Abstract
Developmental dysplasia of the hip (DDH) is a debilitating condition that affects 1–7% of newborns. Children with DDH, not treated early and effectively, will easily lead to disability. A better understanding of the biology of DDH is critical to the development of prognostic biomarkers and novel therapies. The purpose of this study was to establish a biobank of DDH genetic resources, to facilitate clinical and basic scientific research. The biological specimen and clinical data of DDH were collected in Shanghai Children’s Hospital from 2014 to 2021. The collection of blood samples was performed at definitive diagnosis and review, tissue specimens were performed at definitive surgery. The clinical data was collected at the whole stage of DDH patients at diagnosis, treatment and follow-up. A total of 528 patients with DDH were enrolled in this study, 90 were men and 438 were women, with the mean age of 4.67 years. The numbers of tissue and blood specimens reached 2172 and 1490, respectively. The quality test results showed that the DNA concentration decreased slightly with the extension of storage time, but the DNA purity did not change. Meanwhile, the extension of storage time slightly affected the stability of protein of tissue samples but did not affect the expression of the housekeeping gene. The DDH biobank built has the potential of monitoring disease pathogenesis and progress, which could provide specimens to the researchers improving the biological understanding and provide guidance of clinical treatment of this disease to clinicians.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Developmental dysplasia of the hip (DDH) is the most common malformation of hip joint in pediatric orthopedics (Pollet et al. 2017; Feldman et al. 2019). The rate of DDH in the neonatal period of North Central Europe (0–28 days) ranges from 1 to 7% (Woodacre et al. 2016). In England, the incidence rate of DDH was 1.28 per 1000 live births (Broadhurst et al. 2019). In China, the average incidence rate of DDH varies from 1 to 10‰, but the incidence rate varies significantly among different races and regions, which is related to genetic factors, environmental impacts, and lifestyle (Pollet et al. 2017). Children with DDH, but not timely implementation of appropriate treatment, will suffer from early-onset osteoarthritis (OA) of the hip, which is easy to lead to disability in severe cases (Tetsunaga et al. 2017). Owing to its high incidence rate and high disability rate, the early screening and treatment of DDH has become the focus and points in pediatric orthopedic and child health care departments (Azzopardi et al. 2011; Shaw et al. 2016a, b). A better understanding of the biology of DDH need to screening and diagnosis early and identify accurate molecular markers and improve patient outcome. Thus, important equipment for understanding the biology and genetic characteristics of DDH is access to patient materials. However, there is no DDH biobank reported in the literature in the world.
In the 1990s, screening of DDH was listed as one of the important items of routine physical examination for infants and young children in Germany, Britain, North America, and other European and American countries (Stein-Zamir et al. 2008). In Britain, Austria and other European countries, screening of DDH was also protected by legal system (Stein-Zamir et al. 2008). In China's first-tier cities such as Shanghai, Beijing and Tianjin, enough attention has also been paid to the early screening of DDH. However, in remote cities such as Tibet, Yunnan and Guizhou, the screening rate and diagnosis and treatment rate of DDH are still low (Li et al. 2013; Engesaeter et al. 2011). The awareness of families in these areas is even lower (Engesaeter et al. 2011). So, in the past years, we screened DDH patients in Shigatse (Tibet) and the eastern, central, and western regions of China, and obtained many clinical data such as epidemiology and imaging. We found that the prevalence of DDH in Shigatse, Tibet was approximately 174.9/1000 infants (106/606) (Zhao et al. 2019), much higher than that in other parts of China. At the same time, we have set up a medical team to follow up Tibet regularly every year to help Tibet establish a DDH early screening, diagnosis and treatment medical team. Therefore, the public welfare project of “gesanghua’s love” has been formed. The above work helped us establish an early DDH clinical data biobank.
