Abstract
Purpose
Vein graft failure (VGF) is an important limitation for coronary artery bypass graft (CABG) surgery. Inhibition of the excessive proliferation and migration of venous smooth muscle cells (SMCs) is an effective strategy to alleviate VGF during the CABG perioperative period. In the present study, we aimed to explore the role and potential mechanism of all-trans retinoic acid (ATRA) on preventing vein grafts stenosis.
Methods
The autogenous vein grafts model was established in the right jugular artery of rabbits. Immunohistochemistry staining and western blot assays were used to detected the protein expression, while real-time PCR assay was applied for mRNAs expression detection. The interaction between proteins was identified by co-immunoprecipitation assay. The Cell Counting Kit-8 and wound-healing assays were used to investigate the role of ATRA on human umbilical vein smooth muscle cells (HUVSMCs) function. Cell cycle progression was identified by flow cytometry assay.
Results
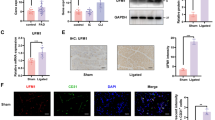
Vein graft stenosis and SMCs hyperproliferation were confirmed in vein grafts by histological and Ki-67 immunohistochemistry assays. Treatment of ATRA (10 mg/kg/day) significantly mitigated the stenosis extent of vein grafts, demonstrated by the decreased thickness of intima-media, and decreased Ki-67 expression. ATRA could repress the PDGF-bb-induced excessive proliferation and migration of HUVSMCs, which was mediated by Rb-E2F dependent cell cycle inhibition. Meanwhile, ATRA could reduce the interaction between KLF5 and RARα, thereby inhibiting the function of cis-elements of KLF5. KLF5-induced inducible nitric oxide synthase (iNOS) expression activation could be significantly inhibited by ATRA.
Conclusions
These results suggested that ATRA treatment may represent an effective prevention and therapy avenue for VGF.





Similar content being viewed by others
References
Woodward LC, Antoniades C, Taggart DP. Intraoperative vein graft preservation: what is the solution? Ann Thorac Surg. 2016;102(5):1736–46.
de Vries MR, Simons KH, Jukema JW, Braun J, Quax PH. Vein graft failure: from pathophysiology to clinical outcomes. Nat Rev Cardiol. 2016;13(8):451–70.
Azzalini L, Jolicoeur EM, Pighi M, Millan X, Picard F, Tadros VX, et al. Epidemiology, management strategies, and outcomes of patients with chronic Total coronary occlusion. Am J Cardiol. 2016;118(8):1128–35.
Ghyselinck NB, Duester G. Retinoic acid signaling pathways. Development. 2019;146(13).
Rhinn M, Dolle P. Retinoic acid signalling during development. Development. 2012;139(5):843–58.
Nedelec B, Rozet JM, Fares Taie L. Genetic architecture of retinoic-acid signaling-associated ocular developmental defects. Hum Genet. 2019;138(8–9):937–55.
Wendland K, Niss K, Kotarsky K, Wu NYH, White AJ, Jendholm J, et al. Retinoic acid signaling in thymic epithelial cells regulates thymopoiesis. J Immunol. 2018;201(2):524–32.
Pogoda HM, Riedl-Quinkertz I, Lohr H, Waxman JS, Dale RM, Topczewski J, et al. Direct activation of chordoblasts by retinoic acid is required for segmented centra mineralization during zebrafish spine development. Development. 2018;145(9).
Lokman NA, Ho R, Gunasegaran K, Bonner WM, Oehler MK, Ricciardelli C. Anti-tumour effects of all-trans retinoid acid on serous ovarian cancer. J Exp Clin Cancer Res. 2019;38(1):10.
Ueki S, Nishikawa J, Yamauchi Y, Konno Y, Tamaki M, Itoga M, et al. Retinoic acids up-regulate functional eosinophil-driving receptor CCR3. Allergy. 2013;68(7):953–6.
