Abstract
Besides LV ejection fraction (LVEF), global longitudinal strain (GLS) and global myocardial work index (GWI) are increasingly important for the echocardiographic assessment of left ventricular (LV) function in athletes. Since exercise testing is frequently performed on a treadmill, we investigated the impact of upright posture on GLS and GWI. In 50 male athletes (mean age 25.7 ± 7.3 years) transthoracic echocardiography (TTE) and simultaneous blood pressure measurements were performed in upright and left lateral position. LVEF (59.7 ± 5.3% vs. 61.1 ± 5.5%; P = 0.197) was not affected by athletes’ position, whereas GLS (− 11.9 ± 2.3% vs. − 18.1 ± 2.1%; P < 0.001) and GWI (1284 ± 283 mmHg% vs. 1882 ± 247 mmHg%; P < 0.001) were lower in upright posture. Longitudinal strain was most frequently reduced in upright posture in the mid-basal inferior, and/or posterolateral segments. Upright posture has a significant impact on LV deformation with lower GLS, GWI and regional LV strain in upright position. These findings need to be considered when performing echocardiography in athletes.
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
To prevent life-threatening events (e.g. sudden cardiac death) in competitive athletes with unknown cardiovascular disease, transthoracic echocardiography (TTE) is recommended by major sport associations [1].
Although left ventricular (LV) ejection fraction (LVEF) is mainly used to characterize LV systolic function, LV global longitudinal strain (GLS) has shown to be more sensitive in the detection of subclinical LV dysfunction than LVEF [, , , 2,3,4,5]. Further, GLS assessed by speckle tracking analysis has shown lower intra- and interobserver variabilities compared to LVEF measurements [6]. In healthy individuals GLS varies from − 22% to − 18% [7], whereas the inter-vendor variability of GLS analyses need to be considered [, 7, 8].
While GLS is dependent on pre- and afterload conditions [9], global myocardial work index (GWI) is a modern echocardiographic parameter which has been shown to be afterload independent with respect to myocardial deformation and contractile function [, 10, 11]. Based on GWI, further parameters such as global myocardial work efficiency (GWE) defined as the percentage ratio of constructive work (GCW) to the sum of GCW and wasted work (GWW) enable the analysis of LV function irrespective of afterload conditions [11]. Normal values of GWI and GCW vary from 1900 to 2100 mmHg % to 2200–2400 mmHg %. Reference values of GWW is defined from 73 to 87 mmHg%, and mean GWE is about 96% [12].
TTE at rest is usually performed in left lateral position. In competitive athletes it is not uncommon that exercise testing is performed in upright posture—especially when testing on a treadmill [13]. While the impact of body position on physiological parameters (e.g. blood pressure, heart rate) is well described [14], the impact of upright posture on LV deformation parameters has not been described yet.
The objective of this study was to analyze the impact of upright posture on echocardiographic parameters of LV deformation in healthy athletes. We hypothesized that upright posture has no significant impact on LV deformation.
Methods
In this study 50 male athletes who underwent TTE during pre-participation screening at the University Hospital Leipzig between March 2018 and August 2021, were included. All athletes were practicing sports at a competitive level with more than 20 h of training per week. They were examined in upright posture and left lateral position and provided informed consent after full explanation of the purpose and order of all procedures (Fig. 1). The study was conducted in accordance with the Declaration of Helsinki and approved by the ethical committee of the University of Leipzig (073/18-ek).
TTE was performed according to a standardized protocol using a Vivid e9 or Vivid e95 ultrasound system with a 4Vc phased array probe (GE Healthcare Vingmed Ultrasound AS, Horten, Norway). All examinations were documented by a board certified cardiologist. Data sets were analyzed by post-processing analyses using the EchoPac software (Version 204, GE Healthcare Vingmed Ultrasound AS, Horten, Norway). Systolic and diastolic blood pressure was measured in all patients after five minutes resting in upright position and in left lateral position.
