrophobic domains exist within polyelectrolytes, and that these tend to vary with the conformational changes (Abe et al. 1977). It was reported that a polyelectrolyte with many hydrophilic ionic sites will show a hydrophobic atmosphere, or a low-polar solvent-like environment, within its domains. The hydrophobicity of a dense polyelectrolyte conformation is higher than for a polyelectrolyte with extended conformation. The presented FCM results would indicate that the polyelectrolyte chains on the surfaces of the PEC particles were less dense around their neutralization points, due to the slight decrease in hydrophobicity. More extended conformations of the polyelectrolytes may have been favored around the neutralization point. Additionally, the determined hydrophobicities of the PECs followed their FSC/SSC values to some extent; increases in structural density of the PECs seemed to be connected to increases in hydrophobicity.
Time dependency: coagulation
To study the time dependency of PECs over a short time scale, a coagulating PEC mixture was chosen for FCM analysis. From the preliminary turbidity measurements, it was known that a mixture of Raifix 01015 SW and 250 kDa CMC, DS 0.9, at a ratio of 5:1, was unstable over time. The two polyelectrolytes were quickly mixed together and diluted. The first sample was collected after 6 s of polyelectrolyte contact time, and it was quickly fed into the FCM apparatus. The initial number of particles in suspension was about 41000/µL, and the measured FSC/SSC value of the PEC population was 3.4 (Fig. 8). The number of particles per µL decreased quite rapidly within the first 5 min of contact time, from 41000 to about 10000/µL, most likely due to secondary aggregation of the initial particles. Within the first 5 min, the FSC/SSC value of the particle population also increased from 3.4 to 8.7, indicating the formation of large, swollen PEC particles with low structural density. The particle count continued to decrease over time, and only about 500/µL remained at 53 min. From the FCM density plots, it was visible that the largest, and most unstable aggregates disappeared over time. The largest aggregates formed macroscopic flocs, that were either left in suspension or attached to the glass beaker walls. The FSC/SSC value of the particle population seemed to stabilize at about 5.5 after 53 min, but the number of particles remaining in suspension was only about 1% of the initial amount.
The large change in the observed PEC population should be of special interest in wet-end chemistry, where the contact times between polycations and polyanions can be extremely short. These studied additives required a short contact time before large, swollen, and easily retainable PEC particles were formed. In adsorption studies at high flux, an initial over-adsorption of polymer has been noted, and the over-adsorption was corrected by partial desorption over time (Fleer et al. 1993). In a previous study, it was noted that the response to additions of salt took up to 60 min for some PEC systems (Dautzenberg and Karibyants 1999). In another study, the PEC mixtures were stirred for 10 min, and allowed to stabilize for 2 h prior to characterization (Mende et al. 2002). Therefore, the time dependency of PECs should be studied in more detail, and also be taken into account when studying mixtures of this nature. The situation at initial polyelectrolyte contact, where a papermaker usually operates, may be very different from the equilibrium states commonly studied at laboratory scale. The salt concentration should also be taken into account, since this will influence the results due to the introduction of screening effects during PEC formation.
It should be mentioned again, that every step of PEC formation will influence the result (Kötz 1993; Feng et al. 2008). Slowly introducing the polycation to its counterpart, which is a common approach in literature, in order to study the cationically stabilized PECs at cation/anion ratios exceeding the neutralization point will cause the interaction path to pass through the unstable region. By suddenly introducing the polycation, it is possible to circumvent this phenomenon and obtain cationically stabilized particles that are not products of secondary aggregation.
Conclusions
Polyelectrolyte complexes (PECs) were prepared from common papermaking additives at different cation/anion ratios. The polycations were different cationic starches and polyDADMACs, while the polyanions were CMCs of various molar masses and charge densities. The colloidal, light scattering PEC particles were studied by turbidity measurements, as well as by flow cytometry (FCM). Turbidity maxima of the different PEC mixtures were located close to the theoretical point of neutralization between the polycation and polyanion. The turbidity response varied with polyelectrolyte charge density. The formed PECs were in most cases stable for 24 h, but certain combinations close to the theoretical point of neutralization resulted in unstable complexes.