DDH is a complex disorder, the etiology of DDH includes both genetic factors and environmental factors (Zhao et al. 2019; Duman et al. 2019). Environmental risk factors include breech presentation, oligohydramnios, primiparity and so on (Chan et al. 1997; Stein-Zamir et al. 2008). Epidemiological studies showed that 67.88% of DDH cases were genetically related (Ceylaner et al. 2008). Some genetic studies had shown that DDH was consistent with some characteristics of autosomal dominant inheritance, but it does not show a simple Mendelian inheritance model (Shi et al. 2012). Maybe because of its complex etiology, DDH often shows incomplete penetrance (Feldman et al. 2013). In a previous study on a large multi-generation family, we identified a variant in bone morphogenetic proteins-2-inducible kinase (BMP2K) known to affect bone metabolism (Zhao et al. 2017) and a nonsense variant in PTGFR (Wang et al. 2018). This termination codon variant in exon 3 of PTGFR gene (c.c922t: p.r308x) located on chromosome 1 may be the pathogenic variant of DDH (Zhao et al. 2017). Although evidence for the genetic cause of DDH has been widely studied and obtained many results, the exact pathogenesis, pathological and molecular mechanisms of DDH remains unclear. Moreover, the incidence of DDH varies greatly in different regions of China. These have caused great deviations to the basic research work. Therefore, the establishment of a multi-component database of patients’ samples of DDH (blood, tissues, and DNA) can effectively change these circumstances, and provide a guarantee for obtaining large research samples in a short time and exploring appropriate treatment strategies based on the natural and original biological characteristics of DDH.
Taken together, the objective of the current study is to establish a biobank of DDH with multi-component samples, to facilitate clinical and basic scientific research. Although the establishment of China's Standardization Organization database has just started, we believe our work will be conducive to standardized DDH biobank establishment. Our biobank was supported by the Orthopedic Department of Shanghai Children’s Hospital affiliated to Shanghai Jiao Tong University, one of the DDH specialized disease treatment centers in China. The DDH biobank we have established is a large sample size biobank publicly reported in China, which will provide valuable information and sample material support for clinical treatment and basic research of DDH.
Materials and methods
Clinical samples of DDH patients were obtained from Shanghai Children’s Hospital Affiliated to Shanghai Jiao Tong University. All samples were collected with the donor being informed completely and with their consent. The procedures were approved by the Institutional Ethical Review Board of the Shanghai Children’s Hospital (2020R043-E01).
Subjects and clinical analysis
Results of clinical exams and radiograph imaging of the hips were evaluated by three pediatric orthopedic surgeons, with clinical opinions of two additional surgeons elicited in any case of disagreement. Radiographic measurements of the hip were taken and affected individuals were identified according to the following criteria: Perkin quadrant (the femoral head is not in the inner lower Perkin quadrant), Acetabular index (> 25°), Shenton’s line (disrupted) and center edge angle (< 20°).
Specimen origins
The collection of the specimens began in 2014. The blood samples were collected at definitive diagnosis and review. The tissue specimens were obtained at the definitive surgery. The clinical data was collected at the whole stage of DDH patients at diagnosis, treatment and follow-up. All data and samples were obtained with the patient's informed consent.
Collection of blood samples
The collection of blood samples was performed at definitive diagnosis and review. 3 mL of the blood sample was collected to the purple top blood collection tube before treatment, and was centrifuged at 3000 g × 10 min to separate plasma and blood cells. Some blood cells are used to extract DNA following the manufacturer's guidelines. Sub package blood cells, DNA, and plasma into small volumes separately and put into tubes, and then store at −80 °C. The workflow of specimens’ collection was shown in Fig. 1.
Collection of tissue specimens
The collection of tissue specimens was performed at definitive surgery. After the surgery schedule was settled, technicians responsible for the specimen collection would be informed. Surgical treatment of DDH patients requires osteotomy. The resected tissue samples, including round ligament, transverse ligament or joint capsule, were firstly examined visually by surgeons, and then the tissue samples were handed over to our technicians for collection. The specimens were cut into small pieces of 1 cm3. A piece in each part is put into cryopreservation tube and then put into the liquid nitrogen tank for quick freezing. The lag between specimen removal and storage should not exceed 30 min. The workflow of specimens’ collection was shown in Fig. 1.
Collection of clinical data
Clinical data were extracted from the electronic databases of Shanghai Children’s Hospital. Most of the stored specimens have corresponding clinical data, including age at enrollment; sex and race; Family history; time to last follow-up. Follow-up was performed every 6 months via outpatient or telephone, up to 3 years. An Excel database was established to record this information and managed by our technicians who received professional training.
The X-ray imaging of DDH patients were performed in parallel with the blood sample collection mentioned above. These image data were also collected in our database, corresponding to tissue specimens.
Sample borrowing process
The borrowers are asked to fill out a request form that includes the following items: the name and institution of the applicant and project leader, the number and type of samples, description of the research project and experiments to be performed. After the submission, our biobank and research office administrators and chief investigator will review the application and sign it. The samples and data must be used only for research as specified in the previous request form and the remaining samples must be returned to our biobank in time. The patient privacy will be protected throughout the whole process.
