Abstract
The contradictory findings of recent prospective randomized controlled trials assessing the impact of percutaneous edge-to-edge repair in patients with functional or secondary mitral regurgitation have triggered a lively discussion about an “integrated” echocardiographic approach for grading severity of mitral regurgitation. In the MITRA-FR trial, the COAPT trial and the REDUCE-FMR trial echocardiographic assessment of the severity of mitral regurgitation was consistent with principles set forth by the current echocardiographic guidelines and analysed in its best settings by expert international leaders in the field of echocardiography. However, serious inconsistencies appeared in the presented echocardiographic assessments regarding cardiac output and regurgitant fraction. A new term “disproportionate functional mitral regurgitation” was introduced describing a situation where the increase of effective regurgitant orifice area exceeds the enlargement of the left ventricular end-diastolic volumes. Further discussion resulted in the idea of a “new conceptional framework” for distinguishing “proportionate” and “disproportionate” functional mitral regurgitation. The aim of this viewpoint is to dispute conclusions based on the term “disproportionate” mitral regurgitation. A “disproportionate” FMR is highly questionable because disproportionateness of flow in communication vessels cannot exist. In addition, a proposal of echocardiographic assessment based on a conventional comprehensive transthoracic echocardiography is given to avoid obvious hemodynamic contradictions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Inconsistencies between the left ventricular (LV) total stroke volume (LVSVtot) obtained with two-dimensional (2D) planimetry by transthoracic echocardiography (TTE), the Doppler-derived effective LV forward SV (LVSVeff) and the mitral regurgitant volume (RegVolMV) obtained with the 2D proximal isovelocity surface area (2D-PISA)-method can be observed in all recent transcatheter mitral valve repair (TMVR) trials [1,2,3]. Taken together, the mean values of the echocardiographic parameters presented in these trials characterize a hemodynamic state resembling conditions below cardiogenic shock index or inconsistent with severe functional mitral regurgitation, FMR [4,5,6,7]. The so-called “integrated approach” of grading FMR severity can be scrutinised because the characterization of the hemodynamics by echocardiography in patients with FMR obviously had failed [8, 9]. Thus, a proposal like „a specific integrative multiparametric MR grading algorithm that could identify a homogeneous population that would benefit from TMVR“ [10] is in doubt. This viewpoint might contribute to the scientific debate for the need of more conclusive echocardiographic FMR assessment as applied in the recent TMVR trials. In light of the fact, that the echocardiography performed in the TMVR trials is presumably divisions above whatever is done in routine practice, and data analysis was performed by international expert leaders, the attempts to explain the differences of inconsistent echocardiographic data [1,2,3,4,5,6,7, 10,11,12,13,14,15,16,17,18,19,20,21,22,23] have to be critically discussed. If we still want to use echocardiography to assess FMR, because it is the most common tool, which can be used, we need substantial methodological improvements.
The first objective of this viewpoint is to discuss whether a “disproportionate” FMR can be possible anyway with the conclusion that disproportionateness of flow in communication vessels cannot exist. The second objective is to propose an extended transparent echocardiographic protocol focusing on hemodynamic plausibility to improve the grading FMR severity.
General rheological considerations in valvular heart diseases
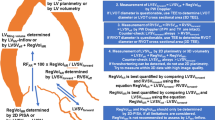
The calculation of the effective orifice area by the continuity equation is an accepted method for echocardiographic grading of aortic valve (AV) severity based on the physical laws of conservation of mass and energy. These principles cannot be neglected meaning that blood flow velocities at defined orifices are proportional in a system of communicating tubes. If we assume “pure” aortic valve stenosis (AS), and if cross sectional areas are known at the level of the left ventricular outflow tract (LVOT) as well as at the level of the stenotic orifice area, LVSVtot can be measured at the level of the LVOT as well as at the level of the stenotic orifice area by Doppler echocardiography, because volume flow has to be the same at both levels [24, 25]. In “pure” mitral regurgitation the same principle can be applied at the regurgitant orifice area; RegVolMV is the calculated difference LVSVtot − LVSVeff. Thus, if forward stroke volume LVSVeff is known, the interrelationship between effective regurgitant orifice area (EROA) and regurgitant flow can be calculated due to physical laws of rheology (Fig. 1).