The light scattering properties of PECs in side direction (SSC) and in forward direction (FSC) were recorded by FCM analyses. It was seen that the SSC and FSC of the analyzed particles were influenced by very different particle properties, and a new term was suggested to better understand the properties of these particles in suspension; FSC/SSC. It was shown that the calculated FSC/SSC values were connected to the structural density of particles. The hydrophobicity of PECs was also analyzed by FCM. All of the measured PEC combinations were very hydrophilic, and it was seen that the hydrophobicity of the particles was lowest close to the theoretical point of neutralization. Finally, a coagulating PEC mixture was studied by FCM analysis as a function of time. It was shown that the initial particle count decreased by 75% over a short period of time. The PECs were initially of medium density, but quickly formed large, swollen particles due to secondary aggregation amongst the initial particles. After 53 min, only about 1% of the particles remained in suspension.
It is suggested that FCM can be used to study polyelectrolyte complexes in suspension. FCM provides relevant information about the structural density and hydrophobicity of these particles. With FCM it would be possible to simultaneous analyze PEC properties and their interactions with other particles in suspension. When dealing with research or production of PECs, FCM techniques can be very useful for categorizing the formed particles.
References
Abe K, Koide M, Tsuchida E (1977) Hydrophobicities in the domains of polyelectrolytes. J Polym Sci, Part A: Polym Chem 15:2469–2477. https://doi.org/10.1002/pol.1977.170151016
Ankerfors C, Wågberg L (2013) Polyelectrolyte complexes for tailoring of wood fibre surfaces. In: Müller M (ed) Polyelectrolyte complexes in the dispersed and solid state II. Springer, Berlin Heidelberg, pp 1–24. https://doi.org/10.1007/12_2012_206
Beck-Candanedo S, Roman M, Gray D (2005) Effect of reaction conditions on the properties and behavior of wood cellulose nanocrystal suspensions. Biomacromol 6:1048–1054. https://doi.org/10.1021/bm049300p
Beghello L, Long L, Eklund D (1997) Laboratory study on carboxymethylcellulose as a wet-end additive in paperboard making. Pap Puu 79:55–57
Buchhammer H-M, Kramer G, Lunkwitz K (1995) Interaction of colloidal dispersions of non-stoichiometric polyelectrolyte complexes and silica particles. Colloids Surf A 95:299–304. https://doi.org/10.1016/0927-7757(94)03036-Y
Chen J, Heitmann A, Hubbe M (2003) Dependency of polyelectrolyte complex stoichiometry on the order of addition. 1. Effect of salt concentration during streaming current titrations with strong poly-acid and poly-base. Colloids Surf A 223:215–230. https://doi.org/10.1016/S0927-7757(03)00222-X
Dautzenberg H (1997) Polyelectrolyte complex formation in highly aggregating systems. 1. Effect of salt: polyelectrolyte complex formation in the presence of NaCl. Macromolecules 30:7810–7815. https://doi.org/10.1021/ma970803f
Dautzenberg H (2000) Polyelectrolyte complex formation: role of a double hydrophilic polymer. Macromol Chem Phys 201:1765–1773. https://doi.org/10.1002/1521-3935(20000901)201:14<1765:AID-MACP1765>3.0.CO;2-W
Dautzenberg H, Karibyants N (1999) Polyelectrolyte complex formation in highly aggregating systems. Effect of salt: response to subsequent addition of NaCl. Macromol Chem Phys 200:118–125. https://doi.org/10.1002/(SICI)1521-3935(19990101)200:1<118::AID-MACP118>3.0.CO;2-K
De Vasconcelos C, Bezerril P, Dos Santos D, Dantas T, Pereira M, Fonseca J (2006) Effect of molecular weight and ionic strength on the formation of polyelectrolyte complexes based on poly(methacrylic acid) and chitosan. Biomacromol 7:1245–1252. https://doi.org/10.1021/bm050963w
Decher G (1997) Fuzzy nanoassemblies: toward layered polymeric multicomposites. Science 277:1232–1237. https://doi.org/10.1126/science.277.5330.1232
