Abstract
Polystyrene supported palladium catalysts were synthesized and their chemical and morphological nature were studied using NMR, XRD, TEM, EDX, and XPS analyses. Using the supported catalyst, the first palladium catalyzed acceptorless dehydrogenative coupling of secondary alcohols in water is reported. This method is green, sustainable, phosphine free, and carried out under aerobic condition. Reusability of the catalyst was shown for both alkylation and quinoline reactions till 7 cycles with marginal decrease in yield. Metal leaching was found to be the cause of decrease in yield in both instances.
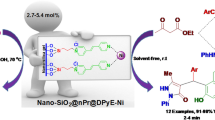
Graphical Abstract
Polystyrene anchored palladium catalysts have been synthesized and used in the acceptorless dehydrogenative coupling of secondary alcohols in aqueous condition. Stability and recyclability of the catalyst was also studied up to 7th cycle in water.






Similar content being viewed by others
References
Selander N, Szabó KJ (2011) Catalysis by palladium pincer complexes. Chem Rev 111(3):2048–2076
Wu X-F, Neumann H, Beller M (2013) Synthesis of heterocycles via palladium-catalyzed carbonylations. Chem Rev 113(1):1–35
Hazari N, Melvin PR, Beromi MM (2017) Well-defined nickel and palladium pre-catalysts for cross-coupling. Nat Rev Chem 1(3):1–16
He J, Wasa M, Chan KS, Shao Q, Yu J-Q (2017) Palladium-catalyzed transformations of alkyl C-H bonds. Chem Rev 117(13):8754–8786
Wang D, Weinstein AB, White PB, Stahl SS (2017) Ligand-promoted palladium-catalyzed aerobic oxidation reactions. Chem Rev 118(5):2636–2679
Devendar P, Qu R-Y, Kang W-M, He B, Yang G-F (2018) Palladium-catalyzed cross-coupling reactions: a powerful tool for the synthesis of agrochemicals. J Agric Food Chem 66(34):8914–8934
Nicolaou K, Bulger PG, Sarlah D (2005) Palladium-catalyzed cross-coupling reactions in total synthesis. Angew Chem Int Ed 44(29):4442–4489
Phan NT, Van Der Sluys M, Jones CW (2006) On the nature of the active species in palladium catalyzed Mizoroki-Heck and Suzuki-Miyaura couplings–homogeneous or heterogeneous catalysis, a critical review. Adv Synth Catal 348(6):609–679
Culkin DA, Hartwig JF (2003) Palladium-catalyzed α-arylation of carbonyl compounds and nitriles. Acc Chem Res 36(4):234–245
Daugulis O, Do H-Q, Shabashov D (2009) Palladium-and copper-catalyzed arylation of carbon− hydrogen bonds. Acc Chem Res 42(8):1074–1086
Brusoe AT, Hartwig JF (2015) Palladium-catalyzed arylation of fluoroalkylamines. J Am Chem Soc 137(26):8460–8468
Sivanandan ST, Shaji A, Ibnusaud I, Seechurn CCJ (2015) Colacot TJ (2015) Palladium-Catalyzed α-Arylation Reactions in Total Synthesis. Eur J Org Chem 1:38–49
Ruiz-Castillo P, Buchwald SL (2016) Applications of palladium-catalyzed C-N cross-coupling reactions. Chem Rev 116(19):12564–12649
Gou B-B, Liu H-F, Chen J, Zhou L (2019) Palladium-catalyzed site-selective C (sp3)–H arylation of phenylacetaldehydes. Org Lett 21(17):7084–7088
Hagui W, Doucet H, Soulé J-F (2019) Application of palladium-catalyzed C (sp2)–H bond arylation to the synthesis of polycyclic (hetero) aromatics. Chem 5(8):2006–2078
Gildner PG, DeAngelis A, Colacot TJ (2016) Palladium-Catalyzed N-Arylation of Cyclopropylamines. Org Lett 18(6):1442–1445
Hong AY (2013) Stoltz BM (2013) The construction of all-carbon quaternary stereocenters by use of Pd-catalyzed asymmetric allylic alkylation reactions in total synthesis. Eur J Org Chem 14:2745–2759
Pritchett BP, Stoltz BM (2018) Enantioselective palladium-catalyzed allylic alkylation reactions in the synthesis of Aspidosperma and structurally related monoterpene indole alkaloids. Nat Prod Rep 35(6):559–574
