Abstract
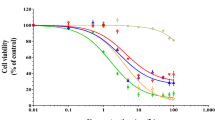
Iron oxide nanoparticles offer unique possibilities due to the change in their physico-chemical parameters when synthesized on the nanoscale (10−9 m) compared to their bulk forms. While novel uses exist for these materials when synthesized as nanoparticles, their unintended effects on the human body and specifically during pregnancy remain ill defined. In this study, an iron oxide nanoparticle, α-Fe2O3, was employed and the potential toxicity due to exposure was assessed in the widely used model human placental cell line BeWo b30. These cells were grown as epithelia, and subsequently assessed for their epithelial integrity, reactive oxygen species production and cellular viability, ultrastructural and morphological disruption, and genotoxicity as a result of exposure to α-Fe2O3 nanoparticles. Transepithelial electrical resistance indicated that exposure to the large (50 and 78 nm), but not small (15 nm) diameter particles of α-Fe2O3 nanomaterial resulted in leakiness of the epithelium. Exposure to the large diameters of 50 and 78 nm resulted in increases in cell death and reactive oxygen species. Disruption of junctional integrity as monitored by immunolocalization of the tight junction protein ZO-1 was found to occur as a consequence of exposure to large diameter NPs. It was found that there was reduction in the number of microvilli responsible for increased surface area for nutrient absorption after exposing the epithelia to large diameter NPs. Finally, genotoxicity as assessed by DNA microarray and confirmed by QPCR indicated that the large diameter particles (78 nm) induce apoptosis in these cells. These data indicate that large (50 and 78 nm), but not small (15 nm) α-Fe2O3 nanoparticles disrupt the barrier function of this epithelium as assessed by in vitro analysis.











Similar content being viewed by others
Abbreviations
- BSA:
-
Bovine serum albumin
- DAPI:
-
4′,6-Diamidino-2-phenylindole
- DLS:
-
Dynamic light scattering
- ICB:
-
Intracellular buffer
- mRNA:
-
Messenger RNA
- NP(s):
-
Nanoparticle
- PBS:
-
Phosphate-buffered saline
- ROS:
-
Reactive oxygen species
- SEM:
-
Scanning electron microscopy
- TEER:
-
Transepithelial electrical resistance
- TEM:
-
Transmission electron microscopy
- ZO-1:
-
Zonula Occludens-1
References
Amstad E, Textor M, Reimhult E. Stabilization and functionalization of iron oxide nanoparticles for biomedical applications. Nanoscale. 2011;3(7):2819–43.
Anderson JM, Van Itallie CM, Peterson MD, Stevenson BR, Carew EA, Mooseker MS. ZO-1 mRNA and protein expression during tight junction assembly in Caco-2 cells. J Cell Biol. 1989;109(3):1047–56.
Aplin J. The cell biology of human implantation. Placenta. 1996;17(5):269–75.
Bement WM, Forscher P, Mooseker MS. A novel cytoskeletal structure involved in purse string wound closure and cell polarity maintenance. J Cell Biol. 1993;121(3):565–78. Epub 1993/05/01.
Berryman M, Gary R, Bretscher A. Ezrin oligomers are major cytoskeletal components of placental microvilli: a proposal for their involvement in cortical morphogenesis. J Cell Biol. 1995;131(5):1231–42.
Bhabra G, Sood A, Fisher B, Cartwright L, Saunders M, Evans WH, et al. Nanoparticles can cause DNA damage across a cellular barrier. Nat Nanotechnol. 2009;4(12):876–83.
Blowes DW, Ptacek CJ, Jambor JL. In-situ remediation of Cr (VI)-contaminated groundwater using permeable reactive walls: laboratory studies. Environ Sci Technol. 1997;31(12):3348–57.
Blume LF, Denker M, Gieseler F, Kunze T. Temperature corrected transepithelial electrical resistance (TEER) measurement to quantify rapid changes in paracellular permeability. Pharmazie. 2010;65:19–24.
Bode CJ, Jin H, Rytting E, Silverstein PS, Young AM, Audus KL. In vitro models for studying trophoblast transcellular transport. Methods Mol Med. 2006;122:225–39. Epub 2006/03/04.
Buyukhatipoglu K, Clyne AM. Superparamagnetic iron oxide nanoparticles change endothelial cell morphology and mechanics via reactive oxygen species formation. J Biomed Mater Res A. 2011;96(1):186–95.
Cartwright L, Poulsen MS, Nielsen HM, Pojana G, Knudsen LE, Saunders M, et al. In vitro placental model optimization for nanoparticle transport studies. Int J Nanomedicine. 2012;7:497–510. Epub 2012/02/16.
