Abstract
Aim
To evaluate the protective effects of allicin on Ang II-induced cardiac hypertrophy.
Methods
Sprague–Dawley male rats were randomized into 3 groups:1)sham group (saline)(n = 12), 2) Ang II group(n = 9), 3) allicin group (Ang II + allicin)(n = 9). They received infusions of either saline or Ang II (250 ng/kg body weight per min) through mini-osmotic pumps implanted subcutaneously for 2 weeks and given a diet containing 180 mg/kg/day of allicin for 8 consecutive weeks. Hemodynamic, morphological, histological, and biochemical changes were evaluated at corresponding time points.
Results
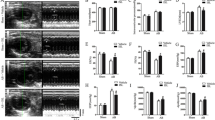
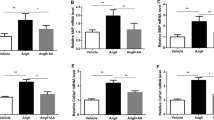
Ang II infusion increased blood pressure, heart rate and heart weight to body weight ratio, and resulted in anatomical and functional changes, such as increased LV mass, posterior wall thickness and LV end-diastolic diameter, and decreased fractional shortening and EF compared with sham rats. Nrf2 and HO-1 in the hearts of rats in the Ang II group were moderately elevated at both mRNA and protein levels compared to sham group mice, but NQO1 andγ-GCS were significantly lower. GPx activities, levels of GSH and T-AOC in the hearts of the rats in the Ang II group were also significantly lower, and the levels of TBARS, reactive oxygen species and protein carbonyl were significant increased. Allicin attenuated LV mass, posterior wall thickness and LV end-diastolic diameter (1.10 ± 0.04 vs. 1.37 ± 0.05, 2.26 ± 0.08 vs. 2.96 ± 0.12, 7.27 ± 0.36 vs. 8.56 ± 0.41, respectively; all P < 0.05), and increased fractional shortening and EF (28.30 ± 3.21 vs. 25.40 ± 2.57, 60.27 ± 5.63 vs. 51.30 ± 4.78, respectively; both P < 0.05) in the Ang II-induced hypertrophic rats compared to the untreated Ang II rats. Furthermore, allicin treatment attenuated the accumulation of interstitial collagen and collagen I/III (P < 0.01 vs. the untreated Ang II group), decreased the levels of reactive oxygen species, protein carbonyl and TBARS and increased GPx activities. Moreover, allicin significantly increased mRNA expression and protein levels of Nrf2, NQO1, and γ-GCS ( P < 0.01, P < 0.05 vs. the untreated Ang II group).
Conclusion
Allicin could prevent the development of cardiac remodeling and the progression of cardiac hypertrophy to cardiac dysfunction caused by enhancing the Nrf2 antioxidant signaling pathways.






Similar content being viewed by others
Abbreviations
- ROS:
-
Reactive oxygen species
- Nrf2:
-
Transcription factor nuclear factor (erythroid-derived 2)-like2
- HO-1:
-
Heme Oxygenase 1
- SOD:
-
Superoxide dismutase
- NQO1:
-
NAD(P)H:- quinine oxidoreductase
- GPx:
-
Glutathione peroxidase
- γ-GCS:
-
γ-glutamylcysteine synthetase
- GSH:
-
Glutathione
- T-AOC:
-
Total antioxidant capability
- TBARS:
-
Thiobarbituric acid reactive substances
References
Maulik SK, Kumar S. Oxidative stress and cardiac hypertrophy: a review. Toxicol Mech Methods. 2012;22:359–66.
Delmar M, Makita N. Cardiac connexins, mutations and arrhythmias. Curr Opin Cardiol. 2012;27:236–41.
Takimoto E, Kass DA. Role of oxidative stress in cardiac hypertrophy and remodeling. Hypertension. 2007;49:241–8.
Schluter KD, Wenzel S. Angiotensin II: a hormone involved in and contributing to pro-hypertrophic cardiac networks and target of anti-hypertrophic cross-talks. Pharmacol Ther. 2008;119:311–25.
Das DK, Maulik N, Engelman RM. Redox regulation of angiotensin II signaling in the heart. J Cell Mol Med. 2004;8:144–52.
Miller ER, Pastor-Barriuso R, Dalal D, Riemersma RA, Appel LJ, Guallar E. Meta-analysis: high-dosage vitamin E supplementation may increase all-cause mortality. Ann Intern Med. 2005;142:37–46.
Lonn E, Bosch J, Yusuf S, Sheridan P, Pogue J, Arnold JM, et al. Effects of long-term vitamin E supplementation on cardiovascular events and cancer: a randomized controlled trial. JAMA. 2005;293:1338–47.
