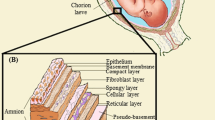
Abstract
The use of fresh amniotic membrane (AM) is not a viable option, as it has many disadvantages. Preserving the AM reduces the risk of cross-infection and maintains its effectiveness for a long time. In order to maximize the therapeutic effects of the AM, the basic need is to preserve its vitality and the bioactive molecules it contains. However, the effect of preservation procedures on cell viability and growth factors is a still matter of debate. Optimum preservation method is expected to be cost-effective, easily-accessible, and most importantly, to preserve the effectiveness of the tissue for the longest time. However, each preservation technique has its advantages and disadvantages over the other, and each one compromises the vitality and bioactive molecules of the tissue to some extent. Therefore, the best method of preservation is still controversial, and the question of ‘how to preserve the AM best?’ has not yet been definitively answered.

Similar content being viewed by others
References
Adds PJ, Hunt CJ, Dart JK (2001) Amniotic membrane grafts, “fresh” or frozen? A clinical and in vitro comparison. Br J Ophthalmol 85:905–907. https://doi.org/10.1136/bjo.85.8.905
Amer MI, Abd-El-Maeboud KHI, Abdelfatah I, Salama FA, Abdallah AS (2010) Human amnion as a temporary biologic barrier after hysteroscopic lysis of severe intrauterine adhesions: pilot study. J Minim Invas Gynecol 17:605–611. https://doi.org/10.1016/j.jmig.2010.03.019
Awan M, Buriak I, Fleck R et al (2020) Dimethyl sulfoxide: a central player since the dawn of cryobiology, is efficacy balanced by toxicity? Regen Med 15:1463–1491. https://doi.org/10.2217/rme-2019-0145
Azuara-Blanco A, Pillai CT, Dua HS (1999) Amniotic membrane transplantation for ocular surface reconstruction. Br J Ophthalmol 83:399–402. https://doi.org/10.1136/bjo.83.4.399
Baradaran-Rafii A, Asl NS, Ebrahimi M et al (2018) The role of amniotic membrane extract eye drop (AMEED) in in vivo cultivation of limbal stem cells. Ocul Surf 16:146–153. https://doi.org/10.1016/j.jtos.2017.11.001
Burman S, Tejwani S, Vemuganti GK, Gopinathan U, Sangwan VS (2004) Ophthalmic applications of preserved human amniotic membrane: a review of current indications. Cell Tissue Bank 5:161–175. https://doi.org/10.1023/B:CATB.0000046067.25057.0a
Davis JS II (1910) Skin grafting at the Johns Hopkins Hospital. Ann Surg 50:542–549. https://doi.org/10.1097/00000658-190909000-00002
De Rötth A (1940) Plastic repair of conjunctival defects with fetal membranes. Arch Ophthalmol 23:522–525. https://doi.org/10.1001/archopht.1940.00860130586006
de Oliveira D, Pena J, de Melo GB, Gomes JAP et al (2007) Ultrastructural and growth factor analysis of amniotic membrane preserved by different methods for ocular surgery. Arq Bras Oftalmol 70:756–762. https://doi.org/10.1590/s0004-27492007000500006
Dhall S, Sathyamoorthy M, Kuang J-Q et al (2018) Properties of viable lyopreserved amnion are equivalent to viable cryopreserved amnion with the convenience of ambient storage. PLoS ONE 13:e0204060. https://doi.org/10.1371/journal.pone.0204060
Dua HS, Gomes JAP, King AJ, Maharajan VS (2004) The amniotic membrane in ophthalmology. Surv Ophthalmol 49:51–77
Duan-Arnold Y, Gyurdieva A, Johnson A, Uveges TE, Jacobstein DA, Danilkovitch A (2015) Retention of endogenous viable cells enhances the anti-inflammatory activity of cryopreserved amnion. Adv Wound Care 4:523–533. https://doi.org/10.1089/wound.2015.0636
Fenelon M, Maurel B, D, Siadous R, et al (2019) Comparison of the impact of preservation methods on amniotic membrane properties for tissue engineering applications. Mater Sci Eng C 104:109903. https://doi.org/10.1016/j.msec.2019.109903
Fenelon M, Catros S, Meyer C et al (2021) Applications of human amniotic membrane for tissue engineering. Membranes (basel) 11(6):1. https://doi.org/10.3390/membranes11060387
