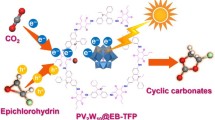
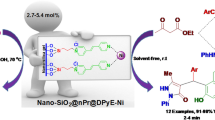
Abstract
Bimetallic Ru:Co nanoparticles stabilized by PEG and imidazolium ionic liquid based [KIT-6] mesoporous silica, denoted as Ru:Co NPs@[KIT-6]-PEG&imid 6, has been synthetized by post grafting method and fully characterized by SEM, TEM, XPS, FT-IR and solid-state 13C and 29Si NMR spectroscopy. The supported Ru:Co mesoporous nanoparticles catalyst 6 revealed excellent catalytic activity (conversions up to 100%), turnover number (TON) of 770 and turnover frequency (TOF) of 7700 h−1 in the Suzuki–Miyaura cross-coupling of p-bromobenzonitrile and phenylboronic acid in H2O:EtOH (1:1) solution under very mild reaction conditions. In addition, the heterogeneous catalyst 6 maintains almost its inherent catalytic activity after the 6th catalytic recycle.
Graphical Abstract






Similar content being viewed by others
References
Miyaura N, Yamada K, Suzuki A (1979) A new stereospecific cross-coupling by the palladium-catalyzed reaction of 1-alkenylboranes with 1-alkenyl or 1-alkynyl halides. Tetrahedron Lett 20:3437–3440
Miyaura N, Suzuki A (1995) Palladium-catalyzed cross-coupling reactions of organoboron compounds. Chem Rev 95:2457–2483
Johansson Seechurn CCC, Kitching MO, Colacot TJ, Snieckus V (2010) Palladium-catalyzed cross-coupling: a historical contextual perspective to the 2010 Nobel Prize. Angew Chem Int Ed 51(2012):5062–5085
Suzuki A (2011) Cross-coupling reactions of organoboranes: an easy way to construct C–C bonds (Nobel Lecture). Angew Chem Int Ed 50:6722–6737
Lasri J, Kopylovich MN, da Silva MFCG, Januário Charmier MA, Pombeiro AJL (2008) Metal-free and PdII-promoted [2+3] cycloadditions of a cyclic nitrone to phthalonitriles: syntheses of oxadiazolines as well as phthalamide–PdII and dihydropyrrolyliminoisoindolinone–PdII complexes with high catalytic activity in Suzuki–Miyaura cross-coupling reactions. Chem Eur J 14:9312–9322
Fernandes RR, Lasri J, da Silva MFCG, Palavra AMF, da Silva JAL, da Silva JJRF, Pombeiro AJL (2011) Oxadiazoline and ketoimine palladium(II) complexes as highly efficient catalysts for Suzuki–Miyaura cross-coupling reactions in supercritical carbon dioxide. Adv Synth Catal 353:1153–1160
Lasri J, da Silva MFCG, Kopylovich MN, Mukhopadhyay S, Januário Charmier MA, Pombeiro AJL (2009) PdII-promoted [2 + 3] cycloaddition of pyrroline N-oxide to organonitriles. Application of (∆4-1,2,4-oxadiazoline)-PdII complexes in the Suzuki–Miyaura reaction. Dalton Trans 2210–2216
Kopylovich MN, Lasri J, da Silva MFCG, Pombeiro AJL (2009) Single-pot template transformations of cyanopyridines on a PdII centre: syntheses of ketoimine and 2,4-dipyridyl-1,3,5-triazapentadiene palladium(II) complexes and their catalytic activity for microwave-assisted Suzuki–Miyaura and Heck reactions. Dalton Trans 3074–3084
Lamblin M, Nassar-Hardy L, Hierso J-C, Fouquet E, Felpin F-X (2010) Recyclable heterogeneous palladium catalysts in pure water: sustainable developments in Suzuki, Heck, Sonogashira and Tsuji-Trost reactions. Adv Synth Catal 352:33–79
Karimi B, Mansouri F, Khorasani M (2016) Recent progress in design and application of functional ordered/periodic mesoporous silicas (OMSs) and organosilicas (PMOs) as catalyst support in carbon-carbon coupling reactions. Curr Org Chem 20:349–380
Giacalone F, Gruttadauria M (2016) Covalently supported ionic liquid phases: an advanced class of recyclable catalytic systems. ChemCatChem 8:664–684