James J, Jackson M, Guiry PJ (2019) Palladium-catalyzed decarboxylative asymmetric allylic alkylation: development, mechanistic understanding and recent advances. Adv Synth Catal 361(13):3016–3049
Noreen S, Zahoor AF, Ahmad S, Shahzadi I, Irfan A, Faiz S (2019) Novel chiral ligands for palladium-catalyzed asymmetric allylic alkylation/asymmetric Tsuji-Trost reaction: a review. Curr Org Chem 23(11):1168–1213
Premi C, Dixit A, Jain N (2015) Palladium-Catalyzed Regioselective Decarboxylative Alkylation of Arenes and Heteroarenes with Aliphatic Carboxylic Acids. Org Lett 17(11):2598–2601
Yadav S, Ramasastry SSV (2021) Palladium-catalysed annulative allylic alkylation for the synthesis of benzannulated heteroarenes. Chem Commun 57(1):77–80
Trost BM, Brennan MK (2007) Palladium-catalyzed regio-and enantioselective allylic alkylation of bis allylic carbonates derived from Morita−Baylis−Hillman adducts. Org Lett 9(20):3961–3964
Fu L, Chen Q, Wang Z, Nishihara Y (2020) Palladium-catalyzed decarbonylative alkylation of acyl fluorides. Org Lett 22(6):2350–2353
Shen Y, Dai Z-Y, Zhang C, Wang P-S (2021) Palladium-catalyzed allylic alkylation via photocatalytic nucleophile generation. ACS Catal 11(12):6757–6762
Craig RA, Loskot SA, Mohr JT, Behenna DC, Harned AM, Stoltz BM (2015) Palladium-catalyzed enantioselective decarboxylative allylic alkylation of cyclopentanones. Org Lett 17(21):5160–5163
Luo Y-C, Yang C, Qiu S-Q, Liang Q-J, Xu Y-H, Loh T-P (2019) Palladium (II)-Catalyzed Stereospecific Alkenyl C-H Bond Alkylation of Allylamines with Alkyl Iodides. ACS Catal 9(5):4271–4276
Zhu J, Wood J, Deplanche K, Mikheenko I, Macaskie LE (2016) Selective hydrogenation using palladium bioinorganic catalyst. ApplCatal B 199:108–122
Mao Z, Gu H, Lin X (2021) Recent advances of Pd/C-catalyzed reactions. Catalysts 11(9):1078
Liu Y, He S, Quan Z, Cai H, Zhao Y, Wang B (2019) Mild palladium-catalysed highly efficient hydrogenation of C [triple bond, length as m-dash] N, C-NO 2, and C [double bond, length as m-dash] O bonds using H 2 of 1 atm in H 2 O. Green Chem 21(4):830–838
Advani JH, Noor-ul HK, Bajaj HC, Biradar AV (2019) Stabilization of palladium nanoparticles on chitosan derived N-doped carbon for hydrogenation of various functional groups. Appl Surf Sci 487:1307–1315
Vilches-Herrera M, Werkmeister S, Junge K, Börner A, Beller M (2014) Selective catalytic transfer hydrogenation of nitriles to primary amines using Pd/C. Catal Sci Technol 4(3):629–632
Lévay K, Madarász J, Hegedűs L (2022) Tuning the chemoselectivity of the Pd-catalysed hydrogenation of pyridinecarbonitriles: an efficient and simple method for preparing pyridyl-or piperidylmethylamines. Catal Sci Technol 12(8):2634–2648
Guo Y, Li J, Zhao F, Lan G, Li L, Liu Y, Si Y, Jiang Y, Yang B, Yang R (2016) Palladium-modified functionalized cyclodextrin as an efficient and recyclable catalyst for reduction of nitroarenes. RSC Adv 6(10):7950–7954
Hegedűs L, Máthé T (2005) Selective heterogeneous catalytic hydrogenation of nitriles to primary amines in liquid phase: Part I. Hydrogenation of benzonitrile over palladium. Appl Catal A Gen 296(2):209–215
Gligorich KM, Sigman MS (2009) Recent advancements and challenges of palladium II-catalyzed oxidation reactions with molecular oxygen as the sole oxidant. Chem Commun 26:3854–3867
Hu M, Wu W, Jiang H (2019) Palladium-catalyzed oxidation reactions of alkenes with green oxidants. Chemsuschem 12(13):2911–2935
Wu W, Jiang H (2012) Palladium-catalyzed oxidation of unsaturated hydrocarbons using molecular oxygen. Acc Chem Res 45(10):1736–1748
Hess W, Burton JW (2010) Palladium-Catalysed Cyclisation of N-Alkynyl Aminomalonates. Chem Eur J 16(41):12303–12306
