Abstract
A cell undergoes many genetic and epigenetic changes as it transitions to malignancy. Malignant transformation is also accompanied by a progressive loss of tissue homeostasis and perturbations in tissue architecture that ultimately culminates in tumor cell invasion into the parenchyma and metastasis to distant organ sites. Increasingly, cancer biologists have begun to recognize that a critical component of this transformation journey involves marked alterations in the mechanical phenotype of the cell and its surrounding microenvironment. These mechanical differences include modifications in cell and tissue structure, adaptive force-induced changes in the environment, altered processing of micromechanical cues encoded in the extracellular matrix (ECM), and cell-directed remodeling of the extracellular stroma. Here, we review critical steps in this “force journey,” including mechanical contributions to tissue dysplasia, invasion of the ECM, and metastasis. We discuss the biophysical basis of this force journey and present recent advances in the measurement of cellular mechanical properties in vitro and in vivo. We end by describing examples of molecular mechanisms through which tumor cells sense, process and respond to mechanical forces in their environment. While our understanding of the mechanical components of tumor growth, survival and motility remains in its infancy, considerable work has already yielded valuable insight into the molecular basis of force-dependent tumor pathophysiology, which offers new directions in cancer chemotherapeutics.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Cancer biologists have long understood that tumor transformation and metastasis are driven by both intrinsic genomic changes in the constituent tumor cells and the integrated response of the tissue or organ to extrinsic soluble cues, such as growth factors, cytokines, and chemotactic stimuli. Indeed, cancer progression is often collectively conceptualized and portrayed as a “journey” in which a cell morphs over time from a benign phenotype into an invasive or metastatic entity, with many potential intermediate steps along the way. In practice, the stages of this journey are marked by a variety of genetic and histopathological checkpoints, including amplification or inactivation of specific genes, expression of tumor markers, and stereotypic alterations in cell and tissue architecture. Over the past two decades, however, the field has begun to appreciate that an important part of this journey involves changes in the mechanical phenotype of the cell and tissue, as reflected both in intrinsic changes in cell and tissue structure and mechanics and in the biophysical properties of the cell’s microenvironment, such as the mechanics, geometry, and topology of the extracellular matrix (ECM) [1–3]. The interplay between the biophysical properties of the cell and ECM establishes a dynamic, mechanical reciprocity between the cell and the ECM in which the cell’s ability to exert contractile stresses against the extracellular environment balances the elastic resistance of the ECM to that deformation (i.e., ECM rigidity or elasticity). It has now become clear that this force balance can regulate a surprisingly wide range of cellular properties that are all critical to tumorigenesis, including structure, motility, proliferation, and differentiation.
Cells sense, process, and respond to mechanical and other biophysical cues from the ECM using an interconnected hierarchy of mechanochemical systems that includes adhesion receptors (e.g., integrins), intracellular focal adhesions, cytoskeletal networks, and molecular motors. The integrated mechanics and dynamics of these systems enable cells to control their shape, generate force, and ultimately remodel the ECM [4–8]. These structural networks also interact in very specific ways with canonical signal transduction pathways to orchestrate cell behavior. For example, mammary epithelial cells (MECs) form normal acinar structures when cultured in ECMs of physiological stiffness but display the structural and transcriptional hallmarks of a developing tumor when cultured in ECMs of a stiffness that more closely resembles tumor stroma. Processing of these signals requires integrin clustering, ERK activation, cytoskeletal remodeling and Rho GTPase-dependent contractility, illustrating functional connections between growth factor signaling, mechanotransductive signaling, and the cell’s cytoskeletal, adhesive, and contractile machinery [9]. In other words, micromechanical signals from the ECM and cell structural control are intimately connected and interface with signal transduction networks to control fundamental behaviors relevant to tumor transformation, invasion, and metastasis.
In this review, we discuss the evolution of the mechanical phenotype of tumor cells, which we conceptualize as a “force journey.” We begin by discussing the various stages of this journey, including mechanical forces that cells within tissues must encounter and generate while transforming from a normal to an invasive or metastatic phenotype. We then review methods for measuring cellular mechanical properties in vitro and in vivo, including a description of probes of both cortical and intracellular mechanics. Finally, we briefly describe emerging molecular mechanisms for mechanotransduction in tumor cells, with a special emphasis on Rho GTPase and focal adhesion kinase.
2 The mechanical force journey of a tumor cell
2.1 Tissue assembly and morphogenesis
Even in tissues that are seemingly static, cells constantly encounter a variety of mechanical forces and, in turn, actively exert mechanical force on their surroundings (Fig. 1A). These forces can originate from neighboring cells or the ECM and are channeled through specific adhesion receptors [10], as well as through mechanical loads applied nonspecifically to the entire tissue, including interstitial forces and shear flows [11]. Indeed, cells continuously interrogate their mechanical microenvironment and integrate these force cues by exerting a reciprocal compensatory contractile force derived from the coordinated action of cytoskeletal remodeling and motor protein activity. At the tissue and organismal levels, these cell-derived contractile forces are essential for sculpting the organism during embryogenesis and organ development. For example, application of mechanical force to the developing Drosophila embryo induces expression of the mechanosensitive gene Twist throughout the embryo and induces ventralization; moreover, developmental deficits in mutants with abnormal Twist expression may be rescued by application of compressive forces [12]. Force transmission between cells in this system may be quantified and directly manipulated through the use of femtosecond laser ablation [13]. These force interactions play similar roles in vertebrate embryogenesis [14] and the development of specific organ systems; for example, pharmacologic disruption of cellular contractility interferes with lung branching morphogenesis [15]. More recently, the mechanical force environment has been exploited as an engineering tool to direct stem cell differentiation in vitro, with an eye towards tissue engineering and regenerative medicine applications. Human mesenchymal stem cells (hMSCs) cultured on highly compliant and rigid ECMs preferentially differentiate into neurons and osteocytes, respectively. In this case, the ECM directs hMSCs to differentiate towards a tissue type whose stiffness matches that of the ECM [16]. Interestingly, these stiffness-dependent lineage effects depend on the specific ECM protein presented to the cells, with compliant ECMs promoting neurogenesis on collagen-based ECMs and adipogenesis on fibronectin-based ECMs [17]. Even stem cell populations not traditionally regarded as “load-bearing” are sensitive to these mechanical cues; for example, when adult neural stem cells are presented with ECMs of various mechanical rigidities and cultured in mixed differentiation media, soft matrices promote neuronal cultures and rigid matrices promote glial cultures [18].
Alterations in the mechanical interactions between cells and their environment contribute to the tissue dysplasia associated with tumor initiation. For example, transformed epithelial cells express vastly different intermediate filament profiles and cytoskeletal architectures than their normal counterparts; indeed, replacement of a keratin-based cytoskeleton with a vimentin-based cytoskeleton is a defining hallmark of epithelial-to-mesenchymal transition in mammary tissue [19–21]. When presented with compliant substrates that suppress spreading and proliferation of normal cells, transformed cells both proliferate extensively [22] and exert abnormally high tractional forces, which can in turn disrupt cell-cell junctional integrity, compromise tissue polarity, promote anchorage-independent survival and enhance invasion (Fig. 2). This increased contractility reflects increased expression and activity of Rho GTPase and its downstream effectors, as well as high levels of growth factor-induced ERK activity. Most compellingly, manipulation of ECM stiffness and stiffness-dependent cell contractility is sufficient to induce epithelial transformation in cultured cells. For example, as discussed earlier, use of high-stiffness ECM gels alters integrin subtype expression, enhances focal adhesion assembly, disrupts acinar architecture, and promotes invasion in cultured MECs through an elevation of Rho- and ERK-dependent contractility [9]. Intriguingly, similar comparative effects may be induced by culturing transformed MECs on collagen gels affixed to a rigid substrate versus gels allowed to freely float [23], implying that intracellular tension channeled through the ECM is a governing cue that regulates tissue assembly and morphogenesis.