Conventional echocardiographic parameters
The left ventricular outflow tract diameter (DLVOT) was determined in the parasternal long axis view in the left lateral position. Relative wall thickness (RWT) was calculated by twice of the LV posterior wall diameter (LVPWD) divided by LV end-diastolic diameter (LVEDD) measured by anatomical M-Mode in parasternal short axis views verified by biplane scanning. LV longitudinal axis diameter (LVLD) was assessed in the apical long axis view by measuring the distance from the apex to the center of the closed mitral valve at end-diastole. LV mass (LVM) was calculated by the Deveraux formula and was indexed to the body surface area (LVMi). Normal ranges of LVMi were defined < 115 g/m2 (males) [15].
The LVOT velocity time integral (VTILVOT) was measured by pulsed waved (PW-) Doppler in the apical long axis view with the sample volume exactly at DLVOT measurement position. LV stroke volume (SVDoppler) was calculated by the cross-sectional area of the LVOT multiplicated by VTILVOT. The LVEF, LV end-diastolic (LVEDV) and end-systolic (LVESV) volumes and LVSVbiplane were assessed by LV biplane planimetry by the modified Simpson’s rule in the apical 2- and 4-chamber view [15]. Regarding both approaches the LVSV was indexed to the body surface area (LVSVi). Left atrial volume index (LAVi) was determined according to current recommendations [15].
Deformation imaging
Deformation imaging was based on speckle tracking analyses using standardized apical views differing by 60° (long axis view, 2-chamber and 4-chamber view) according to current recommendations [16]. The beginning of the QRS complex was set as the reference point, whereas the end-systole was defined by the end of the ejection period obtained from PW Doppler measurements in the LVOT. The region of interest (ROI) of the myocardial tracking area was adjusted to the endocardial and epicardial border. LV longitudinal deformation was assessed to detect regional deformation abnormalities using the 18-segment model comprising all apical views [15]. Regional longitudinal strain values were determined for each LV segment, GLS of > − 16.7% was considered to be normal [17]. GWI, GCW, GWW and GWE of the LV were calculated by post-processing analyses taking systolic and diastolic blood pressure into account.
Statistical analysis
All statistical analyses were performed using SPSS Statistics version 28.0 (IBM, Armonk, NY). Normal distribution was tested using Kolmogorov–Smirnov. Continuous variables were expressed as mean value ± standard deviation (SD) and were compared between groups using Student’s t-test. All categorical variables were expressed as numbers with their percentages (%) and compared using chi-squared or Fisher exact test, as appropriate. Linear regression and Pearson’s r were applied to evaluate association between two linear variables. Data comparisons between more than two groups were performed by one-way Analysis of Variance (ANOVA). A P value < 0.05 was considered statistically significant.
Kappa coefficient (κ) was used to assess intra- and interobserver variabilities in 20 patients under the same conditions. The second investigator was unaware of the results of the first examination.
Results
Baseline demographic characteristics
In 50 male athletes (25.7 ± 7.3 years) systolic (128.3 ± 8.3 mmHg vs. 125.3 ± 9.9 mmHg; P = 0.104) and diastolic blood pressure values (74.3 ± 6.8 mmHg vs. 72.9 ± 12.2 mmHg; P = 0.482) did not differ in upright posture compared to left lateral position. The heart rate was higher in upright posture compared to left lateral position (79.1 ± 13.9 /min vs. 61.0 ± 10.1 /min; P < 0.001). Further demographic data are presented in Table 1.
Conventional echocardiography
Whereas LVMi was not affected by position changing (98.3 ± 21.0 g/m2 vs. 106.0 ± 16.80 g/m2 P = 0.072), RWT was higher in upright posture compared to left lateral position (0.40 ± 0.05 vs. 0.35 ± 0.05; p < 0.001). LVLD, LVEDD and LVESD were lower in upright posture compared to left lateral position (Table 2). Stroke volume measurement LVSVDoppler (65.3 ± 14.2 ml vs. 94.7 ± 15.8 ml; P < 0.001) and LVSVbiplane (69.1 ± 16.3 ml vs. 95.1 ml ± 16.0 ml; P < 0.001) decreased in upright compared to left lateral position.