Tran-Lundmark K, Tannenberg P, Rauch BH, Ekstrand J, Tran PK, Hedin U, et al. Perlecan Heparan sulfate is required for the inhibition of smooth muscle cell proliferation by all-trans-retinoic acid. J Cell Physiol. 2015;230(2):482–7.
Axel DI, Frigge A, Dittmann J, Runge H, Spyridopoulos I, Riessen R, et al. All-trans retinoic acid regulates proliferation, migration, differentiation, and extracellular matrix turnover of human arterial smooth muscle cells. Cardiovasc Res. 2001;49(4):851–62.
Wang GK, Li SH, Zhao ZM, Liu SX, Zhang GX, Yang F, et al. Inhibition of heat shock protein 90 improves pulmonary arteriole remodeling in pulmonary arterial hypertension. Oncotarget. 2016;7(34):54263–73.
Wang Y, Zhao ZM, Zhang GX, Yang F, Yan Y, Liu SX, et al. Dynamic autophagic activity affected the development of thoracic aortic dissection by regulating functional properties of smooth muscle cells. Biochem Biophys Res Commun. 2016;479(2):358–64.
Zhang XH, Zheng B, Han M, Miao SB, Wen JK. Synthetic retinoid Am80 inhibits interaction of KLF5 with RAR alpha through inducing KLF5 dephosphorylation mediated by the PI3K/Akt signaling in vascular smooth muscle cells. FEBS Lett. 2009;583(8):1231–6.
Lv XR, Zheng B, Li SY, Han AL, Wang C, Shi JH, et al. Synthetic retinoid Am80 up-regulates apelin expression by promoting interaction of RARalpha with KLF5 and Sp1 in vascular smooth muscle cells. Biochem J. 2013;456(1):35–46.
Povoleri GAM, Nova-Lamperti E, Scotta C, Fanelli G, Chen YC, Becker PD, et al. Human retinoic acid-regulated CD161(+) regulatory T cells support wound repair in intestinal mucosa. Nat Immunol. 2018;19(12):1403–14.
Gianni M, Terao M, Kurosaki M, Paroni G, Brunelli L, Pastorelli R, et al. S100A3 a partner protein regulating the stability/activity of RARalpha and PML-RARalpha in cellular models of breast/lung cancer and acute myeloid leukemia. Oncogene. 2019;38(14):2482–500.
Nguyen PH, Giraud J, Staedel C, Chambonnier L, Dubus P, Chevret E, et al. All-trans retinoic acid targets gastric cancer stem cells and inhibits patient-derived gastric carcinoma tumor growth. Oncogene. 2016;35(43):5619–28.
Danzl K, Messner B, Doppler C, Nebert C, Abfalterer A, Sakic A, et al. Early inhibition of endothelial retinoid uptake upon myocardial infarction restores cardiac function and prevents cell, tissue, and animal death. J Mol Cell Cardiol. 2019;126:105–17.
Takeda K, Ichiki T, Funakoshi Y, Ito K, Takeshita A. Downregulation of angiotensin II type 1 receptor by all-trans retinoic acid in vascular smooth muscle cells. Hypertension. 2000;35(1 Pt 2):297–302.
Wakino S, Kintscher U, Kim S, Jackson S, Yin F, Nagpal S, et al. Retinoids inhibit proliferation of human coronary smooth muscle cells by modulating cell cycle regulators. Arterioscler Thromb Vasc Biol. 2001;21(5):746–51.
Kosaka C, Sasaguri T, Komiyama Y, Takahashi H. All-trans retinoic acid inhibits vascular smooth muscle cell proliferation targeting multiple genes for cyclins and cyclin-dependent kinases. Hypertens Res. 2001;24(5):579–88.
Shi JH, Zheng B, Chen S, Ma GY, Wen JK. Retinoic acid receptor alpha mediates all-trans-retinoic acid-induced Klf4 gene expression by regulating Klf4 promoter activity in vascular smooth muscle cells. J Biol Chem. 2012;287(14):10799–811.