Claude P. Morphological factors influencing transepithelial permeability: A model for the resistance of thezonula occludens. J Membr Biol. 1978;39(2–3):219–32.
Denker HW. Implantation: a cell biological paradox. J Exp Zool. 1993;266(6):541–58. Epub 1993/09/01.
Ehrenberg MS, Friedman AE, Finkelstein JN, Oberdörster G, McGrath JL. The influence of protein adsorption on nanoparticle association with cultured endothelial cells. Biomaterials. 2009;30(4):603–10.
Ellis RE, Yuan J, Horvitz H. Mechanisms and functions of cell death. Annu Rev Cell Biol. 1991;7(1):663–98.
Faust JJ, Zhang W, Koeneman BA, Chen Y, Capco DG. Commenting on the effects of surface treated- and non-surface treated TiO(2) in the Caco-2 cell model. Part Fibre Toxicol. 2012;9:42. Epub 2012/11/14.
Fischer U, Jänicke R, Schulze-Osthoff K. Many cuts to ruin: a comprehensive update of caspase substrates. Cell Death Differ. 2003;10(1):76–100.
Fulda S, Debatin K. Extrinsic versus intrinsic apoptosis pathways in anticancer chemotherapy. Oncogene. 2006;25(34):4798–811.
Furuse M, Itoh M, Hirase T, Nagafuchi A, Yonemura S, Tsukita S. Direct association of occludin with ZO-1 and its possible involvement in the localization of occludin at tight junctions. J Cell Biol. 1994;127(6):1617–26.
Gaugain B, Barbet J, Capelle N, Roques BP, Le Pecq JB, Le Bret M. DNA bifunctional intercalators. 2. Fluorescence properties and DNA binding interaction of an ethidium homodimer and an acridine ethidium heterodimer. Appendix: Numerical solution of McGhee and von Hippel equations for competing ligands. Biochemistry. 1978;17:5078–88.
Grummer R, Hohn HP, Mareel MM, Denker HW. Adhesion and invasion of three human choriocarcinoma cell lines into human endometrium in a three-dimensional organ culture system. Placenta. 1994;15(4):411–29. Epub 1994/06/01.
Guo H, Stüben D, Berner Z. Removal of arsenic from aqueous solution by natural siderite and hematite. Appl Geochem. 2007;22(5):1039–51.
Gupta AK, Gupta M. Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications. Biomaterials. 2005;26(18):3995–4021.
Gupta AK, Naregalkar RR, Vaidya VD, Gupta M. Recent advances on surface engineering of magnetic iron oxide nanoparticles and their biomedical applications. Nanomedicine (Lond). 2007;2:23–39.
Hannan NJ, Paiva P, Dimitriadis E, Salamonsen LA. Models for study of human embryo implantation: choice of cell lines? Biol Reprod. 2010;82(2):235–45.
He YT, Wan J, Tokunaga T. Kinetic stability of hematite nanoparticles: the effect of particle sizes. J Nanoparticle Res. 2008;10(2):321–32.
Heintzelman MB, Hasson T, Mooseker MS. Multiple unconventional myosin domains of the intestinal brush border cytoskeleton. J Cell Sci. 1994;107(12):3535–43.
Hidalgo IJ, Raub T, Borchardt R. Characterization of the human colon carcinoma cell line (Caco-2) as a model system for intestinal epithelial permeability. Gastroenterology. 1989;96(3):736.
Huppertz B. Nanoparticles: Barrier thickness matters. Nat Nanotechnol. 2011;6(12):758–9.
John NJ, Linke M, Denker HW. Quantitation of human choriocarcinoma spheroid attachment to uterine epithelial cell monolayers. In Vitro Cell Dev Biol Anim. 1993;29A(6):461–8. Epub 1993/06/01.
Jovov B, Wills NK, Lewis SA. A spectroscopic method for assessing confluence of epithelial cell cultures. Am J Physiol. 1991;261:C1196–203.
Kalive M, Zhang W, Chen Y, Capco DG. Human intestinal epithelial cells exhibit a cellular response indicating a potential toxicity upon exposure to hematite nanoparticles. Cell Biol Toxicol. 2012;28(5):343–68. Epub 2012/08/21.
Kievit FM, Zhang M. Surface engineering of iron oxide nanoparticles for targeted cancer therapy. Acc Chem Res. 2011;44(10):853–62.
King BF. Comparative studies of structure and function in mammalian placentas with special reference to maternal-fetal transfer of iron. Am Zool. 1992;32(2):331–42.