Parameters of left ventricular systolic function
LVEF did not differ significantly between upright and left lateral position (61.1 ± 5.5% vs. 59.7 ± 5.3%; P = 0.197, Table 3). GLS, GWI, GCW and GWE were significant lower in upright posture compared to left lateral position (Table 3). GWW was significant higher in upright posture compared to left lateral position. In all athletes a reduction of regional longitudinal strain was observed in upright posture in the inferior and/or posterolateral LV segments (Figs. 2, 3). According to longitudinal strain, a regional reduction of the myocardial work index was observed in the inferior and/or posterolateral segments.
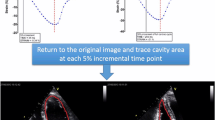
Global longitudinal strain measured by 2D echocardiography in upright (A-C) and left lateral position in the same athlete (D-F). Apical long axis, 2- and 4-chamber view (A1-A3, D1-D3) with anatomical M-Mode in the apical long axis view (B1-B2, E1-E2). Strain curves of all three apical views (C1-C3, F1-F3) with the corresponding bull’s eye (C4, F4). The yellow arrow indicates changes in left ventricular wall motion in comparison to left lateral position. AVO Aortic valve opening, AVC Aortic valve closing, MVO Mitral valve opening, MVC Mitral valve closing
Assessment of global myocardial work index in upright (A) and left lateral position (B). Bull’s eye of global myocardial work index (A1, B1) with the pressure-strain-loop of the mid-lateral segment of the left ventricle (A2, B2). Comparison of constructive and wasted work in the mid-lateral segment of the left ventricle (A3, B3)
Intra- and Interobserver variability
Intraobserver variability showed high agreement for GLS in upright posture (κ = 0.81; z = 4.1, P < 0.001) as well as in left lateral position (κ = 0.88; z = 4.43, P < 0.001). Interobserver variability between two investigators showed equally good agreement for GLS in upright posture (κ = 0.74; z = 4.27, P < 0.001) as well as in left lateral position (κ = 0.86; z = 5.11; P < 0.001). Intra- and interobserver variabilities for the remaining conventional echocardiographic measurements consistently showed good agreement as well.
Discussion
The main findings of the present study are the detection of significant differences of LV morphology and function between upright and left lateral position determined by TTE at rest. These results are based on (1) smaller LV volumes and shorter longitudinal LV axis, (2) significantly reduced GLS—mainly due to regional deformation differences in the basal inferior and posterolateral LV segments, and (3) lower GWI in upright position.
Changes of LV volumes in relation to body position have been reported by Nixon et al. observing a decrease of 30–45% of LVSV as well as an increase of 25–45% in heart rate in healthy young men after mobilization from supine to upright position [18]. Upright position increased long axis shortening, LVEDD and LVESD according to Sundblad and Wranne as shown in healthy subjects [19]. Goodman et al. analyzed left ventricular function after leg cycling and reported an increase of LVEDV, possibly due to increased venous return [20]. These observations were in line with the results of our study and could be physiologically explained by a fast response of the smooth muscles of the venous and arterial vessels under the diaphragm, the autonomic system influencing the general vasoconstrictor tone, the skeletal muscles of the lower limb with its “pump function” and the neurohormonal response [21]. The impact of postural change on LVSV can been described as a manifestation of Frank-Starling mechanism, which has been observed in previous studies as an adaptive response to orthostatic stress [, 22, 23]. Clinically this effect is useful in evaluation of fluid response. The passive leg raising (PLR) test has been used to evaluate volume response in various clinical settings. PLR increased stroke volume > 10% in 45% of the healthy subjects [24]. Sureh et al. reported a significant increase in left ventricular volume and SV after PLR in patients with coronary artery disease and cardiac surgery in a prospective study [25]. Further data is available from critically ill patients where PLR increased the enddiastolic volume and cardiac output predicting fluid response [26]. Corresponding to this pathophysiologic concept, LAVi increased significantly in left lateral position demonstrating the left atrial reservoir function [27].