Ye J, Ma S (2014) Palladium-catalyzed cyclization reactions of allenes in the presence of unsaturated carbon–carbon bonds. Acc Chem Res 47(4):989–1000
Li J, Yang S, Wu W, Jiang H (2019) Palladium-Catalyzed Cascade Cyclization/Alkynylation Reactions. Chem Asian J 14(23):4114–4128
Wang J, Li D, Li J, Zhu Q (2021) Advances in palladium-catalysed imidoylative cyclization of functionalized isocyanides for the construction of N-heterocycles. Org Biomol Chem 19(31):6730–6745
Zhu C, Zhao Y, Wang D, Sun W-Y, Shi Z (2016) Palladium-catalyzed direct arylation and cyclization of o-iodobiaryls to a library of tetraphenylenes. Sci Rep 6(1):33131
Zou S, Gao B, Huang Y, Zhang T, Huang H (2019) Palladium-catalyzed hydrocarbonylative cyclization of 1, 5-dienes. Org Lett 21(16):6333–6336
Yan F, Liang H, Song J, Cui J, Liu Q, Liu S, Wang P, Dong Y, Liu H (2017) Palladium-catalyzed cyclization-Heck reaction of allenamides: an approach to 3-Methylene-5-phenyl-1, 2, 3, 4-tetrahydropyridine derivatives. Org Lett 19(1):86–89
Liu YZ, Wang Z, Huang Z, Zheng X, Yang WL, Deng WP (2020) Palladium-catalyzed asymmetric [4+3] cyclization of trimethylenemethane: regio-, diastereo-, and enantioselective construction of benzofuro [3, 2-b] azepine skeletons. Angew Chem Int Ed 59(3):1238–1242
Js CC, Kitching M, Colacot T, Snieckus V (2012) Palladium-catalyzed cross-coupling: a historical contextual perspective to the 2010 Nobel Prize. Angew Chem Int Ed 51(21):5062–5085
Rullah K, Mohd Aluwi MFF, Yamin BM, Juan JC, Wai LK (2019) Palladium-catalysed cross-coupling reactions for the synthesis of chalcones. Asian J Org Chem 8(8):1174–1193
Helbert H, Visser P, Hermens JG, Buter J, Feringa BL (2020) Palladium-catalysed cross-coupling of lithium acetylides. Nat Catal 3(8):664–671
Vila Descals C, Giannerini M, Hornillos V, Fañanás-Mastral M, Feringa BL (2014) Palladium-catalysed direct cross-coupling of secondary alkyllithium reagents. Chem Sci 5(4):1361–1367
Sore HF, Galloway WR, Spring DR (2012) Palladium-catalysed cross-coupling of organosilicon reagents. Chem Soc Rev 41(5):1845–1866
Türtscher PL, Davis HJ, Phipps RJ (2018) Palladium-catalysed cross-coupling of benzylammonium salts with boronic acids under mild conditions. Synthesis 50(04):793–802
Wen J-H, Li Q, Nie S-Z, Ye J-J, Xu Q, Zhao C-Q (2018) Palladium-catalyzed isomerization-coupling reactions of allyl chloride with amines to generate functionalized phosphorus derivatives. Catalysts 8(5):194
Ren W, Sun F, Chu J, Shi Y (2020) A Pd-catalyzed site-controlled isomerization of terminal olefins. Org Lett 22(5):1868–1873
Hong-Chao Chen YW, Yang Yu, Wang P (2022) Pd-Catalyzed Isomerization of Alkenes. Chinese J Org Chem 42(3):742–757. https://doi.org/10.6023/cjoc202109045
Kocen AL, Brookhart M, Daugulis O (2017) Palladium-catalysed alkene chain-running isomerization Chem Commun 53(72):10010–10013
Larionov E, Lin L, Guenee L, Mazet C (2014) Scope and mechanism in palladium-catalyzed isomerizations of highly substituted allylic, homoallylic, and alkenyl alcohols. J Am Chem Soc 136(48):16882–16894
Biswal P, Samser S, Meher SK, Chandrasekhar V, Venkatasubbaiah K (2022) Palladium-catalyzed synthesis of α-methyl ketones from allylic alcohols and methanol. Adv Synth Catal 364(2):413–419
Lin L, Romano C, Mazet C (2016) Palladium-catalyzed long-range deconjugative isomerization of highly substituted α, β-unsaturated carbonyl compounds. J Am Chem Soc 138(32):10344–10350
Corma A, Navas J, Ródenas T, Sabater MJ (2013) One-Pot Palladium-catalyzed borrowing hydrogen synthesis of thioethers. Chem Eur J 19(51):17464–17471
Hikawa H, Imamura H, Kikkawa S, Azumaya I (2018) A borrowing hydrogen methodology: palladium-catalyzed dehydrative N-benzylation of 2-aminopyridines in water. Green Chem 20(13):3044–3049
