Abstract
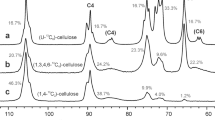
To demonstrate the effectiveness of 13C-detection two-dimensional NMR methods used for the characterization protein structures for the characterization of cellulose derivatives, 13C–13C COSY, 13C–13C INADEQUATE, and 13C–13C TOCSY experiments were applied to 13C-enriched sodium carboxymethyl cellulose and cellulose acetate. These samples were prepared from bacterial cellulose biosynthesized by Gluconacetobacter xylinus ATCC 53582 in medium containing 20 % uniformly 13C-labelled glucose in unlabeled glucose. The combination of 13C–13C COSY (or 13C–13C INADEQUATE) and 13C–13C TOCSY spectra of the cellulose derivatives facilitated the direct and precise assignment of all ring carbons of each anhydroglucose unit comprising the cellulose chain during the short experimental time. In addition, a short spin-lock time (~24 ms) in the 13C–13C TOCSY experiments was revealed to be preferable for achieving improved sensitivity and obtaining correlation signals between all carbons in the anhydroglucose units of the cellulose derivatives.







Similar content being viewed by others
Abbreviations
- 13C:
-
Carbon-13
- 15N:
-
Nitrogen-15
- 1D:
-
One-dimensional
- 1H:
-
Proton
- 2D:
-
Two-dimensional
- AGU:
-
Anhydroglucose unit
- BC:
-
Bacterial cellulose
- CA:
-
Cellulose acetate
- CMC:
-
Carboxymethyl cellulose sodium salt
- COSY:
-
Correlation spectroscopy
- D2O:
-
Deuterium oxide
- DIPSI:
-
Decoupling in the presence of scalar interactions
- DMSO:
-
Dimethyl sulfoxide
- DQ:
-
Double quantum
- DSAC :
-
Substitution degree of acetyl group
- DSCM :
-
Substitution degree of carboxymethyl group
- DSS:
-
4,4-Dimethyl-4-silapentane-1-sulfonic acid
- FID:
-
Free induction decay
- G. xylinus :
-
Gluconacetobacter xylinus
- HSQC:
-
Heteronuclear single quantum coherence
- INADEQUATE:
-
Incredible natural abundance double quantum transfer experiment
- NMR:
-
Nuclear magnetic resonance
- NOESY:
-
Nuclear Overhauser and effect spectroscopy
- S/N:
-
Signal-to-noise ratio
- SQ:
-
Single quantum
- T 2 :
-
Spin–spin relaxation time
- TOCSY:
-
Total correlation spectroscopy
References
Arashida T, Ishino T, Kai K, Hatanaka K, Akaike T, Matsuzaki K, Kaneko Y, Mimura T (1993) Biosynthesis of cellulose from culture media containing 13C-labeled glucose as a carbon source. J Carbohydr Chem 12:641–649. doi:10.1080/07328309308019413
Bax A, Freeman R, Frenkiel TA (1981a) An NMR technique for tracing out the carbon skeleton of an organic molecule. J Am Chem Soc 103:2102–2104. doi:10.1021/ja00398a044
Bax A, Freeman R, Frenkiel TA, Levitt MH (1981b) Assignment of carbon-13 NMR spectra via double-quantum coherence. J Magn Reson 43:478–483. doi:10.1016/0022-2364(81)90060-3
Bax A, Grzesiek S, Gronenborn AM, Clore GM (1994) Isotope-filtered 2D HOHAHA spectroscopy of a peptide-protein complex using heteronuclear Hartmann–Hahn dephasing. J Magn Reson A 106:269–273. doi:10.1006/jmra.1994.1038
Bermel W, Bertini I, Felli IC, Kümmerle R, Pierattelli R (2003) 13C direct detection experiments on the paramagnetic oxidized monomeric copper, zinc superoxide dismutase. J Am Chem Soc 125:16423–16429. doi:10.1021/ja037676p
Bertini I, Felli IC, Kümmerle R, Moskau D, Pierattelli R (2004) 13C–13C NOESY: an attractive alternative for studying large macromolecules. J Am Chem Soc 126:464–465. doi:10.1021/ja0357036
Bromand S, Whalen JK, Janzen HH, Schjoerring JK, Ellert BH (2001) A pulse-labelling method to generate 13C-enriched plant materials. Plant Soil 235:253–257. doi:10.1023/A:1011922103323
Bubb WA (2003) NMR spectroscopy in the study of carbohydrates: characterizing the structural complexity. Concepts Magn Reson A 19A:1–19. doi:10.1002/cmr.a.10080
Bush CA, Martin-Pastor M, Imberty A (1999) Structure and conformation of complex carbohydrates of glycoproteins, glycolipids, and bacterial polysaccharides. Annu Rev Biophys Biomol Struct 28:269–293. doi:10.1146/annurev.biophys.28.1.269
Cavanagh J, Rance M (1992) Suppression of cross-relaxation effects in TOCSY spectra via a modified DIPSI-2 mixing sequence. J Magn Reson 96:670–678. doi:10.1016/0022-2364(92)90357-D