The dependence of LV function - intrinsic contractility and relaxation - on pre- and afterload is an accepted physiological concept [28]. Nafati el al. showed that GLS was affected by different preload conditions in a population of intensive care unit patients [29]. Negishi et al. demonstrated the influence of preload modification on hemodynamic and echocardiographic parameters including GLS by tilt-induced hydrostatic stress emulating different types of gravity in astronauts [30]. Likewise, clinical studies of patients with aortic valve stenosis treated by transcatheter aortic valve implantation or surgical aortic valve replacement, have documented an improvement of GLS due to reduced afterload conditions after therapy [, , 31,32,33]. Roy et al. reported 60 patients with acute circulatory failure, PLR test - suggesting fluid responsiveness - was positive in 55% of the patients. The VTILVOT increased according in this group. The longitudinal strain increased by 19%, but the increase in GLS was also shown in the group with negative PLR [34]. This underlines the preload dependence of GLS. Both, LVEF and GLS, are pre- and afterload dependent, but the models of failing hearts suggest that GLS may be more sensitive to preload than LVEF, whereas the latter is more afterload dependent as shown in 1065 patients with heart failure and reduced LVEF [35]. Moreover, the prognostic value of GLS was superior compared with all other echocardiographic parameters. GLS is supposed to be an important parameter to characterize LV function in comparison to LVEF – especially to detect subclinical LV dysfunction. A GLS-reduction of 15% was shown by Locquet et al. in a prospective study in oncologic patients evaluating cardiotoxicity after chemotherapy before clinical symptoms appeared [4]. Meanwhile, GLS is recommended by current guidelines [36]. At rest, upright position caused a 27% relative GLS reduction compared to left lateral position in this cohort of healthy athletes which was likely caused by regional deformation abnormalities of the inferior and/or posterolateral wall.
Changes of LV wall motion in the mid-basal inferior and/or posterolateral segments in upright posture, have already been described by Sasaki et al. in a cohort of healthy volunteers [37]. The authors hypothesized that the observed changes of LV wall motion in upright posture might be influenced by the anatomic position of the heart, particularly by its proximity to the diaphragm and posterior mediastinum [37]. Sakurai et al. has described a pseudo-asynergy of the LV inferior wall in normal subjects explained by close contact of the heart with the diaphragm [38]. Whereas wall motion abnormalities of the LV in supine position are strongly related with cardiovascular events and death, changes of LV wall motion in upright posture seem to be a physiological response to position changing [39]. The combination of both changes, the anatomical position of the heart and the altered preload in upright posture, could be a plausible explanation for these findings. To our knowledge, the effects of upright posture on echocardiographic parameters of LV deformation have not previously been described. Although these findings obviously cannot be interpreted as “subclinical LV dysfunction”, they are clinically important because exercise testing in athletes is often performed in an upright posture, especially during treadmill testing. Lower strain values in upright posture could be by impaired LV filling conditions, because GLS measurements are load dependent. In an experimental animal model an increase in LV afterload induced by aortic banding led to a GLS decrease [40]. The decreased GLS in the upright posture compared to the left lateral position is probably due to orthostasis and has significant effects on LV deformation.
Longitudinal strain and work index have been advocated as more reliable methods in the assessment of LV function and are able to detect subtle abnormalities. GWI is derived and incorporates afterload information adjusting the strain results to a noninvasive measurement of LV pressure. From the physiological point of view, the pressure–volume–area predicts myocardial oxygen consumption and provides a tool to describe coupling of LV mechanical performance to energy use [41]. More recently, Russell et al. introduced a non-invasive analysis of LV pressure-strain-loops [10]. Since GLS is preload dependent, GWI as function of GLS and afterload should depend on preload as well as afterload. This explains the significant increase in GWI in left lateral position as shown in this study. The same pathophysiological mechanisms of increased preload or autotransfusion as seen in GLS are responsible for this mechanism. More preload shifts the LV function to the right on the pressure–volume–curve and increases myocardial work. To our knowledge this is the first study, which examines this strong effect of pre-stretching the LV cavity on GWI in healthy subjects. We can confirm the pathophysiological assumption of preload dependency.
In conclusion both GLS and GWI are significantly preload dependent. When considering using those parameters in the assessment of LV function, the body position of the patient needs to be taking into consideration. Unfortunately, probably the most reports on normal values are derived in left lateral position. Therefore, there is an urgent need for normal values depending on the body position.