Illustration of the proportionality between cross sectional areas and forward and backward systolic blood flow in the left ventricle as a system of communicating tubes. Transmitral regurgitant blood flow volume is proportional to the regurgitant orifice area, if a single timepoint measurement is performed by echocardiography. CSAAV cross section area of the aortic valve orifice, CSAEROA cross section area of the mitral valve regurgitant orifice, CSALVOT cross section area of the left ventricular outflow tract, CSAMV cross section area at the level of the mitral valve annulus, DAV diameter of the aortic valve orifice, DEROA diameter of the mitral valve regurgitant orifice, DLVOT diameter of the left ventricular outflow tract, DMV diameter at the level of the mitral valve annulus, LVSVeff left ventricular effective stroke volume, LVSVtot left ventricular total stroke volume, RegVolMV transmitral regurgitant volume, VTIAV velocity time integral of the systolic forward blood flow through the aortic valve orifice, VTIEROA velocity time integral of the diastolic backward blood flow through the mitral valve regurgitant orifice, VTILVOT velocity time integral of the systolic forward blood flow through the left ventricular outflow tract, VTIMV velocity time integral of the diastolic forward mitral flow at the level of the mitral annulus, VTI-RegMV velocity time integral of the systolic regurgitant transmitral blood flow at the level of the mitral valve annulus
To introduce into the hemodynamic discussion in FMR the parameters cardiac output (CO) and the cardiac index (CI) determined by the LVSVeff, heart rate (HR) and body surface area (BSA) should be the basis of the physiological thinking in echocardiography. Further important cardiac parameters are the LV end-diastolic volume (LVEDV) and the LV ejection fraction (LVEF) to interpret the cardiovascular physiology. Normal CO is defined within ranges of 4.0 to 4.5 l/min or a CI of about 2.5 l/min/m2. A CI < 2.2 l/min/m2 is a criterium for cardiogenic shock (CO < about 4 l/min). A normal heart rate at rest—and the target heart rate for cardiovascular patients with optimal medical treatment (OMT)—is within the ranges of 50 to 70/min. With respect to these values of a normal cardiovascular physiology a borderline region to differentiate between normal conditions and cardiac decompensation can be marked in a LVSVeff − LVEDV diagram with respect to different LVEF (Fig. 2). To ensure sufficient cardiac output it is obvious that no mitral as well as aortic regurgitation is present, because LVSVtot must be equal to LVSVeff (Fig. 2).
The relation between total left ventricular (LV) stroke volume (LVSVtot) and LV end-diastolic volume (LVEDV) with respect to LV ejection fraction (LVEF) in the presence of no mitral regurgitation (MR) and no aortic regurgitation (AR): LVSVeff LV effective stroke volume, CO cardiac output, CI cardiac index (additional explanations in the text)
If the mean values of LVSVtot and LVEDV of the recent TMVR trials [1,2,3] are put into the diagram of Fig. 2, the colored dots represent the respective relationships (Fig. 3). Table 1 illustrates the hemodynamic parameters of the recent TMVR trials reported in the literature as well as the assumptions resulting from logical calculations of the presented data [1,2,3]. To explain the differences in the COAPT trial version 1 is corresponding to the presented data of the original paper [2], version 2 is corresponding to the authors reply to the letter to the editor [4]. In this reply the authors issued the statement “The actual mean forward stroke volume in the COAPT trial as measured with Doppler was 51 ml, and the regurgitant volume as measured with the use of the PISA method was 59 ml, values that are consistent with severe mitral regurgitation” [4, 17, 18]. However, if the forward stroke volume is subtracted from the LVSVtot, the RegVolMV is only 8 ml, revealing a difference of 51 ml RegVolMV (RegVolMV = LVSVtot − LVSVeff) or a difference of 51 ml LVSVtot (LVSVtot = LVSVeff + RegVolMV) in the device group, which is not explained by the COAPT authors (Table 1).
The relation between total left ventricular (LV) stroke volume (LVSVtot), LV end-diastolic volume (LVEDV) and LV ejection fraction (LVEF) in moderate and severe mitral regurgitation (MR) (e.g. 33% and 50% RF). LVSVeff LV effective stroke volume, CO cardiac output, CI cardiac index. Mean values of LVSVtot and LVEDV presented in recent transcatheter mitral valve repair (TMVR) trials are shown in colored dots (green dots COAPT, blue dots MITRA-FR, and orange dots REDUCE-FMR). The red dot displays a left ventricle with a LVEDV of 200 ml and a LVSVeff of 60 ml; the calculated LVSVtot and LVEDV values in the presence of a LVEF of 30% in a moderate FMR (regurgitant fraction, RF = 33%) are LVSVtot = 90 ml and LVEDV = 300 ml (hollow red dots), in a severe FMR (RF = 50%) are LVSVtot = 120 ml and LVEDV = 400 ml (hollow red dots); the calculated LVSVtot and LVEF values in the presence of LVEDV of 200 ml in a moderate FMR (RF = 33%) LVSVtot has to be 90 ml and LVEF 45% (hollow red dots), in a severe FMR (RF = 50%) LVSVtot has to be 120 ml and LVEF 60% (hollow red dots) (additional explanations in the text)
As mentioned above, CO and CI are represented by the multiplication of LVSVeff × HR and not by the multiplication of LVSVtot × HR. Describing the conditions of a LVEDV of 200 ml and a LVEF of 30% at a normal HR of 65/min, the periphery needs the complete 60 ml of the LVSVtot as LVSVeff to ensure sufficient normal CO or CI as illustrated by the red dot (Fig. 3). If moderate FMR with a regurgitant fraction (RF) of 33% is assumed, this left ventricle is characterized by a LVEDV of 300 ml at a LVEF of 30% or by a LVEDV of 200 ml at a LVEF of 45% (brightened red dot in Fig. 3). If severe FMR with a RF of 50% is assumed, this left ventricle is characterized by a LVEDV of 400 ml at a LVEF of 30% or by a LVEDV of 200 ml at a LVEF of 60% (brightened red dot in Fig. 3).