DNA extraction and the concentration and purity detection
The DNA was extracted from blood cell specimens using the DNeasy Blood Kit (Qiagen, German) following the manufacturer's guidelines. The concentration and purity of DNA extracted from blood were detected by Nanodrop 2000 (Thermo Scientific, USA). Polymerase chain reaction products of β-Actin were amplified with specific primers (Act-F1: atcatgtttgagaccttcaacacc, Act-R1: ccaggaaggaaggctggaagagtg; Act-F2: ctgagcgcaagtactccgtgtgga, Act-R2: ttacacgaaagcaatgctatcacc) and the primers were synthesized by Shanghai Major bio-Co., Ltd.
Protein extraction and housekeeping gene β-Actin expression detection
The whole protein was extracted by RIPA Lysis Buffer (Beyotime Biotechnology, China), and the concentration was detected by BCA protein assay kit (Beyotime Biotechnology, China). Cell lysates were kept on ice for 30 min and centrifuged at 16,000 g for 3–5 min at 4 °C. Supernatants were collected and boiled in 5 × SDS loading buffer, the same amounts of protein were separated by 10% SDS-PAGE and stained by Coomassie or blotted onto polyvinylidene fluoride membranes (Millipore, USA). The primary antibody used in the experiment was β-Actin mouse monoclonal antibody (ab8226, Abcam, USA).
Results
Specimen and clinical data collection
A total of 528 patients with DDH were enrolled in this study from 2014 to 2021. Of these patients, 90 were men and 438 were women. The mean age at diagnosis of DDH was 4.67 years. Among these patients, Tibetans accounted for the majority up to 400, and the rest were Han patients up to 128. The number of DNA was 892. The numbers of tissue and blood specimens reached 2172 and 1490, respectively. The corresponding clinical and demographic data were collected in our electronic database. The characteristics of the biobank are shown in Table 1.
Both clinical data and X-ray imaging data of DDH patients were performed in parallel with the specimen sample collection mentioned above. These image data were also collected in our database, corresponding to tissue specimens (Fig. 2).
DNA extraction and the concentration and purity detection
The stored DNA samples were subpackaged into small volumes and put into tubes, so the number reached 892. The DNA sequencing was performed on these samples to observe the genomic alteration of DDH.
To prove that the samples stored for many years are still valid and available, we selected blood samples from different storage times (1, 3, 5 and 7 years), to extracted DNA and detect their concentration and purity. There are 20 blood samples out of the biobank, with an average storage time of 5 samples. The concentration and purity of DNA extracted from blood were detected by Nanodrop 2000 (Thermo Scientific, USA). We found that the storage time did not affect the purity of DNA, but the concentration of DNA slightly decreased with the extension of storage time (Table 2, Fig. 3). The results of nucleic acid gel electrophoresis showed that the DNA samples of different storage times were intact and unbroken (Fig. 4).
Protein extraction and housekeeping gene β-Actin expression detection
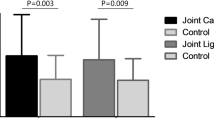
To prove that the stored tissue samples can be used for protein experimental research, we selected the samples stored at different times and extracted the total protein. There are 20 tissue samples out of the biobank from different storage times (1, 3, 5 and 7 years). We found that the concentration of protein slightly decreased with the extension of storage time (Fig. 5). We also detected the expression of housekeeping gene β-Actin of protein samples at different storage times. The result showed that the storage time did not remarkedly affect the expression of β-actin (Fig. 6). However, the result of SDS-PAGE showed that some bands stained with Coomassie bright blue in protein samples stored for 1 and 3 years disappeared in protein samples stored for 5 and 7 years, as shown by the red arrow in Fig. 6. The above results indicated that with the extension of storage time, it would affect the stability of protein in tissue samples, but had no effect on the expression of housekeeping gene.
Correlative studies and published manuscripts
The purpose of our DDH biobank is to facilitate clinical and basic scientific research. Therefore, the quality of this biobank could be evaluated by correlative studies and the resulting publications that included data from our biobank. Table 3 lists the published manuscripts that have used these samples. These studies have contributed to the discovery of more biological characteristics of DDH. These samples in our biobank have played an important role in these studies and contributed to the advance in clinical and basic scientific research of DDH.