Limitations
In this study, only young and healthy athletes were included. Only TTE examinations of athletes with optimal imaging quality were included, athletes with poor imaging were excluded. Thus, the results are not directly transferable on patients with cardiovascular diseases.
Conclusion
Upright posture has a significant impact on LV deformation. The reduction of regional longitudinal strain in the inferior and/or posterolateral LV segments are presumably explained by different LV filling conditions based on orthostasis and cardiac interaction with the diaphragm. A reduction of regional strain at rest in upright posture is not necessarily pathological but has a significant impact on investigating athletes, especially in case of treadmill testing.
References
Mont L, Pelliccia A, Sharma S et al (2017) Pre-participation cardiovascular evaluation for athletic participants to prevent sudden death: position paper from the EHRA and the EACPR, branches of the ESC. endorsed by APHRS, HRS, and SOLAECE. Eur J Prev Cardiol 24:41–69. https://doi.org/10.1177/2047487316676042
Plana JC, Galderisi M, Barac A et al (2014) Expert consensus for multimodality imaging evaluation of adult patients during and after cancer therapy: a report from the American society of echocardiography and the European association of cardiovascular imaging. J Am Soc Echocardiogr 27:911–939. https://doi.org/10.1016/j.echo.2014.07.012
Tops LF, Delgado V, Marsan NA, Bax JJ (2017) Myocardial strain to detect subtle left ventricular systolic dysfunction: LV systolic dysfunction and GLS. Eur J Heart Fail 19:307–313. https://doi.org/10.1002/ejhf.694
Locquet M, Spoor D, Crijns A et al (2022) Subclinical left ventricular dysfunction detected by speckle-tracking echocardiography in breast cancer patients treated with radiation therapy: a six-month follow-up analysis (MEDIRAD EARLY-HEART study). Front Oncol 12:883679. https://doi.org/10.3389/fonc.2022.883679
Biering-Sørensen T, Biering-Sørensen SR, Olsen FJ et al (2017) Global longitudinal strain by echocardiography predicts long-term risk of cardiovascular morbidity and mortality in a low-risk general population: the Copenhagen city heart study. Circ Cardiovasc Imaging. https://doi.org/10.1161/CIRCIMAGING.116.005521
Kouris NT, Kostopoulos VS, Psarrou GA et al (2021) Left ventricular ejection fraction and Global Longitudinal Strain variability between methodology and experience. Echocardiogr Mt Kisco N 38:582–589. https://doi.org/10.1111/echo.15025
Yingchoncharoen T, Agarwal S, Popović ZB, Marwick TH (2013) Normal ranges of left ventricular strain: a meta-analysis. J Am Soc Echocardiogr 26:185–191. https://doi.org/10.1016/j.echo.2012.10.008
Marwick TH (2010) Consistency of myocardial deformation imaging between vendors. Eur J Echocardiogr 11:414–416. https://doi.org/10.1093/ejechocard/jeq006
Hubert A, Le Rolle V, Leclercq C et al (2018) Estimation of myocardial work from pressure–strain loops analysis: an experimental evaluation. Eur Heart J - Cardiovasc Imaging 19:1372–1379. https://doi.org/10.1093/ehjci/jey024
Russell K, Eriksen M, Aaberge L et al (2012) A novel clinical method for quantification of regional left ventricular pressure–strain loop area: a non-invasive index of myocardial work. Eur Heart J 33:724–733. https://doi.org/10.1093/eurheartj/ehs016
Chan J, Edwards NFA, Khandheria BK et al (2019) A new approach to assess myocardial work by non-invasive left ventricular pressure–strain relations in hypertension and dilated cardiomyopathy. Eur Heart J - Cardiovasc Imaging 20:31–39. https://doi.org/10.1093/ehjci/jey131
Truong VT, Vo HQ, Ngo TNM et al (2021) Normal ranges of global left ventricular myocardial work indices in adults: a meta-analysis. J Am Soc Echocardiogr Off Publ Am Soc Echocardiogr S0894–7317(21):00826–00829. https://doi.org/10.1016/j.echo.2021.11.010
