Abstract
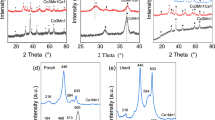
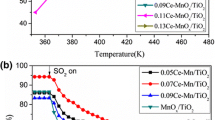
Composite oxide catalysts with Co/Mn/Ce molar ratio of 3:x:2(x = 1, 3, 5 and 7) have been successfully prepared by co-precipitation method. The crystal phase structure, elemental valence, oxygen vacancy and reductivity of the catalysts were characterized by X-ray diffraction (XRD), X-ray photoelectron spectroscopy (XPS), electron paramagnetic resonance (EPR), H2 temperature program reduction(H2-TPR) and in situ DRIFTs. The results demonstrated that when x = 1, the Co–Mn–Ce catalyst had the smallest grain size and oxygen vacancy. A flow reactor experimental system was used to analyze the effect of Mn content on light-off temperature (T10) and complete oxidation temperature (T90) of propane oxidation over Co–Mn–Ce catalysts under anhydrous condition and 5% vol of water vapor. The results showed that when x = 1, the catalyst exhibits the highest activity (T10 = 200 °C, T90 = 307 °C) and water tolerance among the four catalysts. It indicated that when x = 1, the incorporation of Mn content can improve the ability of Co–Mn–Ce catalysts for propane oxidation. Based on the Langmuir–Hinshelwood theory, the surface chemical reaction pathway of propane oxidation was constructed and it revealed that the major active sites of Co–Mn–Ce catalysts mainly depend on surface oxygen vacancy and the surface active species (Mn4+,Oads).
Graphic Abstract











Similar content being viewed by others
References
Guo H, Ling ZH, Cheng HR, Simpson IJ, Lyu XP, Wang XM, Shao M, Lu HX, Ayoko G, Zhang YL, Saunders SM, Lam SHM, Wang JL, Blake DR (2017) Sci Total Environ 574:1021–1043. https://doi.org/10.1016/j.scitotenv.2016.09.116
Riipinen I, Yli-Juuti T, Pierce JR, Petäjä T, Worsnop DR, Kulmala M, Donahue NMJNG (2012) Nat Geosci 5:453–458. https://doi.org/10.1038/ngeo1499
Liu T, Wang X, Deng W, Hu Q, Ding X, Zhang Y, He Q, Zhang Z, Lü S, Bi X, Chen J, Yu J (2015) Atmos Chem Phys 15:9049–9062. https://doi.org/10.5194/acp-15-9049-2015
Everaert K, Baeyens J (2004) J Hazard Mater 109(1–3):113–139. https://doi.org/10.1016/j.jhazmat.2004.03.019
Taylor MN, Zhou W, Garcia T, Solsona B, Carley AF, Kiely CJ, Taylor SH (2012) J Catal 285(1):103–114. https://doi.org/10.1016/j.jcat.2011.09.019
Taylor M, Ndifor EN, Garcia T, Solsona B, Carley AF, Taylor SH (2008) Appl Catal A 350(1):63–70. https://doi.org/10.1016/j.apcata.2008.07.045
Yang JS, Jung WY, Lee GD, Park SS, Jeong ED, Kim HG, Hong SS (2008) J Ind Eng Chem 14(6):779–784. https://doi.org/10.1016/j.jiec.2008.05.008
Li TY, Chiang SJ, Liaw BJ, Chen YZ (2011) Appl Catal B 103(1–2):143–148. https://doi.org/10.1016/j.apcatb.2011.01.020
Genuino HC, Dharmarathna S, Njagi EC, Mei MC, Suib SL (2012) J Phys Chem C 116(22):12066–12078. https://doi.org/10.1021/jp301342f
Bampenrat A, Meeyoo V, Kitiyanan B, Rangsunvigit P, Rirksomboon T (2008) Catal Commun 9(14):2349–2352. https://doi.org/10.1016/j.catcom.2008.05.029
Trovarelli A (1996) Catal Rev 38(4):439–520. https://doi.org/10.1080/01614949608006464
Hu Z, Liu X, Meng D, Guo Y, Guo Y, Lu G (2016) ACS Catal 6(4):2265–2279. https://doi.org/10.1021/acscatal.5b02617
Hu F, Peng Y, Chen J, Liu S, Song H, Li J (2019) Appl Catal B 240:329–336. https://doi.org/10.1016/j.apcatb.2018.06.024
Mo S, Li S, Li J, Peng S, Chen J, Chen Y (2016) Catal Commun 87:102–105. https://doi.org/10.1016/j.catcom.2016.09.017
Zhang W, Hu L, Wu F, Li J (2017) Catal Lett 147(2):407–415. https://doi.org/10.1007/s10562-016-1956-6
Shang DH, Zhong Q, Cai W (2015) Appl Surf Sci 325:211–216. https://doi.org/10.1016/j.apsusc.2014.11.056
Sun YH, Zhang GJ, Xu Y, Zhang RG (2019) Int J Hydrog Energy 44(41):22972–22982. https://doi.org/10.1016/j.ijhydene.2019.07.010
Garcilaso V, Barrientos J, Bobadilla LF, Laguna OH (2019) Renew Energy 132:1141–1150. https://doi.org/10.1016/j.renene.2018.08.080
Konsolakis M, Sgourakis M, Carabineiro SAC (2015) Appl Surf Sci 341:48–54. https://doi.org/10.1016/j.apsusc.2015.02.188