Discussion
DDH is the most common malformation of the hip joint in pediatric orthopedics. Children with DDH, not treated early and effectively, will easily lead to disability. A better understanding of the biology of DDH is critical to the development of prognostic biomarkers and novel therapies. Nevertheless, there is still no reported biobank of DDH in the world. Several biobanks of bone-related diseases including bone health, bone quality and fracture, rheumatoid arthritis, osteoporosis, and so on, have been created worldwide (Darling et al. 2018; Kringelbach et al. 2016; Zahra et al. 2021). However, these studies mainly focus the light on the collection of clinical data not biospecimen. The existing research has not formed a unified standard for the construction of biobank, and the guidance for the establishment of DDH biobank is still lacking. Thus, we established a DDH biobank with multi-component samples including biological specimens and clinical data, to facilitate clinical and basic scientific research.
In the collection of the biobank from 2014 to 2021, 528 patients were included and the corresponding clinical data were obtained in the eastern, central, and western regions of China. This biobank will help to clarify the incidence rate and difference of DDH in different regions of China. The etiology of DDH is complicated and has a close relationship with environment and lifestyle (Palomäki et al. 2020; Harcke et al. 1999; Hatzikotoulas et al. 2018). Therefore, the incidence rate of DDH is obviously different in different ears of China. By analyzing the clinical information of DDH patients screened, we found that among all 528 DDH patients, 400 Tibetans (75.76%) and 128 Han people (24.24%). This result showed that remote area such as Tibet is more likely to induce DDH due to altitude, climate, horse herding, infant swaddling, low medical level, and so on. Additionally, 528 patients included 438 women (82.95%) and 90 men (17.05%). DDH presents high prevalence in Asians, and females are more frequently involved with a sex ratio of 4–10:1 (Li et al. 2017), female gender is the most important risk factor for DDH (Li et al. 2017; Ortiz et al. 2012; Talbot et al. 2013; Shaw et al. 2016a, b). Laxity induced by maternal hormones and limited in utero hip mobility also are the leading causes. In infants with DDH, abnormally increased laxity of the hip capsule and surrounding ligaments have been attributed to the effects of maternal hormone relaxin and a higher concentration of estrogen receptors (Desteli et al. 2013).
Besides environment and lifestyle, genetic factors are also one of the main causes of DDH. Certain chromosomes, genes, loci, and polymorphisms are being associated with variable severity of this disorder (Stefan et al. 2020). To reveal the background of DDH heredity, multiple studies focused on large sample sizes with an emphasis on the correlation between genotype, phenotype, and continuous clinical examination (38). By sequencing and analyzing the blood DNA samples of DDH patients, the researchers found some genes may be the pathogenic genes of DDH (Mabuchi et al. 2006; Feldman et al. 2014). In our biobank, the acquisition of 1490 blood, 2172 tissue specimens and 892 DNA samples will greatly contribute to research and a better understanding of disease pathogenesis, genetic polymorphisms. Therefore, many cross-institutional specimens and data from wider regional areas in China not only meet the sufficiency and diversity of samples, but also ensure the reliability of the research results.
It should be noted that our DDH biobank still has some limitations. Considering the incidence characteristics of DDH, in addition to blood and tissue samples, the inclusion of patients' feces in the sample bank project is helpful to study the relationship between intestinal flora and eating habits and the incidence. Even if the specimen of our biobank includes wider areas of China, increasing communication of samples and data may expose patient privacy to the risk of disclosure, and stronger confidentiality protection will be required.