Gholipourmalekabadi M, Farhadihosseinabadi B, Faraji M, Nourani MR (2020) How preparation and preservation procedures affect the properties of amniotic membrane? How safe are the procedures? Burns 46:1254–1271
Gindraux F, Laurent R, Nicod L et al (2013) Human Amniotic membrane: Clinical uses, patents and marketed products. Rec Pat Regen Med 3:193–214. https://doi.org/10.2174/22102965113039990021
Hennerbichler S, Reichl B, Pleiner D, Gabriel C, Eibl J, Redl H (2007) The influence of various storage conditions on cell viability in amniotic membrane. Cell Tiss Bank 8:1–8. https://doi.org/10.1007/s10561-006-9002-3
Jiang A, Li C, Gao Y et al (2006) In vivo and in vitro inhibitory effect of amniotic extraction on neovascularization. Cornea 25:S36–S40. https://doi.org/10.1097/01.ico.0000247211.78391.af
Jirsova K, Jones GLA (2017) Amniotic membrane in ophthalmology: properties, preparation, storage and indications for grafting-a review. Cell Tissue Bank 18:193–204. https://doi.org/10.1007/s10561-017-9618-5
Kim JC, Tseng SC (1995) Transplantation of preserved human amniotic membrane for surface reconstruction in severely damaged rabbit corneas. Cornea 14:473–484
Kogan S, Sood A, Granick MS (2018) Amniotic membrane adjuncts and clinical applications in wound healing: a review of the literature. Wounds Compend Clin Res Pract 30:168–173
Koh JW, Shin YJ, Oh JY et al (2007) The expression of TIMPs in cryo-preserved and freeze-dried amniotic membrane. Curr Eye Res 32:611–616. https://doi.org/10.1080/02713680701459441
Koizumi N, Inatomi T, Sotozono C, Fullwood NJ, Quantock AJ, Kinoshita S (2000) Growth factor mRNA and protein in preserved human amniotic membrane. Curr Eye Res 20:173–177
Koob TJ, Lim JJ, Zabek N, Massee M (2015) Cytokines in single layer amnion allografts compared to multilayer amnion/chorion allografts for wound healing. J Biomed Mater Res B Appl Biomater 103:1133–1140. https://doi.org/10.1002/jbm.b.33265
Kruse FE, Joussen AM, Rohrschneider K et al (2000) Cryopreserved human amniotic membrane for ocular surface reconstruction. Graefe’s Arch Clin Exp Ophthalmol 238:68–75. https://doi.org/10.1007/s00417005001240
Kubo M, Sonoda Y, Muramatsu R, Usui M (2001) Immunogenicity of human amniotic membrane in experimental xenotransplantation. Invest Ophthalmol vis Sci 42:1539–1546
Leal-Marin S, Kern T, Hofmann N et al (2021) Human amniotic membrane: a review on tissue engineering, application, and storage. J Biomed Mater Res Part B Appl Biomater 109:1198–1215. https://doi.org/10.1002/jbm.b.34782
Lee SH, Tseng SCG (1997) Amniotic membrane transplantation for persistent epithelial defects with ulceration. Am J Ophthalmol 123:303–312. https://doi.org/10.1016/s0002-9394(14)70125-4
Mahbod M, Shahhoseini S, Khabazkhoob M et al (2014) Amniotic membrane extract preparation: What is the best method? J Ophthalmic vis Res 9:314
Malhotra C, Jain AK (2014) Human amniotic membrane transplantation: different modalities of its use in ophthalmology. World J Transpl 4:111–121. https://doi.org/10.5500/wjt.v4.i2.111
Mamede AC, Carvalho MJ, Abrantes AM, Laranjo M, Maia CJ, Botelho MF (2012) Amniotic membrane: From structure and functions to clinical applications. Cell Tissue Res 349:447–458. https://doi.org/10.1007/s00441-012-1424-6
Maral T, Borman H, Arslan H, Demirhan B, Akinbingol G, Haberal M (1999) Effectiveness of human amnion preserved long-term in glycerol as a temporary biological dressing. Burns 25:625–635. https://doi.org/10.1016/s0305-4179(99)00072-8
McDaniel JS, Wehmeyer JL, Cornell LE, Johnson AJ, Zamora DO (2020) Amniotic membrane allografts maintain key biological properties post SCCO2 and lyophilization processing. J Biomater Appl 35:592–601. https://doi.org/10.1177/0885328220952585
Nakamura T, Yoshitani M, Rigby H et al (2004) Sterilized, freeze-dried amniotic membrane: a useful substrate for ocular surface reconstruction. Invest Ophthalmol vis Sci 45:93–99. https://doi.org/10.1167/iovs.03-0752
Riau AK, Beuerman RW, Lim LS, Mehta JS (2010) Preservation, sterilization and de-epithelialization of human amniotic membrane for use in ocular surface reconstruction. Biomaterials 31:216–225