In a CI–HR diagram with respect to different RF a borderline region to differentiate between normal conditions and cardiac decompensation can be marked at the limit of CI of 2.2 l/min/m2 (red bar in Fig. 4). In accordance with the previous diagrams (Figs. 2, 3) LVSVeff must be ≥ 60 ml and HR ≥ 65/min to ensure a CI within normal ranges. The slope of the CI–HR-relationship decreases with increasing RF (dotted lines in Fig. 4). The pathophysiological consequence to ensure a sufficient CO or CI is the increase of HR (red bar in Fig. 4).
The relation between cardiac performance (CO cardiac output, CI cardiac index) and heart rate (HR) with respect to regurgitant fraction (RF): LVSVtot total left ventricular (LV) stroke volume, LVSVeff effective LV stroke volume, RegVolMV regurgitant volume, OMT optimal medical treatment. Mean values of CI and HR presented in recent transcatheter mitral valve repair (TMVR) trials are illustrated by colored dots (green dots COAPT-version 1: LVSVtot = 59 ml, RegVolMV 59 ml, HR presumably about 70/min during OMT; version 2-LVSVtot = 59 ml, LVSVeff = 51 ml, RegVolMV 8 ml, HR presumably about 70/min during OMT), blue dots MITRA-FR (LVSVtot = 82 ml, RegVolMV 45 ml, HR 73/min), and orange dots REDUCE-FMR (LVSVtot = 64 ml, RegVolMV 40 ml, HR 70/min). Thus, calculated HR in the presence of the reported RF to ensure a CI > 2.2 l/min/m2 is illustrated by colored arrows (COAPT-version 1: not possible; version 2 about 77/min), MITRA-FR about 110/min, REDUCE-FMR about 130/min) (additional explanations in the text)
If the mean values of CI and HR of the recent TMVR trials [1,2,3] are put into the diagram, the colored dots represent the respective relationships (Fig. 4). CI-values of the recent TMVR trials were taken or calculated by the LVSVtot, LVSVeff, and RegVolMV-values presented in the literature [1,2,3]. In COAPT HR was not listed, thus, a HR of 65–70/min for cardiovascular patients under OMT was taken for calculation. As obviously shown, all colored dots are below the red bar, which represents the borderline range of CI between normal conditions and cardiogenic shock. As also illustrated by the arrows HR has to be much higher in all recent TMVR trials than presented in the literature or then the HR ranges of OMT to ensure CI > 2.2 l/min/m2 (colored arrows in Fig. 4).
The well-known EROA–LVEDV diagram adapted according to Grayburn et al. [12] should illustrate the proportionality between EROA and LVEDV in patients with severe FMR with a border area. Below this area non severe FMR is characterized. Above this area Grayburn et al. proposed to use the term “disproportionate” severe FMR [12]. Grayburn et al. described the diagnostic scenario that “physicians should seek to determine whether the estimated degree of MR is expected or proportionate to the degree of LV dilatation, or alternatively, whether the severity of MR is unexpected or disproportionate to the degree of LV enlargement” [12].
If the mean values of EROA and LVEDV of the recent TMVR trials [1,2,3] are put into the diagram, the brightened colored dots represent the respective relationships (Fig. 5). However, if these values are corrected with respect to plausible hemodynamics—that means RegVolMV or EROA were reduced to ensure at least borderline CI of 2.2 l/min/m2—all these brightened dots shift into the area of non-severe FMR (colored dots in Fig. 5).
The relation between effective regurgitant orifice area (EROA) and left ventricular (LV) end-diastolic volume (LVEDV) at a LV ejection fraction (LVEF) about 30% [6]: the arrows illustrate the respective calculated EROAs presented in recent transcatheter mitral valve repair (TMVR) trials reduced by the amount in relation to a regurgitant volume (RegVolMV), which ensure at least a cardiac index (CI) > 2.2 l/min/m2. Mean values of EROA and LVEDV are given for green hollow dots COAPT [2, 10, 12], blue hollow dots MITRA-FR [1], and pink hollow dots REDUCE-FMR (assumed mean EROA of 0.26 cm2) [3]. If the EROA is reduced according to laws of rheology and physics, the EROAs of the filled circles have to be assumed in the recent TMVR trials (green dots COAPT, blue dots MITRA-FR, and orange dots REDUCE-FMR) (additional explanations in the text)
In all figures two dots are presented for the COAPT trial. The two dots are explained by the values reported in the literature about COAPT [2, 4]. The first reported LVSVtot by planimetry in COAPT was 51 ml and the RegVolMV was 59 ml which suggests overestimation of EROA and RegVolMV, underestimation of LVEDV, or both due to the impossibility of the specified values [2]. If 51 ml as LVSVeff in the presence of 60 ml of LVSVtot are assumed, a fully different scenario with a mild FMR of 8 ml with a corresponding RF of 14% is described (device cohort).