Collectively, we have established a biobank, the first and largest DDH biobank publicly reported in China, with biological specimen and clinical data, to facilitate clinical and basic scientific research. The establishment of China Standardization Organization database is still in its infancy, we believe our work will be conducive to standardized DDH biobank establishment.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Azzopardi T, Van Essen P, Cundy PJ et al (2011) Late diagnosis of developmental dysplasia of the hip: an analysis of risk factors. J Pediatr Orthop B 20(1):1–7. https://doi.org/10.1097/BPB.0b013e3283415927
Broadhurst C, Rhodes AML, Harper P, Perry DC, Clarke NMP, Aarvold A (2019) What is the incidence of late detection of developmental dysplasia of the hip in England? Bone Joint J 101:281–287. https://doi.org/10.1302/0301-620X.101B3.BJJ-2018-1331.R1
Ceylaner G, Ceylaner S, Ustunkan F, Inan M (2008) Autosomal dominant inheritance of congenital dislocation of the hip in 16 members of a family. Acta Orthop Traumatol Turc 42:289–291. https://doi.org/10.3944/aott.2008.289
Chan A, McCaul KA, Cundy PJ, Haan EA, Byron-Scott R (1997) Perinatal risk factors for developmental dysplasia of the hip. Arch Dis Child Fetal Neonatal Ed 76:F94–F100. https://doi.org/10.1136/fn.76.2.f94
Darling A, Blackbourn D, Ahmadi K, Lanham-New S (2018) Bone health and dietary, lifestyle and anthropometric factors in urban dwelling South Asians (n 2438): data from the UK Biobank cohort. Osteoporos Int 29:624–624. https://doi.org/10.1017/S0029665117002877
Desteli EE, Piskin A, Gulman AB, Kaymaz F, Koksal B, Erdogan M (2013) Estrogen receptors in hip joint capsule and ligamentum capitis femoris of babies with developmental dysplasia of the hip. Acta Orthop Traumatol Turc 47:158–161. https://doi.org/10.3944/aott.2013.2772
Duman S, Camurcu Y, Sofu H, Ucpunar H, Akbulut D, Yildirim T (2019) Arthroscopic versus open, medial approach, surgical reduction for developmental dysplasia of the hip in patients under 18 months of age. Acta Orthop 90:292–296. https://doi.org/10.1080/17453674.2019.1599775
Engesaeter IO, Lehmann T, Laborie LB, Lie SA, Rosendahl K, Engesaeter LB (2011) Total hip replacement in young adults with hip dysplasia: age at diagnosis, previous treatment, quality of life, and validation of diagnoses reported to the norwegian arthroplasty register between 1987 and 2007. Acta Orthop 82:149–154. https://doi.org/10.3109/17453674.2011.566146
Feldman GJ, Parvizi J, Levenstien M, Scott K, Erickson JA, Fortina P, Devoto M, Peters CL (2013) Developmental dysplasia of the hip: linkage mapping and whole exome sequencing identify a shared variant in CX3CR1 in all affected members of a large multigeneration family. J Bone Miner Res 28:2540–2549. https://doi.org/10.1002/jbmr.1999
Feldman GJ, Parvizi J, Sawan H, Erickson JA, Peters CL (2014) Linkage mapping and whole exome sequencing identify a shared variant in CX3CR1 in a large multi-generation family. J Arthroplasty 29:238–241. https://doi.org/10.1016/j.arth.2014.05.014
Feldman G, Kappes D, Mookerjee-Basu J, Freeman T, Fertala A, Parvizi J (2019) Novel mutation in teneurin 3 found to co-segregate in all affecteds in a multi-generation family with developmental dysplasia of the hip. J Orthop Res 37:171–180. https://doi.org/10.1002/jor.24148
Harcke HT (1999) Developmental dysplasia of the hip: a spectrum of abnormality. Pediatrics 103:152. https://doi.org/10.1542/peds.103.1.152
Hatzikotoulas K, Roposch A, Consortium D, Shah KM, Clark MJ, Bratherton S, Limbani V, Steinberg J, Zengini E, Warsame K et al (2018) Genome-wide association study of developmental dysplasia of the hip identifies an association with GDF5. Commun Biol 1:56. https://doi.org/10.1038/s42003-018-0052-4
Kringelbach T, Glintborg B, Johansen J, Høgdall E, Hetland ML (2016) The nationwide danish rheumatologic biobank paving the road to personalized treatment of patients with inflammatory rheumatic disease. Ann Rheum Dis 75:1262–1262. https://doi.org/10.1136/annrheumdis-2016-eular.2666
Li L, Sun K, Zhang L et al (2013) Heritability and sibling recurrent risk of developmental dysplasia of the hip in Chinese population. Eur J Clin Invest 43(6):589–594. https://doi.org/10.1111/eci.12084
Li L, Wang X, Zhao Q, Wang E, Wang L, Cheng J et al (2017) CX3CR1 polymorphisms associated with an increased risk of developmental dysplasia of the hip in human. J Orthop Res 35:377–380. https://doi.org/10.1002/jor.23294