Ricci E, Vanosi G, Lindenmair A et al (2013) Anti-fibrotic effects of fresh and cryopreserved human amniotic membrane in a rat liver fibrosis model. Cell Tissue Bank 14:475–488. https://doi.org/10.1007/s10561-012-9337-x
Rodríguez-Ares MT, López-Valladares MJ, Touriño R et al (2009) Effects of lyophilization on human amniotic membrane. Acta Ophthalmol 87:396–403. https://doi.org/10.1111/j.1755-3768.2008.01261.x
Shojaku H, Fujisaka M, Takakura H et al (2014) The clinical application of amniotic membrane transplantation in surgery of the ear. Pract Otorhinolaryngol (basel) 107:173–180. https://doi.org/10.5631/jibirin.107.173
Singh R, Gupta P, Kumar P, Kumar A, Chacharkar MP (2003) Properties of air dried radiation processed amniotic membranes under different storage conditions. Cell Tissue Bank 4:95–100. https://doi.org/10.1023/B:CATB.0000007030.72031.12
Sorsby A, Symons HM (1946) Amniotic Membrane Grafts in Caustic Burns of the Eye: (Burns of the second degree). Br J Ophthalmol 30:337–345
Spindler R, Wolkers WF, Glasmacher B (2009) Effect of Me(2)SO on membrane phase behavior and protein denaturation of human pulmonary endothelial cells studied by in situ FTIR spectroscopy. J Biomech Eng 131:74517. https://doi.org/10.1115/1.3156802
Tehrani FA, Ahmadiani A, Niknejad H (2013) The effects of preservation procedures on antibacterial property of amniotic membrane. Cryobiology 67:293–298
Thomasen H, Pauklin M, Steuhl KP, Meller D (2009) Comparison of cryopreserved and air-dried human amniotic membrane for ophthalmologic applications. Graefe’s Arch Clin Exp Ophthalmol 247:1691–1700. https://doi.org/10.1007/s00417-009-1162-y
Tseng SC, Prabhasawat P, Lee SH (1997) Amniotic membrane transplantation for conjunctival surface reconstruction. Am J Ophthalmol 124:765–774. https://doi.org/10.1016/s0002-9394(14)71693-9
von Versen-Höynck F, Syring C, Bachmann S, Möller DE (2004) The influence of different preservation and sterilisation steps on the histological properties of amnion allografts - Light and scanning electron microscopic studies. Cell Tissue Bank 5:45–56. https://doi.org/10.1023/B:CATB.0000022276.47180.96
Wagner M, Walter P, Salla S et al (2018) Cryopreservation of amniotic membrane with and without glycerol additive. Graefe’s Arch Clin Exp Ophthalmol Ophthalmol 256:1117–1126. https://doi.org/10.1007/s00417-018-3973-1
Walkden A (2020) Amniotic membrane transplantation in ophthalmology: an updated perspective. Clin Ophthalmol 14:2057–2072. https://doi.org/10.2147/OPTH.S208008
Witt J, Grumm L, Salla S, Geerling G, Menzel-Severing J (2022) Cryopreservation in a standard freezer: −28 °C as alternative storage temperature for amniotic membrane transplantation. J Clin Med 11:1. https://doi.org/10.3390/jcm11041109
Wolbank S, Hildner F, Redl H, van Griensven M, Gabriel C, Hennerbichler S (2009) Impact of human amniotic membrane preparation on release of angiogenic factors. J Tiss Eng Regen Med 3:651–654. https://doi.org/10.1002/term.207
Acknowledgements
None.
Funding
The authors report there are no financial interests to declare.
Author information
Authors and Affiliations
Contributions
MG, IK and OBS were responsible for the conception or design of the work. MG was responsible for the preparing figures. MG and IK were responsible for drafting the article. OBS was responsible for the critical revision of the article. All authors were responsible for the final approval of the version to be published.
Corresponding author
Ethics declarations
Competing interests
The authors report there are no competing interests to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gurdal, M., Korkmaz, I. & Barut Selver, O. An important detail that is still not clear in amniotic membrane applications: How do we store the amniotic membrane best?. Cell Tissue Bank 25, 339–347 (2024). https://doi.org/10.1007/s10561-023-10121-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-023-10121-0