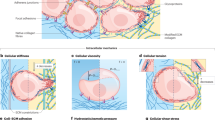
The force journey of a tumor cell. Starting from their participation in normal tissue homeostasis and continuing through all stages of tissue dysplasia, tumor cell invasion, and metasasis, tumor cells both absorb and exert mechanical force. This interplay establishes a dynamic, mechanical reciprocity between tumor cells and their environment (represented schematically as arrows). a Even in normal tissues, such as the epithelium depicted here, cells experience mechanical force from their neighbors and the extracellular matrix, which are often channeled through specific receptor-ligand interactions to trigger signaling events. Cells may also be subject to nonspecific forces applied to the whole tissue, such as interstitial pressure and shear flow. b As a tumor cell detaches from the primary tumor mass and invades the surrounding parenchyma, it continues to exchange mechanical force with its environment, including tractional forces associated with locomotion and protrusive forces of the leading edge of the cell. In some cases, protrusive structures are also used to spatially focus secretion of matrix metalloproteases, e.g., invadopodia. c If a tumor cell escapes its primary tissue and reaches the vasculature, it must withstand shear forces associated with blood flow. Shear has been demonstrated to activate gene programs associated with cytoskeletal remodeling and altered cell-cell adhesion. d In order for a tumor cell to escape the vasculature and metastasize to a distal tissue, it must undergo diapedesis through the endothelial wall, which introduces additional mechanical interactions between the tumor cell and endothelial cells and precedes a transition from cell-cell adhesion to cell-ECM adhesion
Effect of extracellular matrix stiffness on mammary epithelial morphogenesis. Phase and immunofluorescence images of mammary epithelial cells (MECs) cultured on ECM substrates of Young’s moduli of 150 Pa (left column), 1050 Pa (middle column) and >5000 Pa (right column). Cells cultured on substrates with elasticities of 150 Pa, which is similar to the elasticity of normal mammary tissue, form patent acinar structures with clearly defined cell-cell junctions and integrin distributons. A modest increase in ECM elasticity to 1050 Pa leads to loss of luminal patency, disruption of cell-cell contacts, and altered acinar morphology. For ECM elasticities greater than 5000 Pa, acinar organization, cell-cell junctions, and cellular ECM deposition are all completely disrupted. Figure reproduced with permission from [149]
Methods for characterizing the mechanical phenotype. (a-c) Atomic force microscopy (AFM). AFM may be used to obtain topographic images of living cells by scanning a nanoscale probe mounted on a force-sensitive cantilever. This may be accomplished by maintaining the probe in a constant contact or b oscillatory contact with the cell and using the deflections of the cantilever to reconstruct an image. c AFM may also be used to obtain mechanical properties of living cells by indenting the surface of the cell with the probe and recording the resistive force of the cell during indentation. (d-e) Subcellular laser ablation (SLA). In SLA, sub-micron structures inside living cells are irradiated with a high-intensity and tightly-focused laser, resulting in nonlinear absorption and optical breakdown. SLA has been used to d sever and e puncture actomyosin stress fiber bundles in endothelial cells, and the response of these structures (e.g., the retraction of the severed ends) have been used to measure stress fiber mechanical properties and contribution to cell shape. (a)-(c) reproduced from [71] with permission from Blackwell; (d) and (e) reproduced from [80] with permission from Biophysical Society
2.2 Detachment and invasion
As an individual cell frees itself from a tumor and begins to invade the surrounding parenchyma, additional force-generating mechanisms begin to regulate its behavior (Fig. 1B). This phenomenon is perhaps best illustrated by recent work on the role of protrusive processes known as invadopodia in facilitating initial digestion and invasion of the ECM [24, 25]. While the structure and molecular composition of invadopodia remain incompletely understood, from these studies it is now clear that formation of these structures requires highly localized actin polymerization and the coordinated action of multiple actin binding proteins, including cofillin, Arp2/3, and N-WASP [26]. Importantly, although invadopodia share many structural and functional similarities to the filopodia that are observed during two-dimensional migration, they can be distinguished by their ability to spatially focus proteolytic secretion, thereby facilitating the remodeling of existing matrix, secretion of new matrix, and ultimately the establishment of “tracks” that support subsequent invasion. This process was recently captured in real time through elegant multimodal dynamic imaging conducted by Wolf, Friedl, and colleagues [27]. These authors could observe how cells proteolytically degrade and rearrange local ECM fibrils while they migrate through three-dimensional collagen gels. Importantly, broad-spectrum pharmacological inhibition of matrix metalloprotease (MMP) activity forces the cells to “squeeze” through the existing collagen fibers (analogous to amoeboid motion), and this migratory behavior is accompanied by dramatic cell and nuclear deformations. By definition, all of these processes—invadopodia extension, matrix track formation, and cell and nuclear deformation—require local, dramatic, and highly dynamic changes in cytoskeletal organization and cellular mechanics. Quantification of these changes and elucidation of the underlying molecular mechanisms represents a significant and ongoing challenge in this emerging field but also has the potential to uncover novel insights into how cells invade and metastasize, and to identify new therapeutic targets.
The alterations in tumor cell structure and mechanics during detachment and invasion are accompanied by reciprocal changes in ECM topology (organization) and materials properties (mechanics). As described above, cellular contractility can directly promote microscale remodeling of the ECM, which can create matrix bundles or motility tracks that could facilitate three-dimensional cell migration. In addition, tumor formation in vivo is accompanied by a progressive stiffening of the tissue and ECM, as evidenced by the finding that mammary tumor tissue and tumor-adjacent stroma are between 5–20 times stiffer than normal mammary gland, respectively [9]. While such differences in tissue stiffness have not been as well characterized in other tumor systems, they plainly exist and are regularly exploited for cancer diagnosis and therapy. For example, palpable tissue stiffening is routinely used to screen and diagnose virtually all superficial soft-tissue tumors [28]. More recently, ultrasound imaging, which derives its contrast from mechanical compliance differences within tissue, has found a role in tumor diagnosis [29] and intraoperative localization of tumor tissue during resection of gliomas [30, 31]. These increases in ECM stiffness in turn enable cells to generate increased tractional forces on their surroundings, which enhances their growth, survival, and invasion by promoting focal adhesion maturation and signaling through actomyosin contractility [3 9, 32–34]. As described above, the elevated contractility of tumor cells and their associated stromal fibroblasts also induce tension-dependent matrix remodeling to promote the linear reorientation of collagen fibrils surrounding the invasive front of the tumor. Rapidly migrating transformed mammary epithelial cells have been observed on these prominent linear bundles of collagen fibers adjacent to blood vessels [35–37].
2.3 Interstitial forces
In addition to the microscale, molecularly-specific issues described above, an important component of the force journey of a tumor cell involves its ability to withstand nonspecific mechanical forces that arise from the growth of the tumor itself, tissue homeostasis, and transport in the lymphatic system and bloodstream. Even before the initiation of invasion and metastasis, tumor expansion compresses the surrounding ECM, which in turn constricts flow in the vasculature, lymphatic system, and interstitial space. When these compressive stresses occur in the setting of tissue that is highly compliant at baseline, such as pancreas and brain, one observes compromised basement membrane organization and thinning, which combined with the outward projecting compression force, facilitates tumor cell invasion into the parenchyma [9, 38]. These compressive forces clearly also contribute directly to the initial clinical presentation of tumors, such as the symptoms of increased intracranial pressure which commonly prompt presentation in glioblastoma multiforme [39, 40] and the biliary obstructions that are often the initial sign of pancreatic cancer [41]. At the histopathologic level, these stresses can facilitate tumor angiogenesis by enhancing VEGF expression, either through direct upregulation of VEGF secretion or indirectly through induction of hypoxia [42, 43]. Compression forces can also shrink the interstitial space surrounding the ductal structures, which can in turn concentrate growth factors and cytokines to facilitate autocrine and paracrine signaling and promote tumor growth [44]. Tumor-associated changes in interstitial pressure and compressive stress also present significant challenges for drug delivery to solid tumors [45]. These pressures may be compounded by tumor-induced stromal stiffening, which forces the tumor to exert even higher stresses to expand than would be needed in normal tissue. Ironically, while tumor expansion is commonly associated with massive MMP secretion and matrix digestion, tumor-adjacent ECM is frequently quite dense, with increased matrix deposition, crosslinking, and bundling [9].
2.4 Shear Forces
If a tumor cell successfully escapes the confines of its primary tissue of presentation and arrives at the vasculature or lymphatic system en route to metastasis, it must deal with an entirely new set of mechanical forces, in particular those associated with fluid flow and shear (Fig. 1C). Even if the primary tumor is successfully excised, surgical manipulations such as irrigation and suction may subject tumor cells to substantial shear forces or altered patterns of flow [46]. Exposure to shear can activate specific signaling pathways in tumor cells that can in turn induce dramatic reorganization of the cytoskeleton and adhesive machinery and ultimately facilitate reinforcement of cell structure and attachment to the vascular wall [47]. Recently, Basson and colleagues demonstrated that shear can paradoxically enhance adhesion to collagen-based ECM substrates in vitro through a process that involves activation of Src and subsequent assembly of the actin cytoskeleton and formation of focal adhesions [48]. Similarly, Haier and colleagues demonstrated that shear can enhance FAK phosphorylation in colon carcinoma cells, thereby strengthening adhesion to collagen-based ECMs. Impressively, parallel in vivo studies illustrated that overexpression of dominant-negative FAK significantly diminished the ability of tumor cells to adhere to vasculature within the hepatic microcirculation [49].