Conclusions
With respect to hemodynamic implausibility of the echocardiographic data presented in the recent TMVR trials [1,2,3] it might be allowed to search for the reasons for this scenario. If RegVolMV, LVEDV, LVEF and LVSVtot are not conclusive and plausible, the following conclusions can be drawn: RegVolMV is obviously overestimated and planimetry-derived LVEDV is obviously underestimated. Thus, either the methods used should not be applied anymore or they should be applied correctly.
The assessment of MR severity by the echocardiographic “integrated approach” described by the current guideline recommendations is primarily based on semiquantitative analysis of semiquantitative parameters describing MR severity [8, 9, 23]. However, these parameters including the 2D-PISA approach have inherent problems to be methodically prone to errors. The colour flow jet area in the left atrium and its relation to the left atrial size depends on several methodological, anatomic, and pathophysiological factors [26]. Because of many factors, no standardization to adjust the colour flow jet area in MR patients is possible. In consequence, the approach of MR grading based on colour flow jet area is not recommended anymore [8]. The vena contracta (VC) by colour coded Doppler was described in MR patients in the parasternal long axis view because of better axial resolution in comparison to inferior lateral resolution in apical views [27]. Prerequisite of the VC method is the acquisition of the correct longitudinal sectional plane through the regurgitant jet to minimize underestimation beside methodological factors of ultrasound machine settings. Considering the mathematical model of the 2D-PISA method the frequent misuse can be explained e.g. by a inappropriate application in eccentrical jet formations or by an overestimation of RegVolMV by measuring improper PISA radius at false time points and/or at the VC instead of the entry of the EROA [28]. The shape of the EROA and the jet direction are colour coded parameters yielding information to assume a relevant MR [9]. However, both entities should serve as a starting point for a quantitative MR assessment. The systolic flow reversal in the pulmonary veins is influenced by the jet direction towards the respective pulmonary veins, the size of the left atrium, and LV contractility causing over- as well as underestimation of MR severity [29]. The intensity of the MR-signal of the transmitral regurgitation using continuous wave Doppler is not recommended for assessing MR severity due to several practical and methodological limitations [8]. The ratio between transmitral velocity time integral (VTI) and flow velocity within the LVOT (VTIMV/VTILVOT) seems to be suitable for a grading approach of MR severity, if the sample volumes are properly positioned, ultrasound beam alignment is parallel to the blood flow, MV stenosis or aortic valve regurgitation as well as mitral annular dilatation and atrial fibrillation is absent [30]. Due to all these multiple limitations of the semiquantitative parameters it might be necessary to assess quantitatively LVSVtot, LVSVeff and RegVolMV in a similar approach as currently used in cardiac magnetic resonance, CMR [31,32,33]. If echocardiographic measurements provide inconsistent results for LVSVtot, LVSVeff and RegVolMV, the most likely explanation are obviously measurement errors due to methodological factors.
In HF patients with FMR recently two “hemodynamic pathways” have been described to characterize pathophysiological differences [19]. Firstly, LV remodeling defined by LV hypertrophy, LV dilatation, and LV sphericity causes papillary muscle displacement and widening of the mitral annulus with a consecutive FMR. This FMR type should be characterized by a linear relationship between LVEDV and EROA and has been named “proportionate MR”. Secondly, a “disproportionate FMR type” should be possible mainly due to LV dyssynchrony based on electrical conduction delay. This FMR type should be characterized by a greater MR severity than expected solely by LVEDV changes independent of LV geometry [19]. The percentage of HF patient with bundle branch block and FMR in the recent TMVR trials, who primarily are candidates for resynchronization therapy according to recent guidelines [34], has not been transparently presented. However, it was suggested that localized LV remodeling—especially regional wall motion abnormalities of the inferoposterior or lateral wall”—can induce dyssynchronous contraction resulting in a relevant “disproportionate FMR”.
However, the introduction of the terms “proportionate” and “disproportionate” FMR is misleading, because RegVolMV has to be proportional to EROA in a system of communicating vessels (Fig. 1) at a single beat-to-beat measurement. Considering the single timepoint measurement of the PISA radius within the cardiac cycle there are sources of errors in quantifying RegVolMV due to the dynamic nature of MR within systole. However, if RegVolMV is under- or overestimated by this 2D-PISA approach, it is simply a measurement error, which reflects an incorrect assessment of RegVolMV. Inherently, physical laws of conservation of mass and energy cannot be neglected using echocardiography meaning that blood flow velocities at defined orifices are proportional in a system of communicating tubes (Fig. 1). Thus, the basic question raises whether a “disproportionate” FMR can be possible anyway. Instead of arithmetic juggling with inconsistent data the aim of a comprehensive echocardiography should be the correct assessment of conclusive values of LVSVeff, LVSVtot, LVEF and RegVolMV in patients with FMR.