Dragan S, Cristea M (2001) Influence of low-molecular-weight salts on the formation of polyelectrolyte complexes based on polycations with quaternary ammonium salt groups in the main chain and poly(sodium acrylate). Eur Polym J 37:1571–1575. https://doi.org/10.1016/S0014-3057(01)00025-8
Feng X, Leduc M, Pelton R (2008) Polyelectrolyte complex characterization with isothermal titration calorimetry and colloid titration. Colloids Surf A 317:535–542. https://doi.org/10.1016/j.colsurfa.2007.11.053
Fleer G, Cohen Stuart M, Scheutjens J, Cosgrove T, Vincent B (1993) Electrostatic effects: charged surfaces and polyelectrolyte adsorption. In: Polymers at interfaces, 1st edn. Chapman & Hall, London, pp 343–375
Fornue E, Allan G, Quinones H, Gonzalez G, Saucedo J (2011) Fundamental aspects of adhesion between cellulosic surfaces in contact—a review. Papel 72:85–90
Fredriksson H, Silverio J, Andersson R, Eliasson A-C, Aman P (1998) The influence of amylose and amylopectin characteristics on gelatinization and retrogradation properties of different starches. Carbohydr Polym 35:119–134
Gärdlund L, Forsström J, Andreasson B, Wågberg L (2005) Influence of polyelectrolyte complexes on the strength properties of papers from unbleached kraft pulps with different yields. Nord Pulp Pap Res J 20:36–42. https://doi.org/10.3183/NPPRJ-2005-20-01-p036-042
Gernandt R, Wågberg L, Gärdlund L, Dautzenberg H (2003) Polyelectrolyte complexes for surface modification of wood fibres: I. Preparation and characterisation of complexes for dry and wet strength improvement of paper. Colloids Surf A 213:15–25. https://doi.org/10.1016/S0927-7757(02)00335-7
Granberg H, Coppel L, Eita M, de Mayolo A, Arwin H, Wågberg L (2012) Dynamics of moisture interaction with polyelectrolyte multilayers containing nanofibrillated cellulose. Nord Pulp Pap Res J 27:496–499. https://doi.org/10.3183/NPPRJ-2012-27-02-p496-499
Greenspan P, Fowler S (1985) Spectrofluorometric studies of the lipid probe, nile red. J Lipid Res 26:781–789
Häärä M, Konn J, Vähäsalo L, Sundberg A, Willför S (2014) Flow cytometry as a tool to assess inhibitor performance for calcium oxalate scale control. Nord Pulp Pap Res J 29:663–672. https://doi.org/10.3183/NPPRJ-2014-29-04-p663-672
Hubbe M (2006) Bonding between cellulosic fibers in the absence and presence of dry-strength agents—a review. BioResources 1:281–318. https://doi.org/10.15376/biores.1.2.281-318
Hubbe M, Jackson T, Zhang M (2003) Fiber surface saturation as a strategy to optimize dual-polymer dry strength treatment. Tappi J 2:7–12
Hubbe M, Moore S, Lee SY (2005) Effects of charge ratios and cationic polymer nature on polyelectrolyte complex deposition onto cellulose. Ind Eng Chem Res 44:3068–3074. https://doi.org/10.1021/ie048902m
Jose J, Burgess K (2006) Syntheses and properties of water-soluble nile red derivatives. J Org Chem 71:7835–7839. https://doi.org/10.1021/jo061369v
Kekkonen J, Lattu H, Stenius P (2001) Adsorption kinetics of complexes formed by oppositely charged polyelectrolytes. J Colloid Interface Sci 234:384–392. https://doi.org/10.1006/jcis.2000.7326
Kontturi K, Tammelin T, Johansson L-S, Stenius P (2008) Adsorption of cationic starch on cellulose studied by QCM-D. Langmuir 24:4743–4749. https://doi.org/10.1021/la703604j
Kötz J (1993) Phase behavior of polyanion-polycation aggregates and possibilities of utilization. Nord Pulp Pap Res J 8:11–14
Lin Z, Renneckar S (2011) Nanocomposite-based lignocellulosic fibers 3: polyelectrolyte adsorption onto heterogeneous fiber surfaces. Cellulose 18:563–574. https://doi.org/10.1007/s10570-011-9502-6
Lindberg L, Vähäsalo L, Holmbom B (2004) Flow cytometry of bacteria and wood resin particles in paper production. Nord Pulp Pap Res J 19:412–416. https://doi.org/10.3183/NPPRJ-2004-19-04-p412-416
Lofton M, Moore S, Hubbe M, Lee SY (2005) Deposition of polyelectrolyte complexes as a mechanism for developing paper dry strength. Tappi J 4:3–8
Marais A, Utsel S, Gustafsson E, Wågberg L (2014) Towards a super-strainable paper using the layer-by-layer technique. Carbohydr Polym 100:218–224. https://doi.org/10.1016/j.carbpol.2013.03.049