2.5 Diapedesis and distal metastasis
Once a circulating tumor cell has survived the vasculature and adhered to the endothelium of a target tissue, it must cross the endothelial barrier in order to colonize that tissue (Fig. 1D). Much like leukocytes during an inflammatory response, adherent tumor cells undergo diapedesis, a process by which they extend pseudopodial process that penetrate cell-cell junctions in the endothelium, which requires local and dynamic changes in cellular mechanics driven by cytoskeletal remodeling. This in turn is accompanied by rearrangements in the actin cytoskeletons of the endothelial cells (and changes in their own mechanical phenotype), although the molecular details of this complex process are poorly understood [50]. As the tumor cell makes its way towards the subendothelial basement membrane, these cellular rheological changes are accompanied by changes in expression of adhesion molecules, portending a phenotypic switch from cell-cell adhesion to cell-ECM adhesion. The mechanisms underlying this switch may include conformational activation of existing integrins [51] and expression of entirely new integrin subunit combinations [52]. Recently, Mierke et al. screened 51 tumor lines for their ability to transmigrate in an endothelial co-culture system and showed that the propensity of a cell line to invade correlated with (and was enhanced by) expression of the chemokine receptor CXCR2. Importantly, parallel cellular mechanics measurements revealed that siRNA-mediated knockdown of CXCR2 expression increased cytoskeletal remodeling dynamics and contractility, leading to a model in which CXCR2-mediated signaling promotes tumor cell transmigration through modulation of cytoskeletal assembly and contractility [53].
In summary, tumor cells both withstand and exert mechanical force on their environment in their transformative journey, and these processes require profound and highly dynamic changes in cellular mechanical properties. We now discuss specific biophysical methods that permit direct measurement of these properties.
3 Characterizing the mechanical phenotype
3.1 Stress, strain, elasticity, and viscoelasticity
Before embarking on a detailed discussion of how cellular mechanical properties can be measured, it is first necessary to define some terms. Mechanical stress is the force applied per unit area to an object (e.g., a cell), and strain is that object’s deformation normalized by its initial size. Thus, mechanical stress is expressed in units of force/area (e.g., N/m2 or Pascals (Pa)), and strain is a dimensionless quantity. The Young’s Modulus (also known as the elastic modulus or elasticity (E)), a measure of the deformability of the material, is stress divided by strain; the higher the Young’s Modulus, the stiffer the material. Because strain is a dimensionless quantity, the Young’s Modulus has the same units as stress, e.g., Pa. The Young’s Modulus offers a way to quantify mechanical differences between tissues, and indeed the measured bulk elasticities of human tissues span some five orders of magnitude, e.g., fat (17 Pa), mammary gland (160 Pa), brain (260–490 Pa), liver (640 Pa), kidney (2.5 kPa), skeletal muscle (50 kPa), cartilage (950 kPa) [54]. Strictly speaking, elasticity describes the mechanical properties associated with the ability of a material to internally store mechanical energy and is therefore independent of the rate of deformation. However, many biological materials, including living cells, are capable of both storing and dissipating applied mechanical energy through internal frictional interactions, and do so in a way that depends strongly on the rate of deformation. For this reason, when measuring the mechanical properties of these materials, it is critical to capture both the elastic, or “storage” properties and the viscous, or “loss” properties. Such materials are referred to as viscoelastic materials, and the aggregate viscous and elastic response of a material to mechanical deformation is collectively referred to as its rheology [55].
3.2 Measuring cellular rheology in two-dimensional cell culture
Over the past decade, a sophisticated suite of technologies has been developed with the primary goal of quantifying the viscoelastic properties of cultured cells [8, 56]. These include methods for measuring mean rheological properties of whole cells, such as optical stretching [57–60], micropipette aspiration [61–65], traction force microscopy (TFM) [66–69], atomic force microscopy (AFM) [70–73], and magnetic twisting microrheometry [10, 74–77]; and microscale mechanics of portions of cells, such as subcellular laser ablation (SLA) [78–82], micropost array detectors [83–88], and particle tracking microrheometry [89–93]. Some of these methods can be applied to both the subcellular and whole-cell scale; for example, AFM may be used both at low resolution to obtain mean indentational modulus of a population cells and at high resolution to spatially map mechanical properties across the surface of a single cell. All of these methods have been reviewed extensively elsewhere; to offer examples of how these techniques can be applied to cellular rheology in the context of tumor biology, we focus here on AFM and SLA.
-
1.
Atomic force microscopy (AFM)
In atomic force microscopy (AFM), one measures the interaction force between a sample surface, such as a living cell, and a microscale probe (“tip”) attached to a spring-like cantilever (Fig. 3A-C). The encounter between the tip and sample creates a force that deflects the cantilever, which in turn can be optically tracked and converted to an interaction force if the spring constant of the cantilever is known. Because contrast in AFM originates entirely from the interaction force between the tip and sample, it typically requires no fixation or staining and may readily be conducted in cell culture media. Thus, the method is perfectly suited to capture dynamic processes in living systems. One may acquire two types of information from the tip-sample interaction with the AFM: topographical images and force measurements. In the former measurement, the surface of a sample is scanned at constant force, and the compensatory motions of the stage needed to maintain force constant as the sample topography changes can be used to reconstruct an image. In the latter approach, the sample is vertically indented by the tip at a fixed position, and the resistance of the sample to that deformation may be analyzed to extract the material’s viscoelastic properties. AFM has been employed to image superficial cytoskeletal structures in living cells that may not be readily optically imaged, including cortical actin bundles [94, 95]. Similarly, the force measurement capability of AFM has been used quite successfully to quantitatively measure properties relevant to cellular mechanics at length scales ranging from single molecules to whole cells. In the area of single molecule mechanics, AFM has been used to measure both the force-dependent unfolding of ECM proteins [96] and cell-ECM adhesion proteins [97] in an effort to understand how these systems accomplish mechanochemical conversions. AFM has also demonstrated tremendous value for quantifying the indentational rheology of living cells, including cellular elasticity [98], spatial maps of elasticity across the cell surface [99], and transduction of local compressive forces into biochemical signals [100].
One of the more innovative recent applications of AFM to cellular mechanics is the measurement of protrusive forces generated by growing actin networks, such as those found in invadopodia and pseudopodia. For example, Fletcher and coworkers recently nucleated a dendritic actin network from an AFM cantilever and allowed the network to polymerize against a solid support and deflect the cantilever [101]. With this system, they measured network protrusive forces under various applied loads, analogous to a pseudopodium squeezing its way through an endothelial barrier. Surprisingly, these studies show that the growth velocity depends on the loading history of the network and not merely the instantaneous load. These data therefore suggest that these cytoskeletal networks likely remodel to adapt to applied loads (e.g., by recruiting additional actin filaments), and that these remodeling events are progressively recorded in the evolving structure of the network. These investigators later used a similar approach to measure the oscillatory viscoelastic properties of these growing networks and were able to observe predictable and reversible stress-softening phenomena [102]. These results are particularly exciting in light of the parallel efforts of Radmacher and colleagues to measure forces associated with cell migration in living cells [103]. By orienting the AFM cantilever perpendicularly to a glass coverslip containing a culture of migrating keratocytes, these authors could directly measure cellular propulsive forces as individual cells encountered the cantilever during migration and attempted to push the cantilever by extending a lamellipodium against it.
AFM has also recently been employed as a diagnostic tool for measuring stiffness differences in leukemia cells, and for tracking changes in stiffness in response to chemotherapy [104–106]. In these studies, myeloblastic cell lines were found to be more than an order of magnitude stiffer than corresponding lymphoblastic cell lines. Taken together with the clinical observation that acute myeologenous leukemia produces leukostasis much more frequently than acute lymphocytic leukiemia, these observations serve as a conceptual basis for a model in which low cell deformability likely contributes directly to cellular occlusion of blood vessels. This model has been further supported by the observation that when these cells are treated with chemotherapeutic agents and undergo apoptosis, they stiffen further, consistent with the clinical observation that leukostatic episodes often correlate with the induction of chemotherapy.
-
2.
Subcellular laser ablation
Although AFM has yielded much insight into cellular rheological properties relevant to tumor cell invasion and metastasis, it suffers from two important limitations. First, it can only probe the exterior surface of a living cell, thereby offering limited access to the mechanical properties of internal structures. Second, AFM measurements represent the collective contribution of many cytoskeletal filaments and motor proteins and do not permit dissection of the contribution of individual structural elements in localized microscale regions within the cell. As described earlier, the elucidation of specific cytoskeletal structures in specific places and times in the cell (e.g., stress fibers, filopodial actin bundles) are likely to be critical as the cell journeys towards invasion and metastasis.
Subcellular laser ablation (SLA) has emerged as a complementary method that is capable of overcoming both limitations (Fig. 3D-E). First applied towards cell biology by Michael Berns and coworkers [78, 107–111], SLA uses a tightly focused laser beam to irradiate and vaporize nano- to microscale structures in living cells. Upon irradiation, material at the laser focus undergoes nonlinear multiphoton absorption, leading to optical breakdown and material destruction. Importantly, if the pulse energy, pulse width, and repetition rate are chosen correctly, structures in living cells may be selectively incised with sub-micrometer precision without compromising the plasma membrane or killing the cell. For example, it was recently demonstrated that delivery of femtosecond laser pulses at kilohertz repetition rates and at pulse energies ranging from 1.4 nJ—2.3 nJ can produce zones of photodamage as small as ~150 nm [79].