A complete other discussion are dynamic changes of the MR severity with changes of pre- and afterload [35]. It is obviously that RF is altered with increasing CO at the same heart rate, if preload or afterload is decreased, e.g. during sedation. It can be assumed, that RF will disproportionately increase with increasing afterload in FMR patients with advanced HF stages. To proof this concept of an “overproportionate FMR” at minimum two timepoint measurements at different afterload conditions are necessary. It can also be speculated that increased LV wall stress causes more LV remodeling with PM displacement, MV annulus dilatation and leaflet tenting supporting a higher risk of the development of relevant FMR described by the concept of “MR begets MR” in congestive HF [36]. However, again to proof this concept at minimum two timepoint measurements during follow-up under comparable circulatory conditions are necessary. The dynamic changes of MR severity with changes of pre- and afterload underline the importance to standardize measurement conditions during echocardiography—especially in TMVR trials. It is highly questionable to accept a baseline TTE within 90 days and a baseline TEE within 180 days prior to intervention as described in the COAPT Supplementary Appendix [2]. Baseline characteristics defined in this way might be scrutinized with respect to the possible changes of FMR severity due to several reasons. A comparison between a pre-interventional state the day before intervention and a post-interventional state at hospital discharge during comparable conditions—especially at the same heart rate, the same systemic blood pressure and the same drug treatment—should be the prerequisite for a verifiable documentation in clinical trials.
This unfortunate situation of implausible echocardiographic assessment in FMR patients mandates the integration of “hemodynamic conclusiveness” into the recent “integrated approach” [8, 9, 18]. New diagnostic algorithms apart from guidelines [8, 9] to „identify echocardiographic characteristics that predict favourable outcomes after TMVR in heart failure patients with severe secondary MR “ [10] are highly debatable—especially if inconsistent data are followed by treatment recommendations. A greater transparency of all trial data would presumably be helpful for a better understanding. The incongruencies of the reported hemodynamic values in patients with FMR in the recent TMVR trials illustrates the recent echocardiographic weakness in routine practice. The term “disproportionate” FMR is hardly to accept because a disproportionateness of hemodynamics might be just a proof of measuring error or simply a myth.
The objective, reproducible and transparent assessment of echocardiographic parameters for LV function and RF estimation in FMR patients will be the key for a proper decision making.
Proposal of an extended transparent echocardiographic documentation focusing on hemodynamic plausibility in FMR patients FMR
A comprehensive echocardiography should integrate the estimating of cardiovascular parameters by a plausibility-check of the data. Despite the fact, that all cardiologists know, that the accurate assessment of LV volumes and LV function by echocardiography is well possible and methodological limitations in measuring LV volumes and LVEV have to be considered, the echocardiographic documentation with respect to its transparency, reproducibility and objectivity is illustrated regarding to the intention of an imaging journal (Figs. 6, 7, 8, 9).
Illustration of quantitative assessment of left ventricular (LV) volumes in a patient with isolated functional mitral regurgitation (FMR)—part 1: a scheme of LV effective stroke volume (LVSVeff) and regurgitant volume (RegVolMV) in isolated FMR (a); measurements of right ventricular (RV) and LV stroke volume by Doppler echocardiography (RVSVDopp, LVSVDopp) by determination of the diameter of the RV outflow tract (RVOT) (b) and the velocity time integral (VTI) of the RVOT (RVOT VTI) (c) and by determination of the diameter of the LV outflow tract (LVOT) (d) and the LVOT VTI (). In isolated FMR RVSVDopp is equal to RVLVDopp, which represents LVSVeff as well as RVSVeff. LVSVeff is between 65 and 70 ml in this case
Illustration of quantitative assessment of left ventricular (LV) volumes in the same patient with isolated functional mitral regurgitation (FMR) as shown in Fig. 1—part 2: determination of left ventricular (LV) total stroke volume (LVSVtot) by biplane planimetry using Simpson’s method; planimetry of the 2-chamber view during diastole (a, b) and systole (c, d), planimetry of the 4-chamber view during diastole (e, f) and systole (g, h); the biplane LV planimetry enables the determination of LVSVtot, which is the sum of LV effective stroke volume (LVSVeff) and of the regurgitant volume (RegVolMV); LVSVtot is between 85 and 90 ml in this case