Cotrim C, João I, Fazendas P et al (2013) Clinical applications of exercise stress echocardiography in the treadmill with upright evaluation during and after exercise. Cardiovasc Ultrasound 11:26. https://doi.org/10.1186/1476-7120-11-26
Suojanen LJ, Koskela JK, Wilenius M et al (2021) Individual changes of central blood pressure in response to upright posture: different hemodynamic phenotypes. J Hypertens 39:2403–2412. https://doi.org/10.1097/HJH.0000000000002965
Lang RM, Badano LP, Mor-Avi V et al (2015) Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American society of echocardiography and the European association of cardiovascular imaging. J Am Soc Echocardiogr 28:1-39.e14. https://doi.org/10.1016/j.echo.2014.10.003
Voigt J-U, Pedrizzetti G, Lysyansky P et al (2015) Definitions for a common standard for 2D speckle tracking echocardiography: consensus document of the EACVI/ASE/industry task force to standardize deformation imaging. Eur Heart J - Cardiovasc Imaging 16:1–11. https://doi.org/10.1093/ehjci/jeu184
Sugimoto T, Dulgheru R, Bernard A et al (2017) Echocardiographic reference ranges for normal left ventricular 2D strain: results from the EACVI NORRE study. Eur Heart J - Cardiovasc Imaging 18:833–840. https://doi.org/10.1093/ehjci/jex140
Nixon PGF (1988) Human circulation regulation during physical stress. L. B. Rowell. Oxford University Press, London, 1986. No. of pages: 416. Price: £35.00. Stress Med 4:124–125. https://doi.org/10.1002/smi.2460040213
Sundblad P, Wranne B (2002) Influence of posture on left ventricular long- and short-axis shortening. Am J Physiol Heart Circ Physiol 283:H1302-1306. https://doi.org/10.1152/ajpheart.01041.2001
Goodman JM, Freeman MR, Goodman LS (2007) Left ventricular function during arm exercise: influence of leg cycling and lower body positive pressure. J Appl Physiol 102:904–912. https://doi.org/10.1152/japplphysiol.00511.2006
Smith JJ, Porth CM, Erickson M (1994) Hemodynamic response to the upright posture. J Clin Pharmacol 34:375–386. https://doi.org/10.1002/j.1552-4604.1994.tb04977.x
Dickhuth HH, Simon G, Heiss HW et al (1981) Comparative echocardiographic examinations in sitting and supine position at rest and during dynamic exercise. Int J Sports Med 2:178–181. https://doi.org/10.1055/s-2008-1034607
Rodeheffer RJ, Tanaka I, Imada T et al (1986) Atrial pressure and secretion of atrial natriuretic factor into the human central circulation. J Am Coll Cardiol 8:18–26. https://doi.org/10.1016/s0735-1097(86)80086-9
Godfrey GEP, Dubrey SW, Handy JM (2014) A prospective observational study of stroke volume responsiveness to a passive leg raise manoeuvre in healthy non-starved volunteers as assessed by transthoracic echocardiography. Anaesthesia 69:306–313. https://doi.org/10.1111/anae.12560
Suresh V, Sethuraman M, Karunakaran J, Koshy T (2020) Fluid responsiveness to passive leg raising in patients with and without coronary artery disease: a prospective observational study. Ann Card Anaesth 23:439. https://doi.org/10.4103/aca.ACA_73_19
Monnet X, Jabot J, Maizel J et al (2011) Norepinephrine increases cardiac preload and reduces preload dependency assessed by passive leg raising in septic shock patients*. Crit Care Med 39:689–694. https://doi.org/10.1097/CCM.0b013e318206d2a3
Blume GG, Mcleod CJ, Barnes ME et al (2011) Left atrial function: physiology, assessment, and clinical implications. Eur J Echocardiogr 12:421–430. https://doi.org/10.1093/ejechocard/jeq175
Zimmer H-G (2002) Who discovered the Frank-Starling mechanism? Physiology 17:181–184. https://doi.org/10.1152/nips.01383.2002
Nafati C, Gardette M, Leone M et al (2018) Use of speckle-tracking strain in preload-dependent patients, need for cautious interpretation! Ann Intensive Care 8:29. https://doi.org/10.1186/s13613-018-0376-8
Negishi K, Borowski AG, Popović ZB et al (2017) Effect of gravitational gradients on cardiac filling and performance. J Am Soc Echocardiogr 30:1180–1188. https://doi.org/10.1016/j.echo.2017.08.005