In the context of understanding biophysical signaling between capillary endothelial cells and the ECM in tumor angiogenesis [112], SLA has been employed to probe the micromechanical properties of actomyosin stress fiber bundles (stress fibers), which are the contractile structures that anchor and enable endothelial cells to exert tractional forces against the ECM [80]. These tractional forces play central roles in endothelial and epithelial cell shape, polarity, and motility both in vitro [113–116] and in vivo [117, 118]. The actin cytoskeletons of living endothelial cells were visualized using yellow fluorescent protein (YFP)-tagged actin, and selected stress fibers at the cell base were irradiated and severed with femtosecond laser pulses. These studies show that severed stress fibers retract in parallel with the axis of the fiber, providing prima facie evidence that these structures bear tensile loads; and that the quantitative retraction kinetics are consistent with that of a viscoelastic cable. Perhaps the most surprising result to emerge from this study is that the coupling between one fiber and the cytoskeletal architecture and shape of the rest of the cell depend strongly on the stiffness of the ECM onto which cells are cultured. For cells cultured on rigid substrates with an elasticity on the order of 1 MPa—1 GPa (e.g., glass), severing a single stress fiber, or even multiple parallel fibers, does not appreciably alter cell shape. Conversely, severing a stress fiber in cells cultured on relatively soft (~4 kPa) polyacrylamide-based substrates produces a 4–5% elongation of the cell along the axis of the stress fiber, as well as a thinning and extension of cytoskeletal structures tens of microns from the site of incision. Parallel studies with TFM revealed that a single stress fiber contributes to ECM strain across nearly the entire cell-ECM interface and strains the ECM most strongly near the points at which the cytoskeletal element inserts into the focal adhesion. Thus, these studies illustrate how SLA can be used to show direct connections between individual micron-scale cellular contractile structures and tractional forces exerted by cells that are distributed over hundreds of square microns.
3.3 Measuring cellular mechanics in three dimensions and in vivo
The application of AFM and SLA to the measurement of cellular mechanics has largely been limited to cells in two-dimensional culture formats. Recently, however, both of these methods have been extended to more physiologically relevant systems. For example, AFM has been used to measure the regional elasticity of cultured brain slices [119] and excised mammary tissue (VMW, unpublished observations). And as described earlier, laser ablation has been used to disrupt mechanical interactions between groups of cells in the developing three-dimensional embryo [13]. Recently, in an effort to understand biophysical mechanisms regulating cadherin-mediated cell-cell adhesion in living epithelia, Cavey and colleagues successfully used SLA to sever junctional actin networks in Drosophila embryonic epithelia in the presence of actin-severing agents and Rho kinase inhibitors, and in the context of siRNA-mediated knockdown of α-catenin [120]. Similar efforts have been used to extend other cellular mechanics methods to living, three-dimensional organisms, including particle-tracking microrheology [121].
Additional new methods are emerging that enable real-time tracking of cell-directed ECM dynamics during various stages of tumorigenesis. In many cases, this has involved creative extensions of two-dimensional mechanics approaches to three-dimensional cultures. For example, three-dimensional particle tracking microrheology has recently been used to quantify both cellular mechanics [122] and matrix remodeling during migration of cells within hydrogels [123]. Similarly, modified versions of TFM have been used to track ECM stresses and strains in three dimensions [124]. These methods have also been correlated with molecular-scale events during cell migration, such as the formation and disassembly of focal adhesions [125] and generation of contractile forces [126]. An important challenge for the future will be to develop mechanical methodologies that are as quantitatively sophisticated as current two-dimensional approaches but that also allow access to more complex and physiologically relevant ECM environments.
4 Future prospects: Towards molecular mechanisms
One of the central challenges in understanding the role of the mechanical phenotype in cancer is elucidation of the molecular mechanisms that enable tumor cells to modulate their mechanical responses and phenotype and their ability to sense and actively direct the biophysical properties of the ECM. This problem is particularly daunting because it requires facility with cell biology, biophysics, materials science, and imaging. It also requires a willingness to integrate new knowledge about mechanics and mechanobiology into our existing understanding of the molecular and cellular biology of cancer. That said, the field has made tremendous strides over the past decade towards identifying key molecules and signaling pathways relevant to cellular mechanobiology in cancer. While a detailed discussion of these mechanisms is beyond the scope of this review, we briefly discuss evidence for two such systems: Rho GTPase and focal adhesion kinase (FAK).
4.1 Rho GTPase
The small GTPase Rho has long been known to contribute to many steps in cancer progression, including proliferation, evasion of apoptosis, invasion, and metastasis [127]. In the specific context of cell mechanics, Rho can stimulate cellular contractility through its ability to activate Rho-associated kinase (ROCK), which in turn inhibits myosin light chain (MLC) phosphatase and activates MLC kinase, thereby promoting net phosphorylation of MLC. As with all of the small GTPases, Rho acts as a molecular switch in which the GTP-bound form is “active” and the GDP-bound form is “inactive.” Indeed, the expression levels and subtype distributions of accessory factors that facilitate this switching, chiefly guanine nucleotide exchange factors (GEFs) and GTPase activating proteins (GAPs), are frequently markedly altered in tumors. Rho activation has been linked in a wide variety of culture systems to actomyosin contractility, formation of stress fiber bundles, and reinforcement and maturation of focal adhesions [128]. In three-dimensional culture models, Rho GTPases play a central role in both pseudopodial protrusion, focal contact and adhesion formation, and trailing-edge retraction, thereby contributing to amoeboid motion [129]. Rho can also regulate and spatially focus secretion of MMPs, which can in turn facilitate matrix remodeling [130]. Recently, ROCK has been explored as a clinical target; for example, the ROCK inhibitor fasudil has been shown to slow the progression of lung and breast tumors in a series of animal models [131].
4.2 Focal adhesion kinase
As discussed earlier, focal adhesions are micron-scale macromolecular complexes at the intracellular face of the cell-ECM interface that serve the dual purpose of physically anchoring cell adhesion receptors to the cytoskeleton and coordinating mechanotransductive signaling. More than one hundred distinct focal adhesion proteins have been identified to date [132], with a rich diversity of functional properties that includes binding to integrins [133], binding to cytoskeletal proteins [134], binding to membrane lipids [135], internal coordination of other focal adhesion proteins [136], and participation in canonical signal transduction pathways [137]. While focal adhesions seem endlessly complex, a few key proteins appear to play a particularly central role in organizing structure and signaling; one such protein is focal adhesion kinase (FAK) [138]. FAK is a nonreceptor tyrosine kinase that is widely overexpressed and activated in tumor cells [139–142]. For this reason, FAK has emerged as an important therapeutic target in cancer; FAK inhibitors have been demonstrated to inhibit the proliferation of tumor cells in culture [143] and are now currently in phase I clinical trials [138]. In addition to its kinase domain, FAK contains a focal adhesion targeting (FAT) domain that is required for its localization to focal adhesions and binds other focal adhesions proteins (e.g., vinculin) and modulators of Rho GTPase signaling (e.g., p190RhoGEF), and a proline-rich domain that enables docking of SH3-containing proteins (e.g., p130Cas) [144]. FAK also contributes indirectly to focal adhesion structure and function by phosphorylating and functionally activating a wide variety of focal adhesion proteins including the F-actin crosslinking protein α-actinin [145]. While the importance of FAK to regulating all steps in the force journey of tumor cells, including tumor de-adhesion, invasion, and distal metastasis, is well documented, the molecular mechanisms through which FAK senses and transduces mechanical signals remains unclear. Evidence for the importance of FAK in mechanosensing comes from a number of sources. For example, the migration and focal adhesion dynamics of FAK -/- fibroblasts are substantially less sensitive to ECM rigidity than wild-type cells [146], and FAK phosphorylation is dramatically stimulated with application of mechanical force [147]. Recently, Mofrad and colleagues used steered molecular dynamics simulations to show that application of tensile forces to the FAT domain of FAK strongly modulates its binding affinity for vinculin [148].
5 Conclusions
One of the most exciting and challenging developments in cancer biology over the past decade is the recognition that tumor growth, invasion, and metastasis are all intricately tied to the constituent cells’ ability to sense, process, and adapt to mechanical forces in their environment. In this review, we have conceptualized this process as a “force journey” through which a cell progresses that includes dramatic changes in tumor cell shape, mechanics, motility, and actuation of mechanical cues in the tumor microenvironment. It is important to emphasize that while this force journey represents a crucial element in the evolution of a tumor, it exists in an equally important context that includes all of the genetic and epigenetic lesions traditionally associated with cancer, such as genomic disruptions and instability, altered sensitivity to soluble growth and inhibitory factors, and secretion of soluble signals that facilitate matrix remodeling and angiogenesis. The challenge is to determine how these two parallel journeys interact, which portions of each are necessary and sufficient for tumor progression, and under what circumstances elements of one can offset or potentiate elements of the other. An important part of interfacing these two paradigms will be to bring together the quantitative power of mechanobiology with the biological sophistication of traditional cancer biology. In particular, progress in this area will require a willingness to broaden the scope of cancer cell biology to include the concepts, methods, and formalisms normally associated with cellular biophysics and engineering that are needed to synthesize and characterize physically-defined microenvironments, precisely measure mechanobiological properties of living cells, and incorporate applied mechanical force into traditional experimental paradigms. This will also require biophysicists and bioengineers to work closely with traditionally-trained cancer biologists to direct their tools towards experimental problems of maximal physiologic relevance and potential clinical impact. While forging these connections is far from trivial, the examples discussed in this review suggest that the benefits to our understanding of the cellular basis of cancer more than justify the effort.