Illustration of quantitative assessment of left ventricular (LV) volumes in the same patient with isolated functional mitral regurgitation (FMR) as shown in Figs. 1 and 2—part 3: documentation of the FMR with a central jet formation, the jet area, the proximal convergence areas, and the vena contracta (VC) with a Nyquist limit of 47 cm/s (a), VC measurement with 4 mm (b), the basal septal myocardial velocity for calculation of E/E′ (c), the pulsed wave Doppler spectrum for determination of E velocity, the E/A-ratio and the ratio between transmitral velocity time integral (VTI) and flow velocity within the LVOT (VTIMV/VTILVOT) (d), and the continuous wave Doppler spectrum of the transtricuspid regurgitation for estimation of the systolic pulmonary arterial pressure (sPAP) (e); increased E/E′, increased E-velocity, increased VTIMV/VTILVOT and increased sPAP document the secondary cardiac alterations of a relevant MR
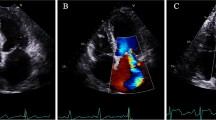
Illustration of quantitative assessment of left ventricular (LV) volumes in the same patient with isolated functional mitral regurgitation (FMR) as shown in Figs. 1, 2 and 3—part 3: determination of regurgitant volume (RegVolMV) by the 2D-PISA method—the jet phenomenon at systole using a Nyquist limit 47 cm/s (a), measurement of the mean and maximum regurgitant velocities (b), labeling of the 2D-PISA radius at a Nyquist limit of 19 cm/s (c), the estimation of RegVolMV in this case, in which the 2D-PISA method is allowed to be used, results in a value of about 15–20 ml; the volumes LVSVtot, LVSVeff, RVSVeff, and RegVolMV have to be conclusive and plausible; exclusion of additional relevant valvular diseases (d–f); color-coded image of the left ventricular outflow tract during diastolic documents the exclusion of aortic regurgitation (AR) (d), the respective continuous wave Doppler spectrum documents no regurgitant signal (e), and a trace tricuspid regurgitation during systole is documented (f)
The quantitative approach of FMR assessment by echocardiography is challenging. The quantitative assessment of LV volumes is highly criticized because of the necessity to determine several parameters, which are all prone to measuring errors that are squared in the respective calculations [8, 9]. Nevertheless, in isolated FMR, the LVSVtot is determined by LV planimetry using the monoplane, biplane, triplane or 3D approach. The LVSVeff is measured by Doppler calculations of forward stroke volume using cross sectional area of the LVOT and the PW-Doppler velocity time integral (VTI) of the LVOT. In patients with combined aortic valve disease the LVSVeff can be determined by Doppler calculations of SV using cross sectional area the right ventricular outflow tract (RVOT) and the respective PW-Doppler VTI of the RVOT which is not common use in clinical routine echocardiography. As illustrated LV volumes and RegVolMV can be correctly calculated by the differences of LVSVtot and LVSVeff or RVSVeff, and in few cases, in which the 2D-PISA method can adequately be used, the calculated RegVolMV corresponds to the RegVolMV determined by the 2D-PISA method (Figs. 6, 7, 8, 9). If all parameters can be assessed, a cross-check can be well performed with respect to plausible hemodynamics [37]. To provide a precise echocardiographic characterization of FMR severity the presented quantitative TTE approach might be additionally added to the “up to now integrated approach” to provide a more reliable and more consistent characterization of the FMR severity.
Summary
-
(1)
The inconsistencies of the echocardiographic characterization of FMR severity make interpretations about FMR characteristics or generation of algorithms based on the trial results difficult.
-
(2)
The term “disproportionate FMR” is not in line with the physical laws of conservation of mass and energy and can only be explained by inconsistent echocardiographic data. Thus, the term is confusing and should therefore be avoided because a “disproportionate FMR” with inconsistent hemodynamics is not possible anyway.
-
(3)
A quantitative approach of FMR grading that includes the accurate quantitative assessment of LVSVtot, LVSVeff, RegVolMV and individual RF by echocardiography should be discussed in future recommendations.
References
Obadia JF, Messika-Zeitoun D, Leurent G, Iung B, Bonnet G, Piriou N, Lefèvre T, Piot C, Rouleau F, Carrié D, Nejjari M, Ohlmann P, Leclercq F, Saint Etienne C, Teiger E, Leroux L, Karam N, Michel N, Gilard M, Donal E, Trochu JN, Cormier B, Armoiry X, Boutitie F, Maucort-Boulch D, Barnel C, Samson G, Guerin P, Vahanian A, Mewton N, MITRA-FR Investigators (2018) Percutaneous repair or medical treatment for secondary mitral regurgitation. N Engl J Med 379:2297–2306