Koeneman BA, Zhang Y, Hristovski K, Westerhoff P, Chen Y, Crittenden JC, et al. Experimental approach for an< i> in vitro</i> toxicity assay with non-aggregated quantum dots. Toxicol In Vitro. 2009;23(5):955–62.
Koeneman BA, Zhang Y, Westerhoff P, Chen Y, Crittenden JC, Capco DG. Toxicity and cellular responses of intestinal cells exposed to titanium dioxide. Cell Biol Toxicol. 2010;26(3):225–38.
Kokkinos MI, Murthi P, Wafai R, Thompson EW, Newgreen DF. Cadherins in the human placenta–epithelial-mesenchymal transition (EMT) and placental development. Placenta. 2010;31(9):747–55. Epub 2010/07/28.
Kreuter J. Drug targeting with nanoparticles. Eur J Drug Metab Pharmacokinet. 1994;19(3):253–6.
Kulvietis V, Zalgeviciene V, Didziapetriene J, Rotomskis R. Transport of nanoparticles through the placental barrier. Tohoku J Exp Med. 2011;225(4):225–34.
Lee N, Tong MK, Leung PP, Chan VW, Leung S, Tam P-C, et al. Kidney claudin-19: localization in distal tubules and collecting ducts and dysregulation in polycystic renal disease. FEBS Lett. 2006;580(3):923.
Li H, van Ravenzwaay B, Rietjens IM, Louisse J. Assessment of an in vitro transport model using BeWo b30 cells to predict placental transfer of compounds. Arch Toxicol. 2013. Epub 2013/05/22.
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods (San Diego, Calif). 2001;25(4):402–8. Epub 2002/02/16.
Mahmoudi M, Sant S, Wang B, Laurent S, Sen T. Superparamagnetic iron oxide nanoparticles (SPIONs): development, surface modification and applications in chemotherapy. Adv Drug Deliv Rev. 2011;63(1):24–46.
Maiorano G, Sabella S, Sorce B, Brunetti V, Malvindi MA, Cingolani R, et al. Effects of cell culture media on the dynamic formation of protein− nanoparticle complexes and influence on the cellular response. ACS Nano. 2010;4(12):7481–91.
Mardon H, Grewal S, Mills K. Experimental models for investigating implantation of the human embryo. Semin Reprod Med. 2007;25:410–7.
Matter K, Aijaz S, Tsapara A, Balda MS. Mammalian tight junctions in the regulation of epithelial differentiation and proliferation. Curr Opin Cell Biol. 2005;17(5):453–8.
Menezes V, Malek A, Keelan AJ. Nanoparticulate drug delivery in pregnancy: placental passage and fetal exposure. Curr Pharm Biotechnol. 2011;12(5):731–42.
Mishra B, Patel BB, Tiwari S. Colloidal nanocarriers: a review on formulation technology, types and applications toward targeted drug delivery. Nanomedicine Nanotechnol Biol Med. 2010;6(1):9–24.
Mooseker MS. Organization, chemistry, and assembly of the cytoskeletal apparatus of the intestinal brush border. Annu Rev Cell Biol. 1985;1(1):209–41.
Morck TJ, Sorda G, Bechi N, Rasmussen BS, Nielsen JB, Ietta F, et al. Placental transport and in vitro effects of Bisphenol A. Reprod Toxicol (Elmsford, NY). 2010;30(1):131–7. Epub 2010/03/11.
Mukherjee T, Squillantea E, Gillespieb M, Shao J. Transepithelial electrical resistance is not a reliable measurement of the Caco-2 monolayer integrity in Transwell. Drug Deliv. 2004;11:11–8.
Mutch DM, Berger A, Mansourian R, Rytz A, Roberts M-A. The limit fold change model: a practical approach for selecting differentially expressed genes from microarray data. BMC Bioinforma. 2002;3(1):17.
Nagata S. Apoptosis by Death Factor Review. Cell. 1997;88(355–365):392.
Pattillo RA, Gey GO. The establishment of a cell line of human hormone-synthesizing trophoblastic cells in vitro. Cancer Res. 1968;28(7):1231–6. Epub 1968/07/01.
Penners N, Koopal L. Preparation and optical properties of homodisperse haematite hydrosols. Colloids Surf. 1986;19(4):337–49.
Pietroiusti A, Campagnolo L, Fadeel B. Interactions of engineered nanoparticles with organs protected by internal biological barriers. Small. 2013;9:1557–72.