Fujiu K, Manabe I, Ishihara A, Oishi Y, Iwata H, Nishimura G, et al. Synthetic retinoid Am80 suppresses smooth muscle phenotypic modulation and in-stent neointima formation by inhibiting KLF5. Circ Res. 2005;97(11):1132–41.
Azami T, Waku T, Matsumoto K, Jeon H, Muratani M, Kawashima A, et al. Klf5 maintains the balance of primitive endoderm versus epiblast specification during mouse embryonic development by suppression of Fgf4. Development. 2017;144(20):3706–18.
Zhao C, Li Y, Qiu W, He F, Zhang W, Zhao D, et al. C5a induces A549 cell proliferation of non-small cell lung cancer via GDF15 gene activation mediated by GCN5-dependent KLF5 acetylation. Oncogene. 2018;37(35):4821–37.
Nagai R, Suzuki T, Aizawa K, Shindo T, Manabe I. Significance of the transcription factor KLF5 in cardiovascular remodeling. J Thromb Haemost. 2005;3(8):1569–76.
Lim HJ, Lee S, Park JH, Lee KS, Choi HE, Chung KS, et al. PPAR delta agonist L-165041 inhibits rat vascular smooth muscle cell proliferation and migration via inhibition of cell cycle. Atherosclerosis. 2009;202(2):446–54.
Meng F, Qian J, Yue H, Li X, Xue K. SUMOylation of Rb enhances its binding with CDK2 and phosphorylation at early G1 phase. Cell Cycle. 2016;15(13):1724–32.
Lo-Coco F, Cicconi L, Breccia M. Current standard treatment of adult acute promyelocytic leukaemia. Br J Haematol. 2016;172(6):841–54.
Kanai F, Obi S, Fujiyama S, Shiina S, Tamai H, Mochizuki H, et al. An open-label phase I/II study of tamibarotene in patients with advanced hepatocellular carcinoma. Hepatol Int. 2014;8(1):94–103.
Boada C, Zinger A, Tsao C, Zhao P, Martinez JO, Hartman K, et al. Rapamycin-loaded biomimetic nanoparticles reverse vascular inflammation. Circ Res. 2020;126(1):25–37.
Zou J, Zhang X, Yang H, Zhu Y, Ma H, Wang S. Rapamycin-loaded nanoparticles for inhibition of neointimal hyperplasia in experimental vein grafts. Ann Vasc Surg. 2011;25(4):538–46.
Zechel C. Synthetic retinoids dissociate coactivator binding from corepressor release. J Recept Signal Transduct Res. 2002;22(1–4):31–61.
Wang N, Zou Q, Xu J, Zhang J, Liu J. Ligand binding and heterodimerization with retinoid X receptor alpha (RXRalpha) induce farnesoid X receptor (FXR) conformational changes affecting coactivator binding. J Biol Chem. 2018;293(47):18180–91.
Funding
This work was supported by the National Natural Science Foundation of China (81470592 and 81800341) and the Science Foundation of Shanghai Municipal Health Planning Commission (20164Y0277).
Author information
Authors and Affiliations
Contributions
Z.X., G. W., and X. L. conceived and designed the experiments. Y. Y., Y. W., and X. F. performed the experiments. Y. Y., Z. S., and F. X. analyzed and confirmed the data. F. Y. performed the statistical analysis. G. W. and Y. Y. wrote the manuscript.
Corresponding authors
Ethics declarations
All animal procedures were approved by the Animal Ethics Committee of Changhai Hospital, Naval Medical University. All experiments were performed in strict compliance with requirements of the Animal Ethics Procedures and Guidelines of China.
Conflict of Interest
The authors declare no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 49 kb)
Rights and permissions
About this article
Cite this article
Yu, Y., Wang, Y., Fei, X. et al. All-Trans Retinoic Acid Prevented Vein Grafts Stenosis by Inhibiting Rb-E2F Mediated Cell Cycle Progression and KLF5-RARα Interaction in Human Vein Smooth Muscle Cells. Cardiovasc Drugs Ther 35, 103–111 (2021). https://doi.org/10.1007/s10557-020-07089-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-020-07089-4