References
Lelievre, S. A., Weaver, V. M., Nickerson, J. A., Larabell, C. A., Bhaumik, A., Petersen, O. W., et al. (1998). Tissue phenotype depends on reciprocal interactions between the extracellular matrix and the structural organization of the nucleus. Proceedings of the National Academy of Sciences of the United States of America, 95(25), 14711–14716.
Nelson, C. A., & Bissell, M. J. (2005). Modeling dynamic reciprocity: Engineering three-dimensional culture models of breast architecture, function, and neoplastic transformation. Seminars in Cancer Biology, 15(5), 342–352.
Paszek, M. J., & Weaver, V. M. (2004). The tension mounts: Mechanics meets morphogenesis and malignancy. Journal of Mammary Gland Biology and Neoplasia, 9(4), 325–342.
Bershadsky, A. D., Balaban, N. Q., & Geiger, B. (2003). Adhesion-dependent cell mechanosensitivity. Annual Review of Cell and Developmental Biology, 19, 677–695.
Giancotti, F. G., & Ruoslahti, E. (1999). Transduction — Integrin signaling. Science, 285(5430), 1028–1032.
Ingber, D. E. (2008). Tensegrity-based mechanosensing from macro to micro. Progress in Biophysics & Molecular Biology, 97(2–3), 163–179.
Janmey, P. A. (1998). The cytoskeleton and cell signaling: Component localization and mechanical coupling. Physiological Reviews, 78(3), 763–781.
Lele, T. P., & Kumar, S. (2007). Brushes, cables, and anchors: Recent insights into multiscale assembly and mechanics of cellular structural networks. Cell Biochemistry and Biophysics, 47(3), 348–360.
Paszek, M. J., Zahir, N., Johnson, K. R., Lakins, J. N., Rozenberg, G. I., Gefen, A., et al. (2005). Tensional homeostasis and the malignant phenotype. Cancer Cell, 8, 241–254.
Wang, N., Butler, J. P., & Ingber, D. E. (1993). Mechanotransduction across the cell surface and through the cytoskeleton. Science, 260(5111), 1124–1127.
Ingber, D. E. (2003). Mechanobiology and diseases of mechanotransduction. Annals of Medicine, 35(8), 564–577.
Farge, E. (2003). Mechanical induction of Twist in the Drosophila foregut/stomodeal primordium. Current Biology, 13(16), 1365–1377.
Supatto, W., Debarre, D., Moulia, B., Brouzes, E., Martin, J. L., Farge, E., et al. (2005). In vivo modulation of morphogenetic movements in Drosophila embryos with femtosecond laser pulses. Proceedings of the National Academy of Sciences of the United States of America, 102(4), 1047–1052.
Wei, L., Roberts, W., Wang, L., Yamada, M., Zhang, S., Zhao, Z., et al. (2001). Rho kinases play an obligatory role in vertebrate embryonic organogenesis. Development, 128(15), 2953–2962.
Ewald, A. J., Brenot, A., Duong, M., Chan, B. S., & Werb, Z. (2008). Collective epithelial migration and cell rearrangements drive mammary branching morphogenesis. Developmental Cell, 14(4), 570–581.
Engler, A. J., Sen, S., Sweeney, H. L., & Discher, D. E. (2006). Matrix elasticity directs stem cell lineage specification. Cell, 126(4), 677–689.
Winer, J. P., Janmey, P. A., McCormick, M. E., & Funaki, M. (2009). Bone Marrow-Derived Human Mesenchymal Stem Cells Become Quiescent on Soft Substrates but Remain Responsive to Chemical or Mechanical Stimuli. Tissue Engineering Part A, 15(1), 147–154.
Saha, K., Keung, A. J., Irwin, E. F., Li, Y., Little, L., Schaffer, D. V., et al. (2008). Substrate modulus directs neural stem cell behavior. Biophysical Journal, 95(9), 4426–4438.
Kokkinos, M. I., Wafai, R., Wong, M. K., Newgreen, D. F., Thompson, E. W., & Waltham, M. (2007). Vimentin and epithelial-mesenchymal transition in human breast cancer—Observations in vitro and in vivo. Cells Tissues Organs, 185(1–3), 191–203.
Pagan, R., Martin, I., Alonso, A., Llobera, M., & Vilaro, S. (1996). Vimentin filaments follow the preexisting cytokeratin network during epithelial-mesenchymal transition of cultured neonatal rat hepatocytes. Experimental Cell Research, 222(2), 333–344.
Willipinski-Stapelfeldt, B., Riethdorf, S., Assmann, V., Woelfle, U., Rau, T., Sauter, G., et al. (2005). Changes in cytoskeletal protein composition indicative of an epithelial-mesenchymal transition in human micrometastatic and primary breast carcinoma cells. Clinical Cancer Research, 11(22), 8006–8014.
Wang, H. B., Dembo, M., & Wang, Y. L. (2000). Substrate flexibility regulates growth and apoptosis of normal but not transformed cells. American Journal of Physiology — Cell Physiology, 279(5), C1345–1350.
Wozniak, M. A., Desai, R., Solski, P. A., Der, C. J., & Keely, P. J. (2003). ROCK-generated contractility regulates breast epithelial cell differentiation in response to the physical properties of a three-dimensional collagen matrix. Journal of Cell Biology, 163(3), 583–595.
Wolf, K., Friedl, P. (2008). Mapping proteolytic cancer cell-extracellular matrix interfaces. Clinical and Expermiental Metastasis, (in press).
Yamaguchi, H., Wyckoff, J., & Condeelis, J. (2005). Cell migration in tumors. Current Opinion in Cell Biology, 17(5), 559–564.
Yamaguchi, H., Lorenz, M., Kempiak, S., Sarmiento, C., Coniglio, S., Symons, M., et al. (2005). Molecular mechanisms of invadopodium formation: the role of the N-WASP-Arp2/3 complex pathway and cofilin. Journal of Cell Biology, 168(3), 441–452.
Wolf, K., Wu, Y. I., Liu, Y., Geiger, J., Tam, E., Overall, C., et al. (2007). Multi-step pericellular proteolysis controls the transition from individual to collective cancer cell invasion. Nature Cell Biology, 9(8), 893–904.
Khaled, W., Reichling, S., Bruhns, O. T., Boese, H., Baumann, M., Monkman, G., et al. (2004). Palpation imaging using a haptic system for virtual reality applications in medicine. Studies in Health Technology and Informatics, 98, 147–153.
Khaled, W., Reichling, S., Bruhns, O. T., & Ermert, H. (2006). Ultrasonic strain imaging and reconstructive elastography for biological tissue. Ultrasonics, 44(Suppl 1), e199–202.
Selbekk, T., Bang, J., & Unsgaard, G. (2005). Strain processing of intraoperative ultrasound images of brain tumours: initial results. Ultrasound in Medicine and Biology, 31(1), 45–51.
Unsgaard, G., Rygh, O. M., Selbekk, T., Muller, T. B., Kolstad, F., Lindseth, F., et al. (2006). Intra-operative 3D ultrasound in neurosurgery. Acta Neurochir (Wien), 148(3), 235–253 discussion 253.
Croft, D. R., Sahai, E., Mavria, G., Li, S., Tsai, J., Lee, W. M., et al. (2004). Conditional ROCK activation in vivo induces tumor cell dissemination and angiogenesis. Cancer Research, 64(24), 8994–9001.
O’Brien, L. E., Jou, T. S., Pollack, A. L., Zhang, Q., Hansen, S. H., Yurchenco, P., et al. (2001). Rac1 orientates epithelial apical polarity through effects on basolateral laminin assembly. Nature Cell Biology, 3(9), 831–838.
Wang, F., Weaver, V. M., Petersen, O. W., Larabell, C. A., Dedhar, S., Briand, P., et al. (1998). Reciprocal interactions between beta1-integrin and epidermal growth factor receptor in three-dimensional basement membrane breast cultures: a different perspective in epithelial biology. Proceedings of the National Academy of Sciences of the United States of America, 95(25), 14821–14826.
Ingman, W. V., Wyckoff, J., Gouon-Evans, V., Condeelis, J., & Pollard, J. W. (2006). Macrophages promote collagen fibrillogenesis around terminal end buds of the developing mammary gland. Developmental Dynamics, 235(12), 3222–3229.
