Abstract
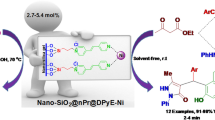
A series of 2-(hetero)arylbenzimidazoles were synthesized by the catalytic condensation of (hetero)aryl aldehydes with 1,2-phenylenediamine derivatives at room temperature in the presence of air as the oxidant. Copper nanoparticles on charcoal was employed as an efficient and mild catalyst for this methodology.
Graphical Abstract



Similar content being viewed by others
References
Tunçbilek M, Kiper T, Altanlar N (2009) Eur J Med Chem 44:1024
Vitale G, Corona P, Loriga M, Carta A, Paglietti G, La Colla P, Busonera B, Marongiu E, Collu D, Loddo R (2009) Med Chem 5:507
Bressi JC, de Jong R, Wu Y, Jennings AJ, Brown JW, O’Connell S, Tari LW, Skene RJ, Vu P, Navre M, Cao X, Gangloff AR (2010) Bioorg Med Chem Lett 20:3138
Tsukamoto G, Yoshino K, Kohno T, Ohtaka H, Kagaya H, Ito K (1980) J Med Chem 23:734
Mederski WWKR, Dorsch D, Anzali S, Gleitz J, Cezanne B, Tsaklakidis C (2004) Bioorg Med Chem Lett 14:3763
Gungor T, Fouquet A, Teulon JM, Provost D, Cazes M, Cloarec A (1992) J Med Chem 35:4455
Navarrete-Vázquez G, Moreno-Diaz H, Aguirre-Crespo F, León-Rivera I, Villalobos-Molina R, Muñoz-Muñiz O, Estrada-Soto S (2006) Bioorg Med Chem Lett 16:4169
Sabnis RW (2008) Handbook of acid–base indicators. CRC Press (Taylor & Francis Group) Boca Raton, London, New York
Pla-Dalmau A (1995) J Org Chem 60:5468
Ueda M, Sato M, Mochizuki A (1985) Macromolecules 18:2723
Phillips MA (1928) J Chem Soc 1928:2393–2399
Hein DW, Alheim RJ, Leavitt JJ (1957) J Am Chem Soc 79:427
Wang R, Lu X–X, Yu X–Q, Shi L, Sun Y (2007) J Mol Cat A: Chem 266:198
Rosen MD, Simon ZM, Tarantino KT, Zhao LX, Rabinowitz MH (2009) Tetrahedron Lett 50:1219
Ben Alloum A, Bougrin K, Soufiaoui M (2003) Tetrahedron Lett 44:5935
Gogoi P, Konwar D (2006) Tetrahedron Lett 47:79
Coppola GM (2008) Synth Commun 38:3500
Han X, Ma H, Wang Y (2008) Russ J Org Chem 44:863
Khan AT, Parvin T, Choudhury LH (2009) Synth Commun 39:2339
Fazaeli R, Aliyan H (2009) Appl Catal A: General 353:74
Gadekar LS, Arbad BR, Lande MK (2010) Chin Chem Lett 21:1053
Luo X, Zhang Z, Yang Y, Xue F, Xiu N, She Y (2009) Front Chem Eng Chin 3:305
Sharghi H, Asemani O, Hossein Tabaei SM (2008) J Heterocyclic Chem 45:1
Sharghi H, Asemani O, Khalifeh R (2008) Synth Commun 38:1128
Sharghi H, Aberi M, Doroodmand MM (2008) Adv Synth Catal 350:2380
Sharghi H, Beyzavi MH, Doroodmand MM (2008) Eur J Org Chem 24:4126–4138
Sharghi H, Hosseini-Sarvari M, Moeini F (2008) Can J Chem 86:1044
Bahrami K, Khodaei MM, Kavianinia I (2007) Synthesis 4:547–550
Rostamizadeh S, Amani AM, Aryan R, Ghaieni HR, Norouzi L (2009) Monatsh Chem 140:547
Saha D, Saha A, Ranu BC (2009) Green Chem 11:733
Dabhade SK, Bora RO, Farooqui M, Gill CH (2009) Chin Chem Lett 20:893
Rostamizadeh S, Aryan R, Ghaieni HR, Amani AM (2009) J Heterocyclic Chem 46:74
Aliyan H, Fazaeli R, Fazaeli N, Mssah AR, Javaherian naghash H, Alizadeh M, Emami G (2009) Heteroatom Chem 20:202
Bahrami K, Khodaei MM, Naali F (2008) J Org Chem 73:6835
Bahrami K, Khodaei MM, Naali F (2009) Synlett 4:569–572
Smith JG, Ho I (1971) Tetrahedron Lett 38:3541
Sharghi H, Khalifeh R, Doroodmand MM (2009) Adv Synth Catal 351:207
Sharghi H, Beyzavi MH, Safavi A, Doroodmand MM, Khalifeh R (2009) Adv Synth Catal 351:2391
Sharghi H, Jokar M (2007) Heterocycles 71:2721
Sharghi H, Khalifeh R (2008) Can J Chem 86:426
Sharghi H, Khalifeh R (2007) Heterocycles 71:1601
Sharghi H, Hosseini Sarvari M (2003) J Org Chem 68:4096
Sharghi H, Jokar M (2010) Can J Chem 88:14
Sharghi H, Khalifeh R, Salimi Beni A (2010) J Iran Chem Soc 7:275
Sharghi H, Salimi Beni AR (2004) Synthesis 17:2900–2904
Sharghi H, Salimi Beni A, Khalifeh R (2007) Helv Chim Acta 90:1373
Sharghi H, Hosseini-Sarvari M, Moeini F, Khalifeh R, Salimi Beni A (2010) Helv Chim Acta 93:435
Sharghi H, Jokar M, Doroodmand MM, Khalifeh R (2010) Adv Syn Cat 352:3031
Sharghi H, Khalifeh R, Moeini F, Beyzavi MH, Salimi Beni A, Doroodmand MM (2011) J Iran Chem Soc 8:S89
Sharghi H, Jokar H, Doroodmand MM (2011) Adv Syn Cat 353:426
Acknowledgments
We gratefully acknowledge the support of this study by the Shiraz University and Shiraz University of Technology Research Council.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Sharghi, H., Khalifeh, R., Mansouri, S.G. et al. Simple, Efficient, and Applicable Route for Synthesis of 2-Aryl(Heteroaryl)-Benzimidazoles at Room Temperature Using Copper Nanoparticles on Activated Carbon as a Reusable Heterogeneous Catalyst. Catal Lett 141, 1845–1850 (2011). https://doi.org/10.1007/s10562-011-0671-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-011-0671-6