Abstract
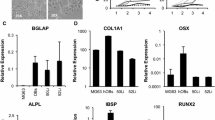
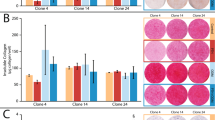
The murine preosteoblastic cell line, MC3T3-E1, is widely used to study bone formation and differentiation in vitro. However, this cell line is unstable in culture. The current study was designed to establish a stable osteoblastic cell line. A mammalian expression vector carrying the SV 40 large T antigen was introduced into a primary culture of cells isolated from the calvaria of newborn mice. Among isolated cell lines, the MN16 cell line was selected for further characterization. The MN16 cell line was cultured for 28 days, and compared with the MC3T3-E1 cell line with or without induction. The expression of bone-related genes was examined using the real-time RT-PCR technique. Alizarin red and von Kossa staining were used to detect mineralization of nodules in the cultures. The cell line showed the characteristics of osteoblastic cells in term of gene expression patterns of various molecular markers and calcium deposition in the cell layer after induction. Furthermore, the MN16 cells showed strong adhesion to the basic domain of collagen, a result that is specific for bone-derived cells. The MN16 cell line was found to be stably differentiated into bone formation cells in vitro and should be useful for studying bone biology.





Similar content being viewed by others
Abbreviations
- DMEM:
-
Dulbecco modified Eagle’s medium
- α-MEM:
-
Modified Eagle’s medium
- SDS:
-
Sodium dodecyl sulfate
References
Aubin JE, Heersche JNH (2002) Osteoprogenitor cell differentiation to mature bone-forming osteoblasts. Drug Dev Res 49:206–215
Baba TT (2000) Restoration of mineral depositions by dexamethasone in the matrix of nonmineralizing osteoblastic cells subcloned from MC3T3-E1 cells. Calcif Tissue Int 67:416–421
Bellows CG, Aubin JE, Heersche JN, Antosz ME (1986) Mineralized bone nodules formed in vitro from enzymatically released rat calvaria cell populations. Calcif Tissue Int 38:143–154
Choi J-Y, Lee B-H, Song K-B, Park R-W, Kim I-S, Sohn K-Y, Jo J-S, Ryoo H-M (1996) Expression patterns of bone-related proteins during osteoblastic differentiation in MC3T3-E1 cells. J Cell Biochem 61:609–618
Czekanska EM, Stoddart MJ, Richards RG, Hayes JS (2012) In search of an osteoblast cell model for in vitro research. Eur Cells Mater 24:1–17
Dahl LK (1952) A simple and sensitive histochemical method for calcium. Proc Soc Exp Biol Med 80:474–479
Gruenert DC, Basbaum CB, Welsh MJ, Li M, Finkbeiner WE, Nadel JA (1988) Characterization of human tracheal cells transformed by an origin-defective simian virus 40. Proc Natl Acad Sci USA 85:5951–5955
Hinoi E, Fujimoto S, Takemori A, Yoneda Y (2002) Cell death by pyruvate deficiency in proliferative cultured calvarial osteoblasts. Biochem Bioph Res Co 294:1177–1183
Karsenty G (2003) The complexities of skeletal biology. Nature 423:316–318
Komori T, Kishimoto T (1998) Cbfa1 in bone development. Curr Opin Genet Dev 8:494–499
Nakashima K, Zhou X, Kunkel G, Zhaoping Z, Deng J-M, Behringer RR, de Crombrugghe B (2002) The novel zinc finger-containing transcription factor Osterix is required for osteoblast differentiation and bone formation. Cell 108:17–29
Page RB, Stromberg AJ (2011) Linear methods for analysis and quality control of relative expression ratios from quantitative real-time polymerase chain reaction experiments. Sci World J 11:1383–1393
Puchtler H, Meloan SN (1978) Demonstration of phosphates in calcium deposits: amodification of von Kossa’s reaction. Histochemistry 56:177–185
Sudo H, Kodama H, Amagai Y, Yamamoto S, Kasai S (1983) In vito differentiation and calcification in a new clonal osteogenic cell line derived from newborn mouse calvaria. J Cell Biol 96:191–198
Yamaguchi K, Matsuo N, Sumiyoshi H, Fujimoto N, Iyama K-I, Yanagisawa S, Yoshioka H (2005) Pro-α3(V) collagen chain is expressed in bone and its basic N-terminal peptide adheres to osteosarcoma cells. Matrix Biol 24:283–294
Acknowledgments
We thank Dr. M. Watanabe, Mr. S. Imi, Ms A. Yasuda and the staff members of Divisions of Life science Research, Laboratory Animal Science and Radioisotope Research, Department of Research Support, Research Promotion Project, Oita University. This work was supported by Grants-In-Aid for Scientific Research (No. 20390402 to H.Y.) from the Ministry of Education, Culture, Sports, Science, and Technology of Japan.
Conflict of interest
The authors report no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nakamura-Ota, M., Hamanaka, R., Yano, H. et al. A new murine osteoblastic cell line immortalized with the SV40 large T antigen. Cell Tissue Bank 15, 373–380 (2014). https://doi.org/10.1007/s10561-013-9394-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-013-9394-9