Hikawa H, Koike T, Izumi K, Kikkawa S, Azumaya I (2016) Borrowing hydrogen methodology for N-benzylation using a π-benzylpalladium system in water. Adv Synth Catal 358(5):784–791
Xie Y, Liu S, Liu Y, Wen Y, Deng G-J (2012) Palladium-catalyzed one-pot diarylamine formation from nitroarenes and cyclohexanones. Org Lett 14(7):1692–1695
Dang TT, Ramalingam B, Shan SP, Seayad AM (2013) An efficient palladium-catalyzed N-alkylation of amines using primary and secondary alcohols. ACS Catal 3(11):2536–2540
Shiraishi Y, Fujiwara K, Sugano Y, Ichikawa S, Hirai T (2013) N-monoalkylation of amines with alcohols by tandem photocatalytic and catalytic reactions on TiO2 loaded with Pd nanoparticles. ACS Catal 3(3):312–320
Yu X, Jiang L, Li Q, Xie Y, Xu Q (2012) Palladium-catalyzed N-alkylation of amides and amines with alcohols employing the aerobic relay race methodology. Chin J Chem 30(10):2322–2332
Mamidala R, Mukundam V, Dhanunjayarao K, Venkatasubbaiah K (2017) Cyclometalated palladium pre-catalyst for N-alkylation of amines using alcohols and regioselective alkylation of sulfanilamide using aryl alcohols. Tetrahedron 73(16):2225–2233
Mamidala R, Samser S, Sharma N, Lourderaj U, Venkatasubbaiah K (2017) Isolation and characterization of regioisomers of pyrazole-based palladacycles and their use in α-alkylation of ketones using alcohols. Organometallics 36(17):3343–3351
Mamidala R, Biswal P, Subramani MS, Samser S, Venkatasubbaiah K (2019) Palladacycle-phosphine catalyzed methylation of amines and ketones using methanol. J Org Chem 84(16):10472–10480
Samser S, Mohapatra O, Biswal P, Venkatasubbaiah K (2021) Palladium-mediated tandem isomerization-methylenation of allyl alcohols: one-pot synthesis of 1, 5-diketones. J Org Chem 86(19):13744–13753
Muzart J (2015) Pd-catalyzed hydrogen-transfer reactions from alcohols to C=C, C=O, and C=N Bonds. Eur J Org Chem 2015(26):5693–5707
Ansari TN, Gallou F, Handa S (2020) Cross‐couplings in water: a better way to assemble new bonds organometallic chemistry in industry: A practical approach. p. 203–238
Dixneuf P, Cadierno V (2013) Metal-catalyzed reactions in water. John Wiley & Sons
Lu S-M, Wang Z, Li J, Xiao J, Li C (2016) Base-free hydrogenation of CO 2 to formic acid in water with an iridium complex bearing a N, N′-diimine ligand. Green Chem 18(16):4553–4558
Fujita K-i, Tamura R, Tanaka Y, Yoshida M, Onoda M, Yamaguchi R (2017) Dehydrogenative oxidation of alcohols in aqueous media catalyzed by a water-soluble dicationic iridium complex bearing a functional N-heterocyclic carbene ligand without using base. ACS Catal 7(10):7226–7230
Vivancos A, Beller M, Albrecht M (2018) NHC-based iridium catalysts for hydrogenation and dehydrogenation of N-heteroarenes in water under mild conditions. ACS Catal 8(1):17–21
Huang M, Li Y, Liu J, Lan X-B, Liu Y, Zhao C, Ke Z (2019) A bifunctional strategy for N-heterocyclic carbene-stabilized iridium complex-catalyzed N-alkylation of amines with alcohols in aqueous media. Green Chem 21(2):219–224
Verma A, Hazra S, Dolui P, Elias AJ (2021) Ruthenium-catalyzed synthesis of α-alkylated ketones and quinolines in an aqueous medium via a hydrogen-borrowing atrategy using ketones and alcohols. Asian J Org Chem 10(3):626–633
Lindstrom UM (2008) Organic reactions in water: principles, strategies and applications. John Wiley & Sons
Prat D, Hayler J, Wells A (2014) A survey of solvent selection guides. Green Chem 16(10):4546–4551
Cornils B, Herrmann WA (2004) Aqueous-phase organometallic catalysis: concepts and applications. Aqueous-phase organometallic catalysis: concepts and applications.