Provenzano, P. P., Inman, D. R., Eliceiri, K. W., Knittel, J. G., Yan, L., Rueden, C. T., et al. (2008). Collagen density promotes mammary tumor initiation and progression. BMC Medicine, 6(1), 1–11.
Wyckoff, J. B., Pinner, S. E., Gschmeissner, S., Condeelis, J. S., & Sahai, E. (2006). ROCK- and myosin-dependent matrix deformation enables protease-independent tumor-cell invasion in vivo. Current Biology, 16(15), 1515–1523.
Ingber, D. E., Madri, J. A., & Jamieson, J. D. (1981). Role of basal lamina in neoplastic disorganization of tissue architecture. Proceedings of the National Academy of Sciences of the United States of America, 78(6), 3901–3905.
Black, P. (1998). Management of malignant glioma: role of surgery in relation to multimodality therapy. Journal of Neurovirology, 4(2), 227–236.
Schankin, C. J., Ferrari, U., Reinisch, V. M., Birnbaum, T., Goldbrunner, R., & Straube, A. (2007). Characteristics of brain tumour-associated headache. Cephalalgia, 27(8), 904–911.
Watanapa, P., & Williamson, R. C. N. (1992). Surgical Palliation for Pancreatic-Cancer—Developments during the Past 2 Decades. British Journal of Surgery, 79(1), 8–20.
Harris, A. L. (2002). Hypoxia—a key regulatory factor in tumour growth. Nature Reviews Cancer, 2(1), 38–47.
Roose, T., Netti, P. A., Munn, L. L., Boucher, Y., & Jain, R. K. (2003). Solid stress generated by spheroid growth estimated using a linear poroelasticity model small star, filled. Microvascular Research, 66(3), 204–212.
Tschumperlin, D. J., Dai, G., Maly, I. V., Kikuchi, T., Laiho, L. H., McVittie, A. K., et al. (2004). Mechanotransduction through growth-factor shedding into the extracellular space. Nature, 429(6987), 83–86.
Minchinton, A. I., & Tannock, I. F. (2006). Drug penetration in solid tumours. Nature Reviews Cancer, 6(8), 583–592.
Marshburn, P. B., & Hulka, J. F. (1990). A simple irrigator-aspirator cannula for laparoscopy: the Stewart system. Obstetrics and Gynecology, 75(3 Pt 1), 458–460.
Davies, P. F., Spaan, J. A., & Krams, R. (2005). Shear stress biology of the endothelium. Annals of Biomedical Engineering, 33(12), 1714–1718.
Thamilselvan, V., Craig, D. H., & Basson, M. D. (2007). FAK association with multiple signal proteins mediates pressure-induced colon cancer cell adhesion via a Src-dependent PI3K/Akt pathway. FASEB Journal, 21(8), 1730–1741.
von Sengbusch, A., Gassmann, P., Fisch, K. M., Enns, A., Nicolson, G. L., & Haier, J. (2005). Focal adhesion kinase regulates metastatic adhesion of carcinoma cells within liver sinusoids. American Journal of Pathology, 166(2), 585–596.
Miles, F. L., Pruitt, F. L., van Golen, K. L., & Cooper, C. R. (2008). Stepping out of the flow: capillary extravasation in cancer metastasis. Clinical and Expermiental Metastasis, 25(4), 305–324.
Stewart, D. A., Cooper, C. R., & Sikes, R. A. (2004). Changes in extracellular matrix (ECM) and ECM-associated proteins in the metastatic progression of prostate cancer. Reproductive Biology and Endocrinology, 2, 2.
Heino, J., & Massague, J. (1989). Transforming growth factor-beta switches the pattern of integrins expressed in MG-63 human osteosarcoma cells and causes a selective loss of cell adhesion to laminin. Journal of Biological Chemistry, 264(36), 21806–21811.
Mierke, C. T., Zitterbart, D. P., Kollmannsberger, P., Raupach, C., Schlotzer-Schrehardt, U., Goecke, T. W., et al. (2008). Breakdown of the endothelial barrier function in tumor cell transmigration. Biophysical Journal, 94(7), 2832–2846.
Levental, I., Georges, P. C., & Janmey, P. A. (2007). Soft biological materials and their impact on cell function. Soft Matter, 3(3), 299–306.
Janmey, P. A., Georges, P. C., & Hvidt, S. (2007). Basic rheology for biologists. Methods in Cell Biology, 83(1), 3–27.
Chown, M. G., & Kumar, S. (2007). Imaging and manipulating the structural machinery of living cells on the micro- and nanoscale. International Journal of Nanomedicine, 2(3), 333–344.
Dao, M., Lim, C. T., & Suresh, S. (2003). Mechanics of the human red blood cell deformed by optical tweezers. Journal of the Mechanics and Physics of Solids, 51(11–12), 2259–2280.
Guck, J., Schinkinger, S., Lincoln, B., Wottawah, F., Ebert, S., Romeyke, M., et al. (2005). Optical deformability as an inherent cell marker for testing malignant transformation and metastatic competence. Biophysical Journal, 88(5), 3689–3698.
Lincoln, B., Wottawah, F., Schinkinger, S., Ebert, S., & Guck, J. (2007). High-throughput rheological measurements with an optical stretcher. Methods in Cell Biology, 83(1), 397–423.
Trepat, X., Grabulosa, M., Puig, F., Maksym, G. N., Navajas, D., & Farre, R. (2004). Viscoelasticity of human alveolar epithelial cells subjected to stretch. American Journal of Physiology-Lung Cellular and Molecular Physiology, 287(5), L1025–L1034.
Drury, J. L., & Dembo, M. (2001). Aspiration of human neutrophils: Effects of shear thinning and cortical dissipation. Biophysical Journal, 81(6), 3166–3177.
Hochmuth, R. M. (2000). Micropipette aspiration of living cells. Journal of Biomechanics, 33(1), 15–22.
Jones, W. R., Ting-Beall, H. P., Lee, G. M., Kelley, S. S., Hochmuth, R. M., & Guilak, F. (1999). Alterations in the Young’s modulus and volumetric properties of chondrocytes isolated from normal and osteoarthritic human cartilage. Journal of Biomechanics, 32(2), 119–127.
Koay, E. J., Shieh, A. C., & Athanasiou, K. A. (2003). Creep indentation of single cells. Journal of Biomechanical Engineering-Transactions of the ASME, 125(3), 334–341.
Trickey, W. R., Baaijens, F. P. T., Laursen, T. A., Alexopoulos, L. G., & Guilak, F. (2006). Determination of the Poisson’s ratio of the cell: recovery properties of chondrocytes after release from complete micropipette aspiration. Journal of Biomechanics, 39(1), 78–87.
Beningo, K. A., Dembo, M., Kaverina, I., Small, J. V., & Wang, Y. L. (2001). Nascent focal adhesions are responsible for the generation of strong propulsive forces in migrating fibroblasts. Journal of Cell Biology, 153(4), 881–887.
Butler, J. P., Tolic-Norrelykke, I. M., Fabry, B., & Fredberg, J. J. (2002). Traction fields, moments, and strain energy that cells exert on their surroundings. American Journal of Physiology-Cell Physiology, 282(3), C595–C605.
Munevar, S., Wang, Y. L., & Dembo, M. (2001). Traction force microscopy of migrating normal and H-ras transformed 3T3 fibroblasts. Biophysical Journal, 80(4), 1744–1757.
Pelham, R. J., & Wang, Y. L. (1999). High resolution detection of mechanical forces exerted by locomoting fibroblasts on the substrate. Molecular Biology of the Cell, 10(4), 935–945.
Hansma, H. G., & Hoh, J. H. (1994). Biomolecular Imaging with the Atomic-Force Microscope. Annual Review of Biophysics and Biomolecular Structure, 23(1), 115–139.
Kumar, S., & Hoh, J. H. (2001). Probing the machinery of intracellular trafficking with the atomic force microscope. Traffic, 2(11), 746–756.
Lal, R., & John, S. A. (1994). Biological Applications of Atomic-Force Microscopy. American Journal of Physiology, 266(1), C1–&.
Radmacher, M. (2002). Measuring the elastic properties of living cells by the atomic force microscope. Methods in Cell Biology, 68(1), 67–90.
An, S. S., Laudadio, R. E., Lai, J., Rogers, R. A., & Fredberg, J. J. (2002). Stiffness changes in cultured airway smooth muscle cells. American Journal of Physiology-Cell Physiology, 283(3), C792–C801.
Ingber, D. E., Prusty, D., Sun, Z. Q., Betensky, H., & Wang, N. (1995). Cell shape, cytoskeletal mechanics, and cell cycle control in angiogenesis. Journal of Biomechanics, 28(12), 1471–1484.