Delgado M, Ruiz M, Mesa D et al (2013) Early improvement of the regional and global ventricle function estimated by two-dimensional speckle tracking echocardiography after percutaneous aortic valve implantation speckle tracking after corevalve implantation. Echocardiography 30:37–44. https://doi.org/10.1111/j.1540-8175.2012.01808.x
Grabskaya E, Becker M, Altiok E et al (2011) Impact of transcutaneous aortic valve implantation on myocardial deformation: myocardial deformation in TAVI. Echocardiography 28:397–401. https://doi.org/10.1111/j.1540-8175.2010.01378.x
Carasso S, Cohen O, Mutlak D et al (2009) Differential effects of afterload on left ventricular long- and short-axis function: insights from a clinical model of patients with aortic valve stenosis undergoing aortic valve replacement. Am Heart J 158:540–545. https://doi.org/10.1016/j.ahj.2009.07.008
Roy C, Duclos G, Nafati C et al (2021) Left ventricular longitudinal strain variations assessed by speckle-tracking echocardiography after a passive leg raising maneuver in patients with acute circulatory failure to predict fluid responsiveness: a prospective, observational study. PLoS One 16:e0257737. https://doi.org/10.1371/journal.pone.0257737
Sengeløv M, Jørgensen PG, Jensen JS et al (2015) Global longitudinal strain is a superior predictor of all-cause mortality in heart failure with reduced ejection fraction. JACC Cardiovasc Imaging 8:1351–1359. https://doi.org/10.1016/j.jcmg.2015.07.013
Lyon AR, López-Fernández T, Couch LS et al (2022) 2022 ESC Guidelines on cardio-oncology developed in collaboration with the European hematology association (EHA), the European society for therapeutic radiology and oncology (ESTRO) and the international cardio-oncology society (IC-OS). Eur Heart J 43:4229–4361. https://doi.org/10.1093/eurheartj/ehac244
Sasaki H, Charuzi Y, Sugiki Y et al (1988) Paradoxical motion of the posterior left ventricular wall seen in healthy subjects in the upright position. J Am Soc Echocardiogr 1:406–409. https://doi.org/10.1016/S0894-7317(88)80022-1
Sakurai S, Takenaka K, Shiojima I et al (2004) Close physical contact of the heart with diaphragm causes Pseudo-Asynergy of left ventricular inferior wall in normal subjects. Echocardiography 21:573–579. https://doi.org/10.1111/j.0742-2822.2004.03058.x
Cicala S, de Simone G, Roman MJ et al (2007) Prevalence and prognostic significance of wall-motion abnormalities in adults without clinically recognized cardiovascular disease: the strong heart study. Circulation 116:143–150. https://doi.org/10.1161/CIRCULATIONAHA.106.652149
Donal E, Bergerot C, Thibault H et al (2009) Influence of afterload on left ventricular radial and longitudinal systolic functions: a two-dimensional strain imaging study. Eur J Echocardiogr 10:914–921. https://doi.org/10.1093/ejechocard/jep095
Takaoka H, Takeuchi M, Odake M, Yokoyama M (1992) Assessment of myocardial oxygen consumption (Vo2) and systolic pressure-volume area (PVA) in human hearts. Eur Heart J 13:85–90. https://doi.org/10.1093/eurheartj/13.suppl_E.85
Funding
Open Access funding enabled and organized by Projekt DEAL. The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
AH performed the standardized echocardiographic examinations. JK and MM wrote the manuscript and performed the echocardiographic measurements. UL, RPM, SS and AH helped writing the manuscript. In addition, SS helped supervise the study. JK and MM contributed equally to the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kandels, J., Metze, M., Hagendorff, A. et al. The impact of upright posture on left ventricular deformation in athletes. Int J Cardiovasc Imaging 39, 1123–1131 (2023). https://doi.org/10.1007/s10554-023-02820-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-023-02820-2