Pijnenborg R. Trophoblast invasion and placentation in the human: morphological aspects. Trophoblast Invasion and Endometrial Receptivity: Springer; 1990. p. 33–47.
Quackenbush J. Microarray data normalization and transformation. Nat Genet. 2002;32:496–501.
Rampon C, Bouillot S, Climescu-Haulica A, Prandini M-H, Cand F, Vandenbrouck Y, et al. Protocadherin 12 deficiency alters morphogenesis and transcriptional profile of the placenta. Physiol Genomics. 2008;34(2):193–204.
Reed JC. Mechanisms of apoptosis. Am J Pathol. 2000;157(5):1415–30.
Rytting E, Audus KL. Novel organic cation transporter 2-mediated carnitine uptake in placental choriocarcinoma (BeWo) cells. J Pharmacol Exp Ther. 2005;312(1):192–8. Epub 2004/08/19.
Rytting E, Audus KL. Effects of low oxygen levels on the expression and function of transporter OCTN2 in BeWo cells. J Pharm Pharmacol. 2007;59(8):1095–102. Epub 2007/08/30.
Rytting E, Audus KL. Contributions of phosphorylation to regulation of OCTN2 uptake of carnitine are minimal in BeWo cells. Biochem Pharmacol. 2008;75(3):745–51. Epub 2007/11/06.
Saunders M. Transplacental transport of nanomaterials. Wiley interdisciplinary reviews. Nanomedicine Nanobiotechnol. 2009;1(6):671–84. Epub 2010/01/06.
Semmler-Behnke M, Fertsch S, Schmid G, Wenk A, Kreyling WG. Uptake of 1.4 nm versus 18 nm gold nanoparticles in secondary target organs is size dependent in control and pregnant rats after intratracheal or intravenous application. EuroNanoForum 2007. 2007:102.
Semmler–Behnke M, Kreyling WG, Lipka J, Fertsch S, Wenk A, Takenaka S, et al. Biodistribution of 1.4– and 18–nm Gold Particles in Rats. Small. 2008;4(12):2108–11.
Silverstein SC, Steinman RM, Cohn ZA. Endocytosis. Annu Rev Biochem. 1977;46(1):669–722.
Sood A, Salih S, Roh D, Lacharme-Lora L, Parry M, Hardiman B, et al. Signalling of DNA damage and cytokines across cell barriers exposed to nanoparticles depends on barrier thickness. Nat Nanotechnol. 2011;6(12):824–33.
Stevenson BR, Siliciano JD, Mooseker MS, Goodenough DA. Identification of ZO-1: a high molecular weight polypeptide associated with the tight junction (zonula occludens) in a variety of epithelia. J Cell Biol. 1986;103(3):755–66.
Takeda K, Suzuki K-I, Ishihara A, Kubo-Irie M, Fujimoto R, Tabata M, et al. Nanoparticles transferred from pregnant mice to their offspring can damage the genital and cranial nerve systems. J Health Sci. 2009;55(1):95–102.
Thorburn A. Death receptor-induced cell killing. Cell Signal. 2004;16(2):139–44.
van der Ende A, du Maine A, Simmons CF, Schwartz AL, Strous GJ. Iron metabolism in BeWo chorion carcinoma cells. Transferrin-mediated uptake and release of iron. J Biol Chem. 1987;262(18):8910–6. Epub 1987/06/25.
van der Ende A, du Maine A, Schwartz AL, Strous GJ. Modulation of transferrin-receptor activity and recycling after induced differentiation of BeWo choriocarcinoma cells. Biochem J. 1990;270(2):451–7. Epub 1990/09/01.
Verma A, Stellacci F. Effect of surface properties on nanoparticle–cell interactions. Small. 2010;6:12–21.
Wahajuddin SA. Superparamagnetic iron oxide nanoparticles: magnetic nanoplatforms as drug carriers. Int J Nanomedicine. 2012;7:3445.
Westerhoff P, Zhang Y, Crittenden J, Chen Y. Properties of commercial nanoparticles that affect their removal during water treatment. Nanoscience and Nanotechnology: Environmental and Health Impacts NJ: John Wiley and Sons. 2008:71–90.
Wice B, Menton D, Geuze H, Schwartz AL. Modulators of cyclic AMP metabolism induce syncytiotrophoblast formation< i> in vitro</i> Exp Cell Res. 1990;186(2):306–16.
Wu C, Yin P, Zhu X, OuYang C, Xie Y. Synthesis of hematite (α-Fe2O3) nanorods: diameter-size and shape effects on their applications in magnetism, lithium ion battery, and gas sensors. J Phys Chem B. 2006;110(36):17806–12.