Duus JØ, Gotfredsen CH, Bock K (2000) Carbohydrate structural determination by NMR spectroscopy: modern methods and limitations. Chem Rev 100:4589–4614. doi:10.1021/cr990302n
Eyler RW, Klug ED, Diephuis F (1947) Determination of degree of substitution of sodium carboxymethylcellulose. Anal Chem 19:24–27. doi:10.1021/ac60001a007
Fernández C, Szyperski T, Ono A, Iwai H, Tate S, Kainosho M, Wüthrich K (1998) NMR with 13C, 15N-doubly-labeled DNA: the antennapedia homeodomain complex with a 14-mer DNA duplex. J Biomol NMR 12:25–37. doi:10.1023/A:1008280117211
Fesik SW, Eaton HL, Olejniczak ET, Zuiderweg ERP, McIntosh LP, Dahlquist FW (1990) 2D and 3D NMR spectroscopy employing carbon-13/carbon-13 magnetization transfer by isotropic mixing. Spin system identification in large proteins. J Am Chem Soc 112:886–888. doi:10.1021/ja00158a069
Fontana C, Kovacs H, Widmalm G (2014) NMR structure analysis of uniformly 13C-labeled carbohydrates. J Biomol NMR 59:95–110. doi:10.1007/s10858-014-9830-6
Friedrich MW (2006) Stable-isotope probing of DNA: insights into the function of uncultivated microorganisms from isotopically labeled metagenomes. Curr Opin Biotech 17:59–66. doi:10.1016/j.copbio.2005.12.003
Gitti R, Long G, Bush CA (1994) Measurement of long-range2 13C–1H coupling constants of 95% uniformly 13C-labeled polysaccharide from streptococcus mitis J22. Biopolymers 34:1327–1338. doi:10.1002/bip.360341005
Hikichi K, Kakuta Y, Katoh T (1995) 1H NMR study on substituent distribution of cellulose diacetate. Polym J 27:659–663. doi:10.1295/polymj.27.659
Hofmann K, Hatakeyama H (1994) 1H n.m.r. relaxation studies and lineshape analysis of aqueous sodium carboxymethylcellulose. Polymer 35:2749–2758. doi:10.1016/0032-3861(94)90303-4
Kjellberg A, Nishida T, Weintraub A, Widmalm G (1998) NMR spectroscopy of 13C-enriched polysaccharides: application of 13C–13C TOCSY to sugars of different configuration. Magn Reson Chem 36:128–131. doi:10.1002/(SICI)1097-458X(199802)36:2<128:AID-OMR226>3.0.CO;2-L
Kjellberg A, Weintraub A, Widmalm G (1999) Structural determination and biosynthetic studies of the O-antigenic polysaccharide from the Enterohemorrhagic Escherichia coli O91 using 13C-enrichment and NMR spectroscopy. Biochemistry 38:12205–12211. doi:10.1021/bi9910629
Kono H (2013a) Chemical shift assignment of the complicated monomers comprising cellulose acetate by two-dimensional NMR spectroscopy. Carbohydr Res 375:136–144. doi:10.1016/j.carres.2013.04.019
Kono H (2013b) 1H and 13C chemical shift assignment of the monomers that comprise carboxymethyl cellulose. Carbohydr Polym 97:384–390. doi:10.1016/j.carbpol.2013.05.031
Kono H, Yunoki S, Shikano T, Fujiwara M, Erata T, Takai M (2002a) CP/MAS 13C NMR study of cellulose and cellulose derivatives. 1. Complete assignment of the 13C resonance for the ring carbons of cellulose triacetate polymorphs. J Am Chem Soc 124:7506–7511. doi:10.1021/ja010704o
Kono H, Erata T, Takai M (2002b) CP/MAS 13C NMR study of cellulose and cellulose derivatives. 2. Complete assignment of the 13C resonance for the ring carbons of cellulose triacetate polymorphs. J Am Chem Soc 124:7512–7518. doi:10.1021/ja010705g
Kono H, Erata T, Takai M (2003a) Determination of the through-bond carbon–carbon and carbon–proton connectivities of the native celluloses in the solid state. Macromolecules 36:5132–5138. doi:10.1021/ma021769u
Kono H, Erata T, Takai M (2003b) Complete assignment of the CP/MAS 13C NMR spectrum of cellulose IIII. Macromolecules 36:3589–3592. doi:10.1021/ma021015f
Kono H, Numata Y, Erata T, Takai M (2004) 13C and 1H Resonance assignment of mercerized cellulose II by two-dimensional MAS NMR spectroscopies. Macromolecules 37:5310–5316. doi:10.1021/ma030465k
Kono H, Hashimoto H, Shimizu Y (2015) NMR characterization of cellulose acetate: chemical shift assignments, substituent effects, and chemical shift additivity. Carbohydr Polym 118:91–100. doi:10.1016/j.carbpol.2014.11.004
Machonkin TE, Westler WM, Markley JL (2002) 13C{13C} 2D NMR: a novel strategy for the study of paramagnetic proteins with slow electronic relaxation rates. J Am Chem Soc 124:3204–3205. doi:10.1021/ja017733j