Mabuchi A, Nakamura S, Takatori Y, Ikegawa S (2006) Familial osteoarthritis of the hip joint associated with acetabular dysplasia maps to chromosome 13q. Am J Hum Genet 79:163–168. https://doi.org/10.1086/505088
Ortiz-Neira CL, Paolucci EO, Donnon T (2012) A meta-analysis of common risk factors associated with the diagnosis of developmental dysplasia of the hip in newborns. Eur J Radiol 81:e344–e351. https://doi.org/10.1016/j.ejrad.2011.11.003
Palomäki A, Paltta J, Pirilä L, Heikkilä HK, Isomäki P, Huhtakangas J, Sokka-Isler T, Kaipiainen SO (2020) Validity of rheumatoid arthritis diagnosis in Finnish biobank patients. Ann Rheum Dis 79:1913–1913. https://doi.org/10.1080/03009742.2021.1967047
Pollet V, Percy V, Prior HJ (2017) Relative risk and incidence for developmental dysplasia of the hip. J Pediatr 181:202–207. https://doi.org/10.1016/j.jpeds.2016.10.017
Shaw BA, Segal LS, Section O (2016a) Evaluation and referral for developmental dysplasia of the hip in infants. Pediatrics. https://doi.org/10.1542/peds.2016-3107
Shaw BA, Segal LS, Section O (2016b) Evaluation and referral for developmental dysplasia of the hip in infants. Pediatrics 138(3107):e1–e10. https://doi.org/10.1542/peds.2016-3107
Shi D, Dai J, Ikegawa S, Jiang Q (2012) Genetic study on developmental dysplasia of the hip. Eur J Clin Invest 42:1121–1125. https://doi.org/10.1111/j.1365-2362.2012.02682.x
Stefan H, Radoslav Z, Milan K, Lubos D (2020) Genetics of developmental dysplasia of the hip. Eur J Med Genet 63(9):103990. https://doi.org/10.1016/j.ejmg.2020.103990
Stein-Zamir C, Volovik I, Rishpon S et al (2008) Developmental dysplasia of the hip: risk markers, clinical screening, and outcome. Pediatr Int 50(3):341–345. https://doi.org/10.1111/j.1442-200X.2008.02575.x
Talbot CL, Paton RW (2013) Screening of selected risk factors in developmental dysplasia of the hip: an observational study. Arch Dis Child 98:692–696. https://doi.org/10.1136/archdischild-2013-303647
Tetsunaga T, Fujiwara K, Endo H, Tetsunaga T, Shiota N, Sato T, Ozaki T (2017) Calcar femorale in patients with osteoarthritis of the hip secondary to developmental dysplasia. Clin Orthop Surg 9:413–419. https://doi.org/10.4055/cios.2017.9.4.413
Wang S, Jiao Q, Zhou ZW, Wu J, Fan LY, Chen MJ, Wang YC, Mang F, Ying H, Zhao LH (2018) Whole genome exon sequencing of developmental dislocation of the hip in a Chinese family. Chin J Pediatr Surg 39:11–17. https://doi.org/10.3760/cma.j.issn.0253-3006.2018.03.003
Woodacre T, Ball T, Cox P (2016) Epidemiology of developmental dysplasia of the hip within the UK: refining the risk factors. J Child Orthop 10:633–642. https://doi.org/10.1007/s11832-016-0798-5
Zahra RE, Luca B, Jackie C et al (2021) Poor bone quality is associated with greater arterialstiffness: insights from the UK biobank. J Bone Min Res 36:90–99. https://doi.org/10.1002/jbmr.4164
Zhao L, Jiao Q, Chen M, Wang Y, Fan L, MA F, Ying H, Wang S (2017) Expression of PTGFR gene in articular capsule and round ligament of children with DDH and its correlation with THE pathogenesis of DDH. Chongqing Med 11. https://doi.org/10.3969/j.issn.1671-8348.2017.33.002
Zhao L, Ma Q, Feng X, Fan L, Jiao Q, Wang S, Ying H, Yang X (2019) Screening for developmental dysplasia of the hip in infants in tibet identifies increased prevalence associated with altitude. Med Sci Monit 25:5771–5775. https://doi.org/10.12659/MSM.916456
Acknowledgements
The authors are grateful for the participation of the family involved, to whom the present study is dedicated. The present study was sponsored by Natural Science Foundation of Shanghai (No. 20ZR1446600), Youth Project of Medical Engineering cross Research Fundation of Shanghai Jiao Tong University (No. YG2021QN115), National Major Science and Technology Infrastructure Project of Translational Medicine of Shanghai Jiao Tong University (No. TMSK-2020-123) and Medicine, Multi-center Clinical Research Project of Shanghai Jiao Tong University (No. DLY201825).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yang, D., Wang, S., Ke, C. et al. Establishment of pediatric developmental dysplasia of the hip biobank: Shanghai children’s hospital experience. Cell Tissue Bank 23, 581–590 (2022). https://doi.org/10.1007/s10561-022-09995-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-022-09995-3