Yamashita K, Yoshioka Y, Higashisaka K, Mimura K, Morishita Y, Nozaki M, et al. Silica and titanium dioxide nanoparticles cause pregnancy complications in mice. Nat Nanotechnol. 2011;6(5):321–8.
Zhang W, Kalive M, Capco DG, Chen Y. Adsorption of hematite nanoparticles onto Caco-2 cells and the cellular impairments: effect of particle size. Nanotechnology. 2010;21(35):355103. Epub 2010/08/10.
Zhang W, Stack AG, Chen Y. Interaction force measurement between< i> E. coli</i> cells and nanoparticles immobilized surfaces by using AFM. Colloids Surf B: Biointerfaces. 2011;82(2):316–24.
Zhang W, Crittenden J, Li K, Chen Y. Attachment Efficiency of Nanoparticle Aggregation in Aqueous Dispersions: Modeling and Experimental Validation. Environ Sci Technol. 2012a;46(13):7054–62.
Zhang W, Hughes J, Chen Y. Impacts of hematite nanoparticle exposure on biomechanical, adhesive, and surface electrical properties of Escherichia coli cells. Appl Environ Microbiol. 2012b;78(11):3905–15.
Zhou F, Kotru S, Pandey R. Pulsed laser-deposited ilmenite–hematite films for application in high-temperature electronics. Thin Solid Films. 2002;408(1):33–6.
Acknowledgments
The authors are indebted to Professor Erik Rytting at UTMB for providing the BeWo b30 cell line employed during this study. Professor Kaushal Rege and Dr. Thrimoorthy Potta are greatly thanked for their helpful discussions of this research. The authors thank Dr. Scott Bingham for his unique willingness to provide assistance in the DNA Core Facility at Arizona State University. The authors thank Mr. David Lowry for his patience training JJF on the scanning scope. All imaging data was procured in the W.M. Keck Bioimaging Facility at Arizona State University. JJF is supported in part by the McKee Award funded by the Delta Sigma Phi Foundation, and the Dr. and Mrs. John Maher Scholarship. This study was partially supported by the US Environmental Protection Agency Science to Achieve Results Program grant RD-83385601 and National Science Foundation Grant CBET-1235166.
Competing interests
The authors declare no competing interests.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental Figure 1
Raw TEER values before percent normalization for epithelia exposed to NPs at concentrations of 100 μg/mL and 10 μg/mL. The histograms indicate that exposure to large diameter NPs result in disruption of TEER. (A) The histogram illustrates the change in TEER after application of different diameters of α-Fe2O3 at a concentration of 100 μg/mL. Both 50- and 78 nm NP treated epithelia follow the same trend, whereas the 15-nm diameter exposure followed the trend of the untreated specimens. (B) Exposure to 10 μg/mL for all α-Fe2O3 diameters tested results in no change compared to the untreated specimens. As indicated in the Methods section, TEER levels off at its maximum value of 40 Ωcm2 3 days after seeding BeWo cells. The NPs were applied after this 3 day culture period which is denoted as t = 0 in the graphs. All experiments were conducted at least three independent times where n = 3 (JPEG 70 kb)
Supplemental Figure 2
Tight junctions, as measured by ZO-1 immunofluorescence, are unperturbed after exposure to 15 nm α-Fe2O3 NPs at a 100 μg/mL concentration at the 1 day time point. After exposure to 15 nm α-Fe2O3 NPs the typical contiguous, honeycomb appearance of ZO-1 is seen in these specimens, albeit with modest discontinuity. (JPEG 86 kb)
Supplemental Figure 3
Morphological analysis of microvilli in 15 nm-treated specimens indicates no change in the number and structure of the microvilli. After exposure to 15 nm NPs the microvilli remain erect and appear to contain a similar number of microvilli compared to controls. (JPEG 32 kb)
Supplemental Table 1
Detailed physico-chemical parameters of the NPs in medium indicates a change in ζ-potential as well as a slight degree of agglomeration over the course of 24 h. In ddH2O the ζ-potential of the nanomaterial remains stable and positive. However, after incubation in electrolyte-containing medium with serum there is a rapid shift to a negative ζ-potential. Further, the peak diameter grows slightly as a function of time. (JPEG 94 kb)
Rights and permissions
About this article
Cite this article
Faust, J.J., Zhang, W., Chen, Y. et al. Alpha-Fe2O3 elicits diameter-dependent effects during exposure to an in vitro model of the human placenta. Cell Biol Toxicol 30, 31–53 (2014). https://doi.org/10.1007/s10565-013-9267-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-013-9267-9