Maksym, G. N., Fabry, B., Butler, J. P., Navajas, D., Tschumperlin, D. J., Laporte, J. D., et al. (2000). Mechanical properties of cultured human airway smooth muscle cells from 0.05 to 0.4 Hz. Journal of Applied Physiology, 89(4), 1619–1632.
Wang, N., Tolic-Norrelykke, I. M., Chen, J. X., Mijailovich, S. M., Butler, J. P., Fredberg, J. J., et al. (2002). Cell prestress. I. Stiffness and prestress are closely associated in adherent contractile cells. American Journal of Physiology-Cell Physiology, 282(3), C606–C616.
Berns, M. W., Aist, J., Edwards, J., Strahs, K., Girton, J., McNeill, P., et al. (1981). Laser microsurgery in cell and developmental biology. Science, 213(4507), 505–13.
Heisterkamp, A., Maxwell, I. Z., Mazur, E., Underwood, J. M., Nickerson, J. A., Kumar, S., et al. (2005). Pulse energy dependence of subcellular dissection by femtosecond laser pulses. Optics Express, 13(10), 3690–3696.
Kumar, S., Maxwell, I. Z., Heisterkamp, A., Polte, T. R., Lele, T. P., Salanga, M., et al. (2006). Viscoelastic retraction of single living stress fibers and its impact on cell shape, cytoskeletal organization, and extracellular matrix mechanics. Biophysical Journal, 90(10), 3762–3773.
Lele, T. P., Pendse, J., Kumar, S., Salanga, M., Karavitis, J., & Ingber, D. E. (2006). Mechanical forces alter zyxin unbinding kinetics within focal adhesions of living cells. Journal of Cellular Physiology, 207(1), 187–194.
Shen, N., Colvin, M., Genin, F., Huser, T., Cortopassi, G. A., Stearns, T., et al. (2004). Using femtosecond laser subcellular surgery to studycell biology. Biophysical Journal, 86(1), 520A–520A.
du Roure, O., Saez, A., Buguin, A., Austin, R. H., Chavrier, P., Siberzan, P., et al. (2005). Force mapping in epithelial cell migration. Proceedings of the National Academy of Sciences of the United States of America, 102(7), 2390–2395.
Nelson, C. M., Jean, R. P., Tan, J. L., Liu, W. F., Sniadecki, N. J., Spector, A. A., et al. (2005). Emergent patterns of growth controlled by multicellular form and mechanics. Proceedings of the National Academy of Sciences of the United States of America, 102(33), 11594–11599.
Poujade, M., Grasland-Mongrain, E., Hertzog, A., Jouanneau, J., Chavrier, P., Ladoux, B., et al. (2007). Collective migration of an epithelial monolayer in response to a model wound. Proceedings of the National Academy of Sciences of the United States of America, 104(41), 15988–15993.
Rabodzey, A., Alcaide, P., Luscinskas, F. W., & Ladoux, B. (2008). Mechanical forces induced by the transendothelial migration of human neutrophils. Biophysical Journal, 95(3), 1428–1438.
Sniadecki, N. J., Anguelouch, A., Yang, M. T., Lamb, C. M., Liu, Z., Kirschner, S. B., et al. (2007). Magnetic microposts as an approach to apply forces to living cells. Proceedings of the National Academy of Sciences of the United States of America, 104(37), 14553–14558.
Tan, J. L., Tien, J., Pirone, D. M., Gray, D. S., Bhadriraju, K., & Chen, C. S. (2003). Cells lying on a bed of microneedles: An approach to isolate mechanical force. Proceedings of the National Academy of Sciences of the United States of America, 100(4), 1484–1489.
Bausch, A. R., Ziemann, F., Boulbitch, A. A., Jacobson, K., & Sackmann, E. (1998). Local measurements of viscoelastic parameters of adherent cell surfaces by magnetic bead microrheometry. Biophysical Journal, 75(4), 2038–2049.
Tseng, Y., Kole, T. P., & Wirtz, D. (2002). Micromechanical mapping of live cells by multiple-particle-tracking microrheology. Biophysical Journal, 83(6), 3162–3176.
Valentine, M. T., Perlman, Z. E., Gardel, M. L., Shin, J. H., Matsudaira, P., Mitchison, T. J., et al. (2004). Colloid surface chemistry critically affects multiple particle tracking measurements of biomaterials. Biophysical Journal, 86(6), 4004–4014.
Yamada, S., Wirtz, D., & Kuo, S. C. (2000). Mechanics of living cells measured by laser tracking microrheology. Biophysical Journal, 78(4), 1736–1747.
Yap, B., & Kamm, R. D. (2005). Mechanical deformation of neutrophils into narrow channels induces pseudopod projection and changes in biomechanical properties. Journal of Applied Physiology, 98(5), 1930–1939.
Pesen, D., & Hoh, J. H. (2005). Modes of remodeling in the cortical cytoskeleton of vascular endothelial cells. FEBS Letters, 579(2), 473–476.
Pesen, D., & Hoh, J. H. (2005). Micromechanical architecture of the endothelial cell cortex. Biophysical Journal, 88(1), 670–679.
Oberhauser, A. F., Badilla-Fernandez, C., Carrion-Vasquez, M., & Fernandez, J. M. (2002). The mechanical hierarchies of fibronectin observed with single-molecule AFM. Journal of Molecular Biology, 319, 433–447.
Carl, P., Kwok, C. H., Manderson, G., Speicher, D. W., & Discher, D. E. (2001). Forced unfolding modulated by disulfide bonds in the Ig domains of a cell adhesion molecule. Proceedings of the National Academy of Sciences of the United States of America, 98, 1565–1570.
Rotsch, C., Jacobson, K., & Radmacher, M. (1999). Dimensional and mechanical dynamics of active and stable edges in motile fibroblasts investigated by using atomic force microscopy. Proceedings of the National Academy of Sciences of the United States of America, 96(3), 921–926.
A-Hassan, E., Heinz, W. F., Antonik, M. D., D’Costa, N. P., Nageswaran, S., Schoenenberger, C. A., et al. (1998). Relative microelastic mapping of living cells by atomic force microscopy. Biophysical Journal, 74(3), 1564–1578.
Charras, G. T., & Horton, M. A. (2002). Single cell mechanotransduction and its modulation analyzed by atomic force microscope indentation. Biophysical Journal, 82(6), 2970–2981.
Parekh, S. H., Chaudhuri, O., Theriot, J. A., & Fletcher, D. A. (2005). Loading history determines the velocity of actin-network growth. Nature Cell Biology, 7(12), 1219–1223.
Chaudhuri, O., Parekh, S. H., & Fletcher, D. A. (2007). Reversible stress softening of actin networks. Nature, 445(7125), 295–298.
Prass, M., Jacobson, K., Mogilner, A., & Radmacher, M. (2006). Direct measurement of the lamellipodial protrusive force in a migrating cell. Journal of Cell Biology, 174(6), 767–772.
Lam, W. A., Rosenbluth, M. J., & Fletcher, D. A. (2008). Increased leukaemia cell stiffness is associated with symptoms of leucostasis in paediatric acute lymphoblastic leukaemia. British Journal of Haematology, 142(3), 497–501.
Lam, W. A., Rosenbluth, M. J., & Fletcher, D. A. (2007). Chemotherapy exposure increases leukemia cell stiffness. Blood, 109(8), 3505–3508.
Rosenbluth, M. J., Lam, W. A., & Fletcher, D. A. (2006). Force microscopy of nonadherent cells: A comparison of leukemia cell deformability. Biophysical Journal, 90(8), 2994–3003.
Strahs, K. R., Burt, J. M., & Berns, M. W. (1978). Contractility changes in cultured cardiac cells following laser microirradiation of myofibrils and the cell surface. Experimental Cell Research, 113(1), 75–83.
Strahs, K. R., & Berns, M. W. (1979). Laser microirradiation of stress fibers and intermediate filaments in non-muscle cells from cultured rat heart. Experimental Cell Research, 119(1), 31–45.
Koonce, M. P., Strahs, K. R., & Berns, M. W. (1982). Repair of laser-severed stress fibers in myocardial non-muscle cells. Experimental Cell Research, 141(2), 375–384.
Burt, J. M., Strahs, K. R., & Berns, M. W. (1979). Correlation of cell surface alterations with contractile response in laser microbeam irradiated myocardial cells. A scanning electron microscope study. Experimental Cell Research, 118(2), 341–351.
Botvinick, E. L., Venugopalan, V., Shah, J. V., Liaw, L. H., & Berns, M. W. (2004). Controlled ablation of microtubules using a picosecond laser. Biophysical Journal, 87(6), 4203–4212.
Ingber, D. E. (2002). Mechanical signalling and the cellular response to extracellular matrix in angiogenesis and cardiovascular physiology. Circulation Research, 91(10), 877–887.
Assoian, R. K., & Klein, E. A. (2008). Growth control by intracellular tension and extracellular stiffness. Trends in Cell Biology, 18(7), 347–352.
Burridge, K., & ChrzanowskaWodnicka, M. (1996). Focal adhesions, contractility, and signaling. Annual Review of Cell and Developmental Biology, 12, 463–518.