Malm CJ, Tanghe LJ, Laird BC (1946) Preparation of cellulose acetate—action of sulfuric acid. Ind Eng Chem 38:77–82. doi:10.1021/ie50433a033
Martin-Pastor M, Bush CA (1999) New strategy for the conformational analysis of carbohydrates based on NOE and 13C NMR coupling constants. Application to the flexible polysaccharide of Streptococcus mitis J22. Biochemistry 38:8045–8055. doi:10.1021/bi9904205
Miyazaki T, Sato H, Sakakibara T, Kajihara Y (2000) An approach to the precise chemoenzymatic synthesis of 13C-labeled sialyloligosaccharide on an intact glycoprotein: a novel one-pot [3-13C]-labeling method for sialic acid analogues by control of the reversible aldolase reaction, enzymatic synthesis of [3-13C]-NeuAc-α-(2 → 3)-[U-13C]-Gal-β-(1 → 4)-GlcNAc-β-sequence onto glycoprotein, and its conformational analysis by developed NMR techniques. J Am Chem Soc 122:5678–5694. doi:10.1021/ja994211j
Numata Y, Kono H, Kawano S, Erata T, Takai M (2003) Cross-polarization/magic-angle spinning 13C nuclear magnetic resonance study of cellulose I–ethylenediamine complex. J Biosci Bioeng 96:461–466. doi:10.1016/S1389-1723(03)70132-7
Opella ST (2006) Structural biology: designer labels. Nature 440:40. doi:10.1038/440040a
Roudaut G, van Dusschoten D, As HV, Hemminga MA, Meste ML (1998) Mobility of lipids in low moisture bread as studied by NMR. J Cereal Sci 28:147–155. doi:10.1006/jcrs.1998.0196
Schlufter K, Schmauder H-P, Dorn S, Heinze T (2006) Efficient homogeneous chemical modification of bacterial cellulose in the ionic liquid 1-N-butyl-3-methylimidazolium chloride. Macromol Rapid Comm 27:1670–1676. doi:10.1002/marc.200600463
Schramm M, Gromet Z, Hestrin S (1957) Synthesis of cellulose by Acetobacter xylinum. 3. Substrates and inhibitors. Biochem J 67:669–679
Tugarinov V, Kanelis V, Kay LE (2006) Isotope labeling strategies for the study of high-molecular-weight proteins by solution NMR spectroscopy. Nat Protoc 1:749–754. doi:10.1038/nprot.2006.101
Walker TE, London RE, Whaley TW, Barker R, Matwiyoff NA (1976) Carbon-13 nuclear magnetic resonance spectroscopy of [1-13C] enriched monosaccharides. Signal assignments and orientational dependence of geminal and vicinal carbon–carbon and carbon–hydrogen spin–spin coupling constants. J Am Chem Soc 98:5807–5813. doi:10.1021/ja00435a011
Witter R, Sternberg U, Hesse S, Kondo T, Koch F-T, Ulich AS (2006) 13C chemical shift constrained crystal structure refinement of cellulose Iα and its verification by NMR anisotropy experiments. Macromolecules 39:6125–6132. doi:10.1021/ma052439n
Yamaguchi Y, Kato K, Shindo M, Aoki S, Furusho K, Koga K, Takahashi N, Arata Y, Shimada I (1998) Dynamics of the carbohydrate chains attached to the Fc portion of immunoglobulin G as studied by NMR spectroscopy assisted by selective 13C labeling of the glycans. J Biomol NMR 12:385–394. doi:10.1023/A:1008392229694
Yu L, Goldman R, Sullivan P, Walker GF, Fesik SW (1993) Heteronuclear NMR studies of 13C-labeled yeast cell wall β-glucan oligosaccharides. J Biomol NMR 3:429–441. doi:10.1007/BF00176009
Yunoki S, Osada Y, Kono H, Takai M (2004) Role of ethanol in improvement of bacterial cellulose production: Analysis using 13C-labeled carbon sources. Food Sci Technol Res 10:307–313. doi:10.3136/fstr.10.307
Zhou Z, Kümmerle R, Qiu X, Redwine D, Cong R, Taha A, Baugh D, Winniford B (2007) A new decoupling method for accurate quantification of polyethylene copolymer composition and triad sequence distribution with 13C NMR. J Magn Reson 187:225–233. doi:10.1016/j.jmr.2007.05.005
Acknowledgments
This work was supported in part by Grants-in-Aid for Scientific Research C-25410134 from the Japan Society for Promotion of Science (JSPS).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kono, H., Anai, H., Hashimoto, H. et al. 13C-detection two-dimensional NMR approaches for cellulose derivatives. Cellulose 22, 2927–2942 (2015). https://doi.org/10.1007/s10570-015-0697-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-015-0697-9