Doherty S, Knight JG, Backhouse T, Abood E, Al-shaikh H, Clemmet AR, Ellison JR, Bourne RA, Chamberlain TW, Stones R, Warren NJ, Fairlamb IAJ, Lovelock KRJ (2018) Heteroatom donor-decorated polymer-immobilized ionic liquid stabilized palladium nanoparticles: efficient catalysts for room-temperature Suzuki–Miyaura cross-coupling in aqueous media. Adv Synth Catal 360:3716–3731
Lasri J, Mac Leod TCO, Pombeiro AJL (2011) Oxadiazoline and ketoimine palladium(II) complexes supported on a chitosan membrane and their catalytic activity for the microwave-assisted Suzuki–Miyaura cross-coupling in water. Appl Catal A 397:94–102
Zhou JY, Liu RQ, Wang CY, Zhu YM (2020) Synthesis of biaryls via decarbonylative nickel-catalyzed Suzuki–Miyaura cross-coupling of aryl anhydrides. J Org Chem 85:14149–14157
Desrosiers JN, Wen J, Tcyrulnikov S, Biswas S, Qu B, Hie L, Kurouski D, Wu L, Grinberg N, Haddad N, Busacca CA, Yee NK, Song JJ, Garg NK, Zhang X, Kozlowski MC, Senanayake CH (2017) Enantioselective nickel-catalyzed Mizoroki–Heck cyclizations to generate quaternary stereocenters. Org Lett 19:3338–3341
Crockett MP, Tyrol CC, Wong AS, Li B, Byers JA (2018) Iron-catalyzed Suzuki–Miyaura cross-coupling reactions between alkyl halides and unactivated arylboronic esters. Org Lett 20:5233–5237
Jiang SP, Dong XY, Gu QS, Ye L, Li ZL, Liu XY (2020) Copper-catalyzed enantioconvergent radical Suzuki–Miyaura C(sp3)–C(sp2) cross-coupling. J Am Chem Soc 142:19652–19659
Babu SG, Neelakandeswari N, Dharmaraj N, Jackson SD, Karvembu R (2013) Copper(II) oxide on aluminosilicate mediated Heck coupling of styrene with aryl halides in water. RSC Adv 3:7774–7781
Kumar LM, Bhat BR (2017) Cobalt pincer complex catalyzed Suzuki–Miyaura cross coupling—a green approach. J Organomet Chem 827:41–48
Hajipour AR, Rezaei F, Khorsandi Z (2017) Pd/Cu-free Heck and Sonogashira cross-coupling reaction by Co nanoparticles immobilized on magnetic chitosan as reusable catalyst. Green Chem 19:1353–1361
Salam N, Kundu SK, Roy AS, Mondal P, Ghosh K, Bhaumik A, Islam SM (2014) A ruthenium-grafted triazine functionalized mesoporous polymer: a highly efficient and multifunctional catalyst for transfer hydrogenation and the Suzuki–Miyaura cross-coupling reactions. Dalton Trans 43:7057–7068
Durap F, Gülen Y, Abay A, Bulut A, Yurderi M, Aydemir M, Baysal A, Zahmakiran M (2021) Bimetallic PdxNi1−x and PdxCo1−x nanoparticles supported on K-OMS-2: highly active, environmentally friendly and reusable nanocatalysts for the Suzuki–Miyaura cross-coupling reactions in water. Appl Organomet Chem 35:e6096
Yamada YM, Takeda K, Takahashi H, Ikegami S (2003) Highly active catalyst for the heterogeneous Suzuki−Miyaura reaction: assembled complex of palladium and non-cross-linked amphiphilic polymer. J Org Chem 68:7733–7741
Durap F, Rakap M, Aydemir M, Özkar S (2010) Room temperature aerobic Suzuki cross-coupling reactions in DMF/water mixture using zeolite confined palladium (0) nanoclusters as efficient and recyclable catalyst. Appl Catal A 382:339–344
Varma RS, Naicker KP, Liesen PJ (1999) Palladium chloride and tetraphenylphosphonium bromide intercalated clay as a new catalyst for the Heck reaction. Tetrahedron Lett 40:2075–2078
Seki M (2006) Recent advances in Pd/C-catalyzed coupling reactions. Synthesis 2006:2975–2992