Stone GW, Lindenfeld J, Abraham WT, Kar S, Lim DS, Mishell JM, Whisenant B, Grayburn PA, Rinaldi M, Kapadia SR, Rajagopal V, Sarembock IJ, Brieke A, Marx SO, Cohen DJ, Weissman NJ, Mack MJ, COAPT Investigators (2018) Transcatheter mitral-valve repair in patients with heart failure. N Engl J Med 379:2307–2318. https://doi.org/10.1056/nejmoa1806640
Klaus K, Witte KK, Lipiecki J, Siminiak T, Meredith JT, Malkin CJ, Goldberg SL, Stark MA, von Bardeleben RS, Cremer PC, Jaber WA, Celermajer DS, Kaye DM, Sievert H (2019) The REDUCE FMR Trial: a randomized sham-controlled study of percutaneous mitral annuloplasty in functional mitral regurgitation. JACC Heart Fail 7:945–955. https://doi.org/10.1016/j.jchf.2019.06.011
Crestanello JA, Oh JK, Schaff HV (2019) Transcatheter mitral-valve repair in patients with heart failure. N Engl J Med 380:1978. https://doi.org/10.1056/NEJMc1903982
Doenst T, Bargenda S, Kirov H, Moschovas A, Tkebuchava S, Safarov R, Diab M, Faerber G (2019) Cardiac surgery 2018 reviewed. Clin Res Cardiol 108:974–989. https://doi.org/10.1007/s00392-019-01470-6
Hahn RT (2019) Disproportionate emphasis on proportionate mitral regurgitation—are there better measures of regurgitant severity? JAMA Cardiol. https://doi.org/10.1001/jamacardio.2019.6235
Hagendorff A, Doenst T, Falk V (2019) Echocardiographic assessment of functional mitral regurgitation: opening Pandora’s box? ESC Heart Fail 6:678–685. https://doi.org/10.1002/ehf2.12491
Lancellotti P, Tribouilloy C, Hagendorff A, Popescu BA, Edvardsen T, Pierard LA, Badano L, Zamorano JL, Scientific Document Committee of the European Association of Cardiovascular Imaging (2013) Recommendations for the echocardiographic assessment of native valvular regurgitation: an executive summary from the European Association of Cardiovascular Imaging. Eur Heart J Cardiovasc Imaging 14:611–644
Zoghbi WA, Adams D, Bonow RO, Enriquez-Sarano M, Foster E, Grayburn PA, Hahn RT, Han Y, Hung J, Lang RM, Little SH, Shah DJ, Shernan S, Thavendiranathan P, Thomas JD, Weissman NJ (2017) Recommendations for noninvasive evaluation of native valvular regurgitation: a report from the American Society of Echocardiography developed in collaboration with the Society for Cardiovascular Magnetic Resonance. J Am Soc Echocardiogr 30:303–371
Asch AF, Grayburn PA, Siegel RJ, Kar S, Lim DS, Zaroff JG, Mishell JM, Whisenant B, Mack MJ, JoAnn Lindenfeld JA, Abraham WT, Stone GW, Weissman NJ, on behalf of the COAPT Investigators (2019) Echocardiographic outcomes after transcatheter leaflet approximation in patients with secondary mitral regurgitation: the COAPT trial. J Am Coll Cardiol 74:2969–2979. https://doi.org/10.1016/j.jacc.2019.09.017
Gaasch WH, Aurigemma GP, Meyer TE (2020) An appraisal of the association of clinical outcomes with the severity of regurgitant volume relative to end-diastolic volume in patients with secondary mitral regurgitation. JAMA Cardiol. https://doi.org/10.1001/jamacardio.2019.5980
Grayburn PA, Sannino A, Packer M (2019) Proportionate and disproportionate functional mitral regurgitation: a new conceptual framework that reconciles the results of the MITRA-FR and COAPT trials. JACC Cardiovasc Imaging 12:353–362
Arora G, Patel N, Arora P (2018) Futile MITRA-FR and a positive COAPT trial: where does the evidence leave the clinicians? Int J Cardiol Heart Vasc 22:18–19
Bartko PE, Arfsten H, Heitzinger G, Pavo N, Toma A, Strunk G, Hengstenberg C, Hülsmann M, Goliasch G (2019) A unifying concept for the quantitative assessment of secondary mitral regurgitation. J Am Coll Cardiol 73:2506–2517. https://doi.org/10.1016/j.jacc.2019.02.075
Praz F, Grasso C, Taramasso M, Baumbach A, Piazza N, Tamburino C, Windecker S, Maisano F, Prendergast B (2019) Mitral regurgitation in heart failure: time for a rethink. Eur Heart J 40:2189–2193. https://doi.org/10.1093/eurheartj/ehz222
Drake DH, Zimmerman KG, Sidebotham DA (2019) Transcatheter mitral-valve repair in patients with heart failure. N Engl J Med 380:1978–1979. https://doi.org/10.1056/NEJMc1903982
Garbi M, Lancelotti P (2019) Transcatheter mitral-valve repair in patients with heart failure. N Engl J Med 380:1979. https://doi.org/10.1056/NEJMc1903982
Kalavrouziotis D, Voisine P, Mohammadi S (2019) Transcatheter mitral-valve repair in patients with heart failure. N Engl J Med 380:1979–1980. https://doi.org/10.1056/NEJMc1903982
Packer M, Grayburn PA (2019) Contrasting effects of pharmacological, procedural, and surgical interventions on proportionate and disproportionate functional mitral regurgitation in chronic heart failure. Circulation 140:779–789. https://doi.org/10.1161/CIRCULATIONAHA.119.039612
Packer M, Grayburn PA (2020) New evidence supporting a novel conceptual framework for distinguishing proportionate and disproportionate functional mitral regurgitation. JAMA Cardiol. https://doi.org/10.1001/jamacardio.2019.5971