Sharma S, Buchbinder NW, Braje WM, Handa S (2020) Fast amide couplings in water: Extraction, column chromatography, and crystallization not required. Org Lett 22(15):5737–5740
Breslow R (2006) The hydrophobic effect in reaction mechanism studies and in catalysis by artificial enzymes. J Phys Org Chem 19(12):813–822
Butler RN, Coyne AG (2016) Organic synthesis reactions on-water at the organic–liquid water interface. Org Biomol Chem 14(42):9945–9960
Lipshutz BH, Ghorai S, Cortes-Clerget M (2018) The hydrophobic effect applied to organic synthesis: recent synthetic chemistry “in water.” Chem Eur J 24(26):6672–6695
Kitanosono T, Masuda K, Xu P, Kobayashi S (2018) Catalytic organic reactions in water toward sustainable society. Chem Rev 118(2):679–746
Benaglia M (2009) Recoverable and recyclable catalysts. John Wiley & Sons
Nasrollahzadeh M, Motahharifar N, Ghorbannezhad F, Bidgoli NSS, Baran T, Varma RS (2020) Recent advances in polymer supported palladium complexes as (nano) catalysts for Sonogashira coupling reaction. Mol Cat 480:110645
Leadbeater NE, Marco M (2002) Preparation of polymer-supported ligands and metal complexes for use in catalysis. Chem Rev 102(10):3217–3274
McNamara CA, Dixon MJ, Bradley M (2002) Recoverable catalysts and reagents using recyclable polystyrene-based supports. Chem Rev 102(10):3275–3300
Drabina P, Svoboda J, Sedlák M (2017) Recent advances in C-C and C–N bond forming reactions catalysed by polystyrene-supported copper complexes. Molecules 22(6):865
Bai L, Wang J-X (2005) Environment friendly Suzuki aryl-aryl cross-coupling reaction. Curr Org Chem 9(6):535–553
Reed-Berendt BG, Latham DE, Dambatta MB, Morrill LC (2021) Borrowing hydrogen for organic synthesis. ACS Cent Sci 7(4):570–585
Irrgang T, Kempe R (2018) 3d-Metal catalyzed N-and C-alkylation reactions via borrowing hydrogen or hydrogen autotransfer. Chem Rev 119(4):2524–2549
Corma A, Navas J, Sabater MJ (2018) Advances in one-pot synthesis through borrowing hydrogen catalysis. Chem Rev 118(4):1410–1459
Wang R, Ma J, Li F (2015) Synthesis of a-alkylated ketones via tandem acceptorless dehydrogenation/a-alkylation from secondary and primary alcohols catalyzed by metal-ligand bifunctional iridium complex [Cp* Ir (2, 2′-bpyO)(H2O)]. J Org Chem 80(21):10769–10776
Musa S, Ackermann L, Gelman D (2013) Dehydrogenative cross-coupling of primary and secondary alcohols. Adv Synth Catal 355(14–15):3077–3080
Sahoo AR, Lalitha G, Murugesh V, Bruneau C, Sharma GV, Suresh S, Achard M (2017) Ruthenium phosphine–pyridone catalyzed cross-coupling of alcohols to form α-alkylated ketones. J Org Chem 82(19):10727–10731
Chang W, Gong X, Wang S, Xiao L-P, Song G (2017) Acceptorless dehydrogenation and dehydrogenative coupling of alcohols catalysed by protic NHC ruthenium complexes. Org Biomol Chem 15(16):3466–3471
Jumde VR, Gonsalvi L, Guerriero A, Peruzzini M (2015) Taddei M (2015) A Ruthenium-Based Catalytic System for a Mild Borrowing-Hydrogen Process. Eur J Org Chem 8:1829–1833
Genç S, Günnaz S, Çetinkaya B, Sl G, Gülcemal D (2018) Iridium (I)-catalyzed alkylation reactions to form α-alkylated ketones. J Org Chem 83(5):2875–2881
Akhtar WM, Cheong CB, Frost JR, Christensen KE, Stevenson NG, Donohoe TJ (2017) Hydrogen borrowing catalysis with secondary alcohols: a new route for the generation of β-branched carbonyl compounds. J Am Chem Soc 139(7):2577–2580