Zhao F, Bhanage BM, Shirai M, Arai M (2000) Heck reactions of iodobenzene and methyl acrylate with conventional supported palladium catalysts in the presence of organic and/and inorganic bases without ligands. Chem Eur J 6:843–848
Biffis A, Zecca M, Basato M (2001) Metallic palladium in the Heck reaction: active catalyst or convenient precursor? Eur J Inorg Chem 2001:1131–1133
Kilic A, Gezer E, Durap F, Aydemir M, Baysal A (2019) Pd(II) supported dioxime functionalized Fe3O4 nanoparticles as efficient, eco-friendly and reusable catalysts for the Suzuki–Miyaura cross-coupling reaction in water. J Organomet Chem 896:129–138
Polshettiwar V, Molnár A (2007) Silica-supported Pd catalysts for Heck coupling reactions. Tetrahedron 30:6949–6976
Mao M, Lv H, Li Y, Yang Y, Zeng M, Li N, Zhao X (2016) Metal support interaction in Pt nanoparticles partially confined in the mesopores of microsized mesoporous CeO2 for highly efficient purification of volatile organic compounds. ACS Catal 6:418–427
Yamada YM, Sarkar SM, Uozumi Y (2012) Amphiphilic self-assembled polymeric copper catalyst to parts per million levels: click chemistry. J Am Chem Soc 134:9285–9290
Anantharaman SB, Jo K, Jariwala D (2021) Exciton-photonics: from fundamental science to applications. ACS Nano 15:12628–12654
Rostamnia S, Doustkhah E (2014) Nanoporous silica-supported organocatalyst: a heterogeneous and green hybrid catalyst for organic transformations. RSC Adv 4:28238–28248
Mehdi A (2014) Pores functionalized mesoporous silica: an original approach for hybrid catalytic materials. Curr Org Chem 18:2451–2460
Attard GS, Corker JM, Göltner CG, Henke S, Templer RH (1997) Liquid-crystal templates for nanostructured metals. Angew Chem Int Ed 36:1315–1317
Attard GS, Bartlett PN, Coleman NRB, Elliott JM, Owen JR, Wang JH (1997) Mesoporous platinum films from lyotropic liquid crystalline phases. Science 278:838–840
Jiang J, Kucernak A (2003) Electrooxidation of small organic molecules on mesoporous precious metal catalysts: II: CO and methanol on platinum–ruthenium alloy. J Electroanal Chem 543:187–199
Yamauchi Y, Kuroda K (2008) Rational design of mesoporous metals and related nanomaterials by a soft-template approach. Chem Asian J 664:664–676
Yamauchi Y, Yokoshima T, Momma T, Osaka T, Kuroda K (2004) Platinum thin film with a highly ordered mesostructure by contact plating. Chem Lett 33:1576–1577
Boo H, Park S, Ku B, Kim Y, Park JH, Kim HC, Chung TD (2004) Ionic strength-controlled virtual area of mesoporous platinum electrode. J Am Chem Soc 126:4524–4525
Wang C, Chen D, Jiao X (2009) Lyotropic liquid crystal directed synthesis of nanostructured materials. Sci Technol Adv Mater 10:023001
Liu Z, Sakamoto Y, Ohsuna T, Hiraga K, Terasaki O, Ko CH, Shin HJ, Ryoo R (2000) TEM studies of platinum nanowires fabricated in mesoporous silica MCM-41. Angew Chem Int Ed 39:3107–3110
Liu Z, Terasaki O, Ohsuna T, Hiraga K, Shin HJ, Ryoo R (2001) An HREM study of channel structures in mesoporous silica SBA-15 and platinum wires produced in the channels. ChemPhysChem 2:229–231
Shin HJ, Ryoo R, Liu Z, Terasaki O (2001) Template synthesis of asymmetrically mesostructured platinum networks. J Am Chem Soc 123:1246–1247
Doi Y, Takai A, Sakamoto Y, Terasaki O, Yamauchi Y, Kuroda K (2010) Tailored synthesis of mesoporous platinum replicas using double gyroid mesoporous silica (KIT-6) with different pore diameters via vapor infiltration of a reducing agent. Chem Commun 46:6365–6367