Zou G, Fan Z, Yao X, Zhang Y, Zhang Z, Chen M, Shangguan W (2017) Chin J Catal 38(3):564–572. https://doi.org/10.1016/s1872-2067(17)62758-x
Wang C, Zhang C, Hua W, Guo Y, Lu G, Gil S, Giroir-Fendler A (2017) Chem Eng J 315:392–402. https://doi.org/10.1016/j.cej.2017.01.007
Mukherjee D, Devaiah D, Venkataswamy P, Vinodkumar T, Smirniotis PG, Reddy BM (2018) New J Chem 42:14149–14156. https://doi.org/10.1039/C8NJ01184B
Gómez DM, Galvita VV, Gatica JM, Hilario V, Marin GB (2014) Phys Chem Chem Phys 16(23):11447–11455. https://doi.org/10.1039/c4cp00886c
Li X, Li X, Zeng X, Zhu T (2019) Appl Catal A 572:61–70. https://doi.org/10.1016/j.apcata.2018.12.026
Zhang S, Liu S, Zhu X, Yang Y, Hu W, Zhao H, Qu R, Zheng C, Gao X (2019) Appl Surf Sci 479:1132–1140. https://doi.org/10.1016/j.apsusc.2019.02.118
Kim SC, Shim W (2010) Appl Catal B 98(3–4):180–185. https://doi.org/10.1016/j.apcatb.2010.05.027
Álvarez-Galván MC, dela Peña O’Shea VA, Fierro JLG, Arias PL (2003) Catal Commun 4(5):223–228. https://doi.org/10.1016/s1566-7367(03)00037-2
Castaño MH, Molina R, Moreno S (2017) Mol Catal 443:117–124. https://doi.org/10.1016/j.mcat.2017.09.015
Tang W, Wu X, Li S, Li W, Chen Y (2014) Catal Commun 56:134–138. https://doi.org/10.1016/j.catcom.2014.07.023
Faure B, Alphonse P (2016) Appl Catal B 180:715–725. https://doi.org/10.1016/j.apcatb.2015.07.019
Todorova S, Kolev H, Holgado JP, Kadinov G, Bonev C, Pereñíguez R, Caballero A (2010) Appl Catal B 94(1–2):46–54. https://doi.org/10.1016/j.apcatb.2009.10.019
Cuo Z, Deng Y, Li W, Peng S, Zhao F, Liu H, Chen Y (2018) Appl Surf Sci 456:594–601. https://doi.org/10.1016/j.apsusc.2018.06.207
Wang X, Kang Q, Li D (2008) Catal Commun 9(13):2158–2162. https://doi.org/10.1016/j.catcom.2008.04.021
Wang Y, Deng W, Wang Y, Guo L, Ishihara T (2018) Mol Catal 459:61–70. https://doi.org/10.1016/j.mcat.2018.08.022
Morales M, Barbero B, Cadus L (2006) Appl Catal B 67(3–4):229–236. https://doi.org/10.1016/j.apcatb.2006.05.006
Kim MH, Cho KH, Shin CH, Kang SE, Ham SW (2011) Korean J Chem Eng 28(4):1139–1143. https://doi.org/10.1007/s11814-011-0035-3
Lin J, Guo Y, Chen X, Li C, Lu S, Liew KM (2017) Catal Lett 148(1):181–193. https://doi.org/10.1007/s10562-017-2227-x
Kan J, Deng L, Li B, Huang Q, Zhu S, Shen S, Chen Y (2017) Appl Catal A 530:21–29. https://doi.org/10.1016/j.apcata.2016.11.013
Bae J, Kim BS, Jeong H, Lee H (2019) Mol Catal 467:9–15. https://doi.org/10.1016/j.mcat.2019.01.025
Fuentes RO, Baker RT (2008) J Phys Chem C 113(3):914–924. https://doi.org/10.1021/jp808825c
Liu Y, Dai H, Deng J, Du Y, Xi L, Zhao Z, Wang Y, Gao B, Yang H, Guo G (2013) Appl Catal B 140–141:493–505. https://doi.org/10.1016/j.apcatb.2013.04.051
Tang W, Wu X, Li S, Shan X, Liu G, Chen Y (2015) Appl Catal B 162:110–121. https://doi.org/10.1016/j.apcatb.2014.06.030
Velu S, Shah N, Jyothi TM, Sivasanker S (1999) Microporus Mesoporus Mater 33:61–75. https://doi.org/10.1016/S1387-1811(99)00123-7
Zhao J, Zhang Y, Zhang S, Wang Q, Chen M, Hu T, Meng C (2018) Microporus Mesoporus Mater 268:16–24. https://doi.org/10.1016/j.micromeso.2018.04.009
Song W, Poyraz AS, Meng Y, Ren Z, Chen SY, Suib SL (2014) Chem Mater 26(15):4629–4639. https://doi.org/10.1021/cm502106v
Tang W, Li W, Li D, Liu G, Wu X, Chen Y (2014) Catal Lett 144(11):1900–1910. https://doi.org/10.1007/s10562-014-1340-3
Acknowledgements
This work is supported by the National Natural Science Fund (51676090) and the National Engineering Laboratory Open Fund for Mobile Source Pollution Emission Control Technology (NELMS2018A18) for financial supports.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, P., Cui, C., Li, K. et al. The Effect of Mn Content on Catalytic Activity of the Co–Mn–Ce Catalysts for Propane Oxidation: Importance of Lattice Defect and Surface Active Species. Catal Lett 150, 1505–1514 (2020). https://doi.org/10.1007/s10562-019-03061-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-019-03061-6