Widder JD, Fraccarollo D, Galuppo P, Hansen JM, Jones DP, Ertl G, et al. Attenuation of angiotensin II-induced vascular dysfunction and hypertension by overexpression of Thioredoxin 2. Hypertension. 2009;54:338–44.
Chen SY. Analysis of nrf2-mediated transcriptional induction of antioxidant response in early embryos. Methods Mol Biol. 2012;889:277–90.
Li J, Ichikawa T, Villacorta L, Janicki JS, Brower GL, Yamamoto M, et al. Nrf2 protects against maladaptive cardiac responses to hemodynamic stress. Arterioscler Thromb Vasc Biol. 2009;29:1843–50.
Ichikawa T, Li J, Meyer CJ, Janicki JS, Hannink M, Cui T. Dihydro-CDDO-trifluoroethyl amide (dh404), a novel Nrf2 activator, suppresses oxidative stress in cardiomyocytes. PLoS One. 2009;4:e8391.
Li J, Zhang C, Xing Y, Janicki JS, Yamamoto M, Wang XL, et al. Up-regulation of p27(kip1) contributes to Nrf2-mediated protection against angiotensin II-induced cardiac hypertrophy. Cardiovasc Res. 2011;90:315–24.
Hybertson BM, Gao B, Bose SK, McCord JM. Oxidative stress in health and disease: the therapeutic potential of Nrf2 activation. Mol Aspects Med. 2011;32:234–46.
Alfieri A, Srivastava S, Siow RC, Modo M, Fraser PA, Mann GE. Targeting the Nrf2-Keap1 antioxidant defence pathway for neurovascular protection in stroke. J Physiol. 2011;589:4125–36.
Li XH, Li CY, Lu JM, Tian RB, Wei J. Allicin ameliorates cognitive deficits ageing-induced learning and memory deficits through enhancing of Nrf2 antioxidant signaling pathways. Neurosci Lett. 2012;514:46–50.
Leonardo CC, Dore S. Dietary flavonoids are neuroprotective through Nrf2-coordinated induction of endogenous cytoprotective proteins. Nutr Neurosci. 2011;14:226–36.
Liu C, Cao F, Tang QZ, Yan L, Dong YG, Zhu LH, et al. Allicin protects against cardiac hypertrophy and fibrosis via attenuating reactive oxygen species-dependent signaling pathways. J Nutr Biochem. 2010;21:1238–50.
Gul R, Park JH, Kim SY, Jang KY, Chae JK, Ko JK, et al. Inhibition of ADP-ribosyl cyclase attenuates angiotensin II-induced cardiac hypertrophy. Cardiovasc Res. 2009;81:582–91.
Lu J, Wu DM, Zheng ZH, Zheng YL, Hu B, Zhang ZF. Troxerutin protects against high cholesterol-induced cognitive deficits in mice. Brain. 2011;134(Pt 3):783–97.
Porter KE, Turner NA. Cardiac fibroblasts: at the heart of myocardial remodeling. Pharmacol Ther. 2009;123:255–78.
Li J, Ichikawa T, Jin Y, Hofseth LJ, Nagarkatti P, Nagarkatti M, et al. An essential role of Nrf2 in American ginseng-mediated anti-oxidative actions in cardiomyocytes. J Ethnopharmacol. 2010;130:222–30.
Piao CS, Gao S, Lee GH, Kim DS, Park BH, Chae SW, et al. Sulforaphane protects ischemic injury of hearts through antioxidant pathway and mitochondrial K(ATP) channels. Pharmacol Res. 2010;61:342–8.
Sussan TE, Rangasamy T, Blake DJ, Malhotra D, El-Haddad H, Bedja D, et al. Targeting Nrf2 with the triterpenoid CDDO-imidazolide attenuates cigarette smoke-induced emphysema and cardiac dysfunction in mice. Proc Natl Acad Sci U S A. 2009;106:250–5.
Author information
Authors and Affiliations
Corresponding authors
Additional information
This work was supported by the grants (no. JSU-CX-2010-38) from the college students’ research learning and innovative experiment plan project of JiShou University and Office of Science and Technology of Xiangxi autonomous region.
Rights and permissions
About this article
Cite this article
Li, XH., Li, CY., Xiang, ZG. et al. Allicin Ameliorates Cardiac Hypertrophy and Fibrosis through Enhancing of Nrf2 Antioxidant Signaling Pathways. Cardiovasc Drugs Ther 26, 457–465 (2012). https://doi.org/10.1007/s10557-012-6415-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-012-6415-z