Wang H, Jeong HY, Imura M, Wang L, Radhakrishnan L, Fujita N, Castle T, Terasaki O, Yamauchi Y (2011) Shape- and size-controlled synthesis in hard templates: sophisticated chemical reduction for mesoporous monocrystalline platinum nanoparticles. J Am Chem Soc 133:14526–14529
Wang H, Imura M, Nemoto Y, Park SE, Yamauchi Y (2012) Synthesis of olive-shaped mesoporous platinum nanoparticles (MPNs) with a hard-templating method using mesoporous silica (SBA-15). Chem Asian J 7:802–808
Mohammad N, Bepari S, Aravamudhan S, Kuila D (2019) Catalysts kinetics of Fischer–Tropsch synthesis in a 3-D printed stainless steel microreactor using different mesoporous silica supported Co–Ru catalysts. Catalysts 9:872
Udumula V, Tyler JH, Davis DA, Wang H, Linford MR, Minson PS, Michaelis DJ (2015) Dual optimization approach to bimetallic nanoparticle catalysis: impact of M1/M2 ratio and supporting polymer structure on reactivity. ACS Catal 5:3457–3462
Kilic A, Kilinc D, Tas E, Yilmaz I, Durgun M, Ozdemir I, Yasar S (2010) The orthopalladation dinuclear [Pd (L1)(μ-OAc)]2,[Pd (L2)(μ-OAc)]2 and mononuclear [Pd (L3)2] complexes with [N, C, O] or [N, O] containing ligands: synthesis, spectral characterization, electrochemistry and catalytic properties. J Organomet Chem 695:697–706
Tas E, Kilic A, Durgun M, Yilmaz I, Ozdemir I, Gurbuz N (2009) Mono- and dinuclear Pd(II) complexes of different salicylaldimine ligands as catalysts of transfer hydrogenation of nitrobenzene with cyclohexene and Suzuki–Miyaura coupling reactions. J Organomet Chem 694:446–454
Kilic A, Yilmaz I, Ulusoy M, Tas E (2008) Synthesis, spectral characterization, electrochemical studies and catalytic properties in Suzuki–Miyaura coupling reactions of the mononuclear PdII, trinuclear PdII(BPh2)2 and RuII–PdII–RuII type complexes containing 4-amino-1-benzyl piperidine and phenyl groups. Appl Organomet Chem 22:494–502
Kilic A, Durap F, Aydemir M, Baysal A, Tas E (2008) Ru(II) with chelating containing N4-type donor quadridentate Pd-oxime metal complexes: syntheses, spectral characterization, thermal and catalytic properties. J Organomet Chem 693:2835–2842
González-Abrego DO, Zuno-Cruz FJ, Carpio-Granillo M, Andrade-Lopez N, Cruz-Borbolla J, Martinez-Macias C, Mendoza-Espinosa D, Rosales-Hoz MJ, Leyva MA, Torres-Lubian JR, Lopez-Jimenez JA, Jancik V, Sánchez-Cabrera G (2017) Synthetic, spectroscopic and structural behavior of unsaturated functionalized N-heterocyclic carbene complexes of group 11. Polyhedron 137:97–111
Acknowledgements
This project was funded by the Deanship of Scientific Research (DSR) at King Abdulaziz University, Jeddah, under Grant No. (KEP-2-130-42). The authors, therefore, acknowledge with thanks DSR for technical and financial support.
Author information
Authors and Affiliations
Contributions
HAl-s: Experimental work, Formal analysis, Methodology, Funding acquisition. JL: Conceptualization, Validation of the results, Writing the original draft, Funding acquisition. JGK: Review & editing, Supervision.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Al-shaikh, H., Lasri, J. & Knight, J.G. Bimetallic Ru:Co Mesoporous Nanoparticles Stabilized by PEG and Imidazolium Ionic Liquid Based [KIT-6] as an Efficient Heterogeneous Catalyst for Suzuki–Miyaura Cross-Couplings in H2O:EtOH Solution. Catal Lett 152, 3761–3771 (2022). https://doi.org/10.1007/s10562-022-03951-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-022-03951-2