Pibarot P, Delgado V, Bax JJ (2019) MITRA-FR vs. COAPT: lessons from two trials with diametrically opposed results. Eur Heart J Cardiovasc Imaging 20:620–624. https://doi.org/10.1093/ehjci/jez073
Senni M, Adamo M, Metra M, Alfieri O, Vahanian A (2019) Treatment of functional mitral regurgitation in chronic heart failure: can we get a ‘proof of concept’ from the MITRA-FR and COAPT trials? Eur J Heart Fail 21:852–861. https://doi.org/10.1002/ejhf.1491
Grayburn PA, Carabello B, Hung J, Gillam LD, Liang D, Mack MJ, McCarthy PM, Miller DC, Trento A, Siegel RJ (2014) Defining “severe” secondary mitral regurgitation: emphasizing an integrated approach. J Am Coll Cardiol 64:2792–2801. https://doi.org/10.1016/j.jacc.2014.10.016
Baumgartner H, Hung J, Bermejo J, Chambers JB, Edvardsen T, Goldstein S, Lancellotti P, LeFevre M, Miller F, Otto CM (2017) Recommendations on the echocardiographic assessment of aortic valve stenosis: a focused update from the European Association of Cardiovascular Imaging and the American Society of Echocardiography. Eur Heart J Cardiovasc Imaging 18:254–275. https://doi.org/10.1093/ehjci/jew335
Hagendorff A, Knebel F, Helfen A, Knierim J, Sinning C, Stöbe S, Fehske W, Ewen S (2020) Expert consensus document on the assessment of the severity of aortic valve stenosis by echocardiography to provide diagnostic conclusiveness by standardized verifiable documentation. Clin Res Cardiol 109:271–288. https://doi.org/10.1007/s00392-019-01539-2
Baumgartner H, Schima H, Kuhn P (1991) Value and limitations of proximal jet dimensions for the quantitation of valvular regurgitation: an in vitro study using Doppler flow imaging. J Am Soc Echocardiogr 4:57–66
Hall SA, Brickner ME, Willett DL, Irani WN, Afridi I, Grayburn PA (1997) Assessment of mitral regurgitation severity by Doppler color flow mapping of the vena contracta. Circulation 95:636–642
Bargiggia GS, Tronconi L, Sahn DJ, Recusani F, Raisaro A, De Servi S, Valdes-Cruz LM, Montemartini C (1991) A new method for quantitation of mitral regurgitation based on color flow Doppler imaging of flow convergence proximal to regurgitant orifice. Circulation 84:1481–1489
Enriquez-Sarano M, Dujardin KS, Tribouilloy CM, Seward JB, Yoganathan AP, Bailey KR, A J Tajik AJ. (1999) Determinants of pulmonary venous flow reversal in mitral regurgitation and its usefulness in determining the severity of regurgitation. Am J Cardiol 83:535–541
Tribouilloy C, Shen WF, Rey JL, Adam MC, Lesbre JP (1994) Mitral to aortic velocity–time integral ratio. A non-geometric pulsed-Doppler regurgitant index in isolated pure mitral regurgitation. Eur Heart J 15:1335–1339
Thavendiranathan P, Phelan D, Thomas JD, Flamm SD, Marwick TH (2012) Quantitative assessment of mitral regurgitation: validation of new methods. J Am Coll Cardiol 60:1470–1483. https://doi.org/10.1016/j.jacc.2012.05.048
Uretsky S, Gillam L, Lang R, Chaudhry FA, Argulian E, Supariwala A, Gurram S, Jain K, Subero M, Jang JJ, Cohen R, Wolff SD (2015) Discordance between echocardiography and MRI in the assessment of mitral regurgitation severity: a prospective multicenter trial. J Am Coll Cardiol 65:1078–1088. https://doi.org/10.1016/j.jacc.2014.12.047
Uretsky S, Argulian E, Narula J, Wolff SD (2018) Use of cardiac magnetic resonance imaging in assessing mitral regurgitation: current evidence. J Am Coll Cardiol 71:547–563. https://doi.org/10.1016/j.jacc.2017.12.009
Baumgartner H, Falk V, Bax JJ, De Bonis M, Hamm C, Holm PJ, Iung B, Lancellotti P, Lansac E, Muñoz DR, Rosenhek R, Sjögren J, Mas PT, Vahanian A, Walther T, Wendler O, Windecker S, Zamorano JL, ESC Scientific Document Group (2017) 2017 ESC/EACTS Guidelines for the management of valvular heart disease. Eur Heart J 38:2739–2791. https://doi.org/10.1093/eurheartj/ehx391
Kizilbash AM, Willett DL, Brickner ME, Heinle SK, Grayburn PA (1998) Effects of afterload reduction on vena contracta width in mitral regurgitation. J Am Coll Cardiol 32:427–431
Borger MA, Alam A, Murphy PM, Doenst T, David TE (2006) Chronic ischemic mitral regurgitation: repair, replace or rethink? Ann Thorac Surg 81:1153–1161. https://doi.org/10.1016/j.athoracsur.2005.08.080
Stöbe S, Kreyer K, Jurisch D, Pfeiffer D, Lavall D, Farese G, Laufs U, Hagendorff A (2020) Echocardiographic analysis of acute effects of percutaneous mitral annuloplasty on severity of secondary mitral regurgitation. ESC Heart Fail. https://doi.org/10.1002/ehf2.12719
Funding
Open access funding provided by Projekt DEAL.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Hagendorff Disproportionate FMR—Another Myth?
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hagendorff, A., Knebel, F., Helfen, A. et al. Disproportionate mitral regurgitation: another myth? A critical appraisal of echocardiographic assessment of functional mitral regurgitation. Int J Cardiovasc Imaging 37, 183–196 (2021). https://doi.org/10.1007/s10554-020-01975-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-020-01975-6