Bhattacharyya D, Sarmah BK, Nandi S, Srivastava HK, Das A (2021) Selective catalytic synthesis of α-alkylated ketones and β-disubstituted ketones via acceptorless dehydrogenative cross-coupling of alcohols. Org Lett 23(3):869–875
Thiyagarajan S, Vijaya Sankar R, Gunanathan C (2020) Ruthenium-catalyzed α-alkylation of ketones using secondary alcohols to β-disubstituted ketones. Org Lett 22(20):7879–7884
Chakraborty P, Garg N, Manoury E, Poli R, Sundararaju B (2020) C-alkylation of various carbonucleophiles with secondary alcohols under CoIII-catalysis. ACS Catal 10(14):8023–8031
Mukundam V, Kumar A, Dhanunjayarao K, Ravi A, Peruncheralathan S, Venkatasubbaiah K (2015) Tetraaryl pyrazole polymers: versatile synthesis, aggregation induced emission enhancement and detection of explosives. Polym Chem 6(44):7764–7770
Eghbali P, Nişancı B, Metin Ö (2018) Graphene hydrogel supported palladium nanoparticles as an efficient and reusable heterogeneous catalysts in the transfer hydrogenation of nitroarenes using ammonia borane as a hydrogen source. Pure Appl Chem 90(2):327–335
Mondal J, Gomes R, Modak A, Bhaumik A (2013) Pd-anchored functionalized mesoporous materials as robust and recyclable heterogeneous catalysts for a series of CC bond forming reactions. Recyclable Catalysis 1:10–33
Demir MM, Gulgun MA, Menceloglu YZ, Erman B, Abramchuk SS, Makhaeva EE, Khokhlov AR, Matveeva VG, Sulman MG (2004) Palladium nanoparticles by electrospinning from poly (acrylonitrile-co-acrylic acid)−PdCl2 solutions. Relations between preparation conditions, particle size, and catalytic activity. Macromolecules 37(5):1787–1792
Ovezova M, Eroğlu Z, Metin Ö, Çetinkaya B, Gülcemal S (2021) Unveiling the catalytic nature of palladium-N-heterocyclic carbene catalysts in the α-alkylation of ketones with primary alcohols. Dalton Trans 50(31):10896–10908
Lisowski W, Keim EG (2010) Vacuum annealing phenomena in ultrathin TiD y/Pd bi-layer films evaporated on Si (100) as studied by TEM and XPS. Anal Bioanal Chem 396(8):2797–2804
Farooq MU, Novosad V, Rozhkova EA, Wali H, Ali A, Fateh AA, Neogi PB, Neogi A, Wang Z (2018) Gold nanoparticles-enabled efficient dual delivery of anticancer therapeutics to HeLa cells. Sci Rep 8(1):1–12
Yu W, Hou H, Xin Z, Niu S, Xie Y, Ji X, Shao L (2017) Nanosizing Pd on 3D porous carbon frameworks as effective catalysts for selective phenylacetylene hydrogenation. RSC Adv 7(25):15309–15314
Li F, Han M, Dai P, Xu W, He J, Tao X, Wu Y, Tong X, Xia X, Guo W (2021) Distinct mechanisms for TMPRSS2 expression explain organ-specific inhibition of SARS-CoV-2 infection by enzalutamide. Nat Commun 12(1):1–14
Acknowledgements
KV thank Department of Atomic Energy (DAE) for financial support. S.S. thanks DST for an INSPIRE fellowship. P.B and SKM thank CSIR for a research fellowship. We thank Marimuthu Rajendiran and Dr. Jiban Krushna Das for XPS measurement and Abhishek Padhy and Ranjit Mishra for TEM and EDX measurement respectively.
Funding
Department of Atomic Energy, Government of India
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shaikh, S., Biswal, P., Meher, S.K. et al. Polystyrene Supported Pyrazole-based Palladium Catalysts/Precatalysts for Acceptorless Dehydrogenative Coupling of Alcohols in Water. Catal Lett 154, 737–748 (2024). https://doi.org/10.1007/s10562-023-04316-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-023-04316-z