Mende M, Petzold G, Buchhammer H-M (2002) Polyelectrolyte complex formation between poly(diallyldimethyl-ammonium chloride) and copolymers of acrylamide and sodium-acrylate. Colloid Polym Sci 280:342–351. https://doi.org/10.1007/s00396-001-0614-7
Myllytie P, Yin J, Holappa S, Laine J (2009) The effect of different polysaccharides on the development of paper strength during drying. Nord Pulp Pap Res J 24:469–477. https://doi.org/10.3183/NPPRJ-2009-24-04-p469-477
Petzold G, Lunkwitz K (1995) The interaction between polyelectrolyte complexes made from poly(dimethyldiallylammonium chloride) (PDMDAAC) and poly(maleic acid-co-α-methylstyrene) (P(MS-α-MeSty)) and cellulosic materials. Colloids Surf A 98:225–233. https://doi.org/10.1016/0927-7757(95)03133-X
Petzold G, Buchhammer H-M, Lunkwitz K (1996) The use of oppositely charged polyelectrolytes as flocculants and retention aids. Colloids Surf A 119:87–92. https://doi.org/10.1016/S0927-7757(96)03740-5
Philipp B, Dautzenberg H, Linow K-J, Kötz J, Dawydoff W (1989) Polyelectrolyte complexes—recent developments and open problems. Prog Polym Sci 14:91–172. https://doi.org/10.1016/0079-6700(89)90018-X
Piculell L, Lindman B (1992) Association and segregation in aqueous polymer/polymer, polymer/surfactant, and surfactant/surfactant mixtures: similarities and differences. Adv Colloid Interface Sci 41:149–178. https://doi.org/10.1016/0001-8686(92)80011-L
Renneckar S, Zhou Y (2009) Nanoscale coatings on wood: polyelectrolyte adsorption and layer-by-layer assembled film formation. ACS Appl Mater Interfaces 1:559–566. https://doi.org/10.1021/am800119q
Shapiro H (2003) Practical flow cytometry, 4th edn. Wiley, Hoboken
Shibata I, Isogai A (2003) Depolymerization of cellouronic acid during TEMPO-mediated oxidation. Cellulose 10:151–158
Strand A, Lindqvist H, Vähäsalo L, Blomquist M, Sundberg A (2013) Analysis of interactions between colloidal wood pitch and various mineral particles by flow cytometry. BioResources 8:3884–3900. https://doi.org/10.15376/biores.8.3.3884-3900
Strand A, Khakalo A, Kouko J, Oksanen A, Ketola A, Salminen K, Rojas O, Retulainen E, Sundberg A (2017) The effect of chemical additives on the strength, stiffness and elongation potential of paper. Nord Pulp Pap Res J 32:324–335. https://doi.org/10.3183/NPPRJ-2017-32-03-p324-335
Stratton R (1989) Dependence of sheet properties on the location of adsorbed polymer. Nord Pulp Pap Res J 4:104–112
Sundberg A, Pranovich A, Holmbom B (2000) Distribution of anionic groups in TMP suspensions. J Wood Chem Technol 20:71–92. https://doi.org/10.1080/02773810009349625
Tammelin T, Merta J, Johansson L-S, Stenius P (2004) Viscoelastic properties of cationic starch adsorbed on quartz studied by QCM-D. Langmuir 20:10900–10909. https://doi.org/10.1021/la0487693
Torgnysdotter A, Wågberg L (2006) Tailoring of fibre/fibre joints in order to avoid the negative impacts of drying on paper properties. Nord Pulp Pap Res J 21:411–418. https://doi.org/10.3183/NPPRJ-2006-21-03-p411-418
Vähäsalo L, Holmbom B (2005) Factors affecting white pitch deposition. Nord Pulp Pap Res J 20:164–168. https://doi.org/10.3183/NPPRJ-2005-20-02-p164-168
Vähäsalo L, Degerth R, Holmbom B (2003) The use of flow cytometry in wet end research. Pap Technol 44:45–49
Van de Steeg H, Cohen Stuart M, De Keizer A, Bijsterbosch B (1992) Polyelectrolyte adsorption: a subtle balance of forces. Langmuir 8:2538–2546. https://doi.org/10.1021/la00046a030
Acknowledgments
This work is a part of the project ExtBioNet, appointed by the Academy of Finland. The work is part of the activities of the Process Chemistry Centre (PCC) at Åbo Akademi University. Special thanks to Frida Sjögren, MSc worker at the laboratory of Wood and Paper Chemistry, Åbo Akademi University, for skillful laboratory work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Strand, A., Vähäsalo, L., Ketola, A. et al. In-situ analysis of polyelectrolyte complexes by flow cytometry. Cellulose 25, 3781–3795 (2018). https://doi.org/10.1007/s10570-018-1832-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-018-1832-1