Etienne-Manneville, S., & Hall, A. (2002). Rho GTPases in cell biology. Nature, 420(6916), 629–635.
Hotulainen, P., & Lappalainen, P. (2006). Stress fibers are generated by two distinct actin assembly mechanisms in motile cells. Journal of Cell Biology, 173(3), 383–394.
Sipkema, P., van der Linden, P. J. W., Westerhof, N., & Yin, F. C. P. (2003). Effect of cyclic axial stretch of rat arteries on endothelial cytoskeletal morphology and vascular reactivity. Journal of Biomechanics, 36(5), 653–659.
Hayes, A. J., Benjamin, M., & Ralphs, J. R. (1999). Role of actin stress fibres in the development of the intervertebral disc: Cytoskeletal control of extracellular matrix assembly. Developmental Dynamics, 215(3), 179–189.
Elkin, B. S., Azeloglu, E. U., Costa, K. D., & Morrison, B. (2007). Mechanical heterogeneity of the rat hippocampus measured by atomic force microscope indentation. Journal of Neurotrauma, 24(5), 812–822.
Cavey, M., Rauzi, M., Lenne, P. F., & Lecuit, T. (2008). A two-tiered mechanism for stabilization and immobilization of E-cadherin. Nature, 453(7196), 751–U2.
Daniels, B. R., Masi, B. C., & Wirtz, D. (2006). Probing single-cell micromechanics in vivo: The microrheology of C-elegans developing embryos. Biophysical Journal, 90(12), 4712–4719.
Panorchan, P., Lee, J. S. H., Kole, T. P., Tseng, Y., & Wirtz, D. (2006). Microrheology and ROCK signaling of human endothelial cells embedded in a 3D matrix. Biophysical Journal, 91(9), 3499–3507.
Bloom, R. J., George, J. P., Celedon, A., Sun, S. X., & Wirtz, D. (2008). Mapping local matrix remodeling induced by a migrating tumor cell using 3-D multiple-particle tracking. Biophysical Journal, 95(8), 4077–4088.
Roy, P., Petroll, W. M., Cavanagh, H. D., & Jester, J. V. (1999). Exertion of tractional force requires the coordinated up-regulation of cell contractility and adhesion. Cell Motility and the Cytoskeleton, 43(1), 23–34.
Petroll, W. M., Ma, L., & Jester, J. V. (2003). Direct correlation of collagen matrix deformation with focal adhesion dynamics in living corneal fibroblasts. Journal of Cell Science, 116(8), 1481–1491.
Vanni, S., Lagerholm, B. C., Otey, C., Taylor, D. L., & Lanni, F. (2003). Internet-Based image analysis quantifies contractile behavior of individual fibroblasts inside model tissue. Biophysical Journal, 84(4), 2715–2727.
Vega, F. M., & Ridley, A. J. (2008). Rho GTPases in cancer cell biology. FEBS Letters, 582(14), 2093–2101.
Raftopoulou, M., & Hall, A. (2004). Cell migration: Rho GTPases lead the way. Developmental Biology, 265(1), 23–32.
Friedl, P., & Wolf, K. (2003). Tumour-cell invasion and migration: Diversity and escape mechanisms. Nature Reviews Cancer, 3(5), 362–374.
Lozano, E., Betson, M., & Braga, V. M. M. (2003). Tumor progression: small GTPases and loss of cell-cell adhesion. Bioessays, 25(5), 452–463.
Ying, H., Biroc, S. L., Li, W. W., Alicke, B., Xuan, J. A., Pagila, R., et al. (2006). The Rho kinase inhibitor fasudil inhibits tumor progression in human and rat tumor models. Molecular Cancer Therapeutics, 5(9), 2158–2164.
Bershadsky, A., Kozlov, M., & Geiger, B. (2006). Adhesion-mediated mechanosensitivity: a time to experiment, and a time to theorize. Current Opinion in Cell Biology, 18(5), 472–481.
Schlaepfer, D. D., Hanks, S. K., Hunter, T., & Vandergeer, P. (1994). Integrin-Mediated Signal-Transduction Linked to Ras Pathway by Grb2 Binding to Focal Adhesion Kinase. Nature, 372(6508), 786–791.
Ridley, A. J., & Hall, A. (1992). The Small Gtp-Binding Protein Rho Regulates the Assembly of Focal Adhesions and Actin Stress Fibers in Response to Growth-Factors. Cell, 70(3), 389–399.
Sechi, A. S., & Wehland, J. (2000). The actin cytoskeleton and plasma membrane connection: PtdIns(4,5)P-2 influences cytoskeletal protein activity at the plasma membrane. Journal of Cell Science, 113(21), 3685–3695.
Gilmore, A. P., & Burridge, K. (1996). Regulation of vinculin binding to talin and actin by phosphatidyl-inositol-4-5-bisphosphate. Nature, 381(6582), 531–535.
Schwartz, M. A., Schaller, M. D., & Ginsberg, M. H. (1995). Integrins: Emerging paradigms of signal transduction. Annual Review of Cell and Developmental Biology, 11, 549–599.
Tilghman, R. W., & Parsons, J. T. (2008). Focal adhesion kinase as a regulator of cell tension in the progression of cancer. Seminars in Cancer Biology, 18(1), 45–52.
Gabarra-Niecko, V., Schaller, M. D., & Dunty, J. M. (2003). FAK regulates biological processes important for the pathogenesis of cancer. Cancer and Metastasis Reviews, 22(4), 359–374.
Mitra, S. K., Hanson, D. A., & Schlaepfer, D. D. (2005). Focal adhesion kinase: In command and control of cell motility. Nature Reviews Molecular Cell Biology, 6(1), 56–68.
Parsons, J. T. (2003). Focal adhesion kinase: the first ten years. Journal of Cell Science, 116(8), 1409–1416.
Schlaepfer, D. D., Mitra, S. K., & Ilic, D. (2004). Control of motile and invasive cell phenotypes by focal adhesion kinase. Biochimica et Biophysica Acta-Molecular Cell Research, 1692(2–3), 77–102.
Liu, T. J., LaFortune, T., Honda, T., Ohmori, O., Hatakeyama, S., Meyer, T., et al. (2007). Inhibition of both focal adhesion kinase and insulin-like growth factor-I receptor kinase suppresses glioma proliferation in vitro and in vivo. Molecular Cancer Therapeutics, 6(4), 1357–1367.
Shen, T. L., & Guan, J. L. (2001). Differential regulation of cell migration and cell cycle progression by FAK complexes with Src, PI3K, Grb7 and Grb2 in focal contacts. FEBS Letters, 499(1–2), 176–181.
Izaguirre, G., Aguirre, L., Hu, Y. P., Lee, H. Y., Schlaepfer, D. D., Aneskievich, B. J., et al. (2001). The cytoskeletal/non-muscle isoform of alpha-actinin is phosphorylated on its actin-binding domain by the focal adhesion kinase. Journal of Biological Chemistry, 276(31), 28676–28685.
Wang, H. B., Dembo, M., Hanks, S. K., & Wang, Y. L. (2001). Focal adhesion kinase is involved in mechanosensing during fibroblast migration. Proceedings of the National Academy of Sciences of the United States of America, 98(20), 11295–11300.
Hamasaki, K., Mimura, T., Furuya, H., Morino, N., Yamazaki, T., Komuro, I., et al. (1995). Stretching Mesangial Cells Stimulates Tyrosine Phosphorylation of Focal Adhesion Kinase Pp125(Fak). Biochemical and Biophysical Research Communications, 212(2), 544–549.
Mofrad, M. R., Golji, J., Abdul Rahim, N. A., & Kamm, R. D. (2004). Force-induced unfolding of the focal adhesion targeting domain and the influence of paxillin binding. Mechanics and Chemistry of Biosystems, 1(4), 253–65.
Johnson, K. R., Leight, J. L., & Weaver, V. M. (2007). Demystifying the effects of a three-dimensional microenvironment in tissue morphogenesis. Methods in Cell Biology, 83(1), 547–583.
Acknowledgments
We apologize to the many authors whose work is not cited due to space limitations. SK acknowledges grant support from the University of California Cancer Research Coordinating Committee, the Arnold and Mabel Beckman Foundation, the American Heart Association (0765128Y), the National Science Foundation (CMMI-0727420), and the NIH Director’s New Innovator Award (1DP2OD004213), a part of the NIH Roadmap for Medical Research. VMW acknowledges grant support from NIH NCI grant 7R01CA078731-07, DOD Breast Cancer Research Era of Hope grant W81XWH-05-1-330 (BC044791), CIRM grant RS1-00449 and DOE Low Dose Radiation grant A107165.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Kumar, S., Weaver, V.M. Mechanics, malignancy, and metastasis: The force journey of a tumor cell. Cancer Metastasis Rev 28, 113–127 (2009). https://doi.org/10.1007/s10555-008-9173-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10555-008-9173-4