Abstract
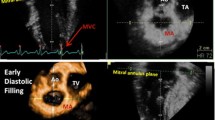
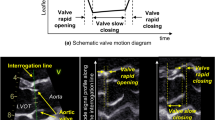
Cardiac valve plane displacement (CVPD) reflects longitudinal LV function. The purpose of the present study was to determine regional heterogeneity of CVPD in healthy adults to provide normal values by cardiac magnetic resonance (CMR). We measured the anterior aortic plane systolic excursion (AAPSE); the anterior, anterolateral, inferolateral, inferior, and inferoseptal mitral annular plane systolic excursion (MAPSE); and the lateral tricuspid annulus plane systolic excursion (TAPSE). Systolic excursion was measured as the distance from peak end-diastolic to peak end-sysstolic annular position (peak-to-peak) in cine images acquired in 2-, 3- and 4-chamber views. Echocardiographic measurements of CVPD were performed in M-Mode as previously described. We retrospectively analyzed 209 healthy Caucasians (57% men), who participated in the Heidelberg normal cohort between March 2009 and September 2014. The analysis was possible in all participants. Mean values were: AAPSE = 14 ± 3 mm (8–20); MAPSEanterior = 14 ± 3 mm (8–20); MAPSEanterolateral = 16 ± 3 mm (10–22); MAPSEinferolateral = 16 ± 3 mm (10–22); MAPSEinferior = 17 ± 3 mm (11–23); MAPSEinferoseptal = 13 ± 3 mm (7–19) and TAPSE = 26 ± 4 mm (18–34) respectively. MAPSE was significantly elevated in lateral compared to septal regions (p = 0.0001). Sex-differences for CVPD were not found. Age-dependency of CVPD revealed distinct regional differences. AAPSE decreased the most with age (B=−0.48; p = 0.0001), whereas MAPSEinferior was the least age-dependent site (B=−0.17; p = 0.01). AAPSE revealed favorable intra-/interobserver reproducibility and interstudy agreement. Intermethod-comparison of CMR and M-Mode echocardiography showed good agreement between both measurements of CVPD. Age-stratified normal values of regional CVPD are provided. AAPSE revealed the most pronounced age-related decrease and provided favorable reproducibility compared to other regions of cardiac valve plane.



Similar content being viewed by others
References
Greenbaum RA, Ho SY, Gibson DG et al (1981) Left ventricular fibre architecture in man. Br Heart J 45:248–263. doi:10.1136/hrt.45.3.248
Torrent-Guasp F, Kocica MJ, Corno AF et al (2005) Towards new understanding of the heart structure and function. Eur J Cardiothorac Surg 27:191–201. doi:10.1016/j.ejcts.2004.11.026
Carlsson M, Ugander M, Mosén H et al (2007) Atrioventricular plane displacement is the major contributor to left ventricular pumping in healthy adults, athletes, and patients with dilated cardiomyopathy. AJP 292:H1452–H1459. doi:10.1152/ajpheart.01148.2006
Matos J, Kronzon I, Panagopoulos G, Perk G (2012) Mitral annular plane systolic excursion as a surrogate for left ventricular ejection fraction. J Am Soc Echocardiogr 25:969–974. doi:10.1016/j.echo.2012.06.011
Elnoamany MF, Abdelhameed AK (2006) Mitral annular motion as a surrogate for left ventricular function: correlation with brain natriuretic peptide levels. Eur J Echocardiogr 7:187–198. doi:10.1016/j.euje.2005.05.005
Pai RG, Bodenheimer MM, Pai SM et al (1991) Usefulness of systolic excursion of the mitral anulus as an index of left ventricular systolic function. Am J Cardiol 67:222–224. doi:10.1016/0002-9149(91)90453-R
Ochs MM, Riffel J, Kristen AV et al (2016) Anterior aortic plane systolic excursion: a novel indicator of transplant-free survival in systemic light-chain amyloidosis. J Am Soc Echocardiogr 29:1188–1196. doi:10.1016/j.echo.2016.09.003
Brand B, Rydberg E, Ericsson G et al (2002) Prognostication and risk stratification by assessment of left atrioventricular plane displacement in patients with myocardial infarction. Int J Cardiol 83:35–41. doi:10.1016/S0167-5273(02)00007-4
Sveälv BG, Olofsson EL, Andersson B (2008) Ventricular long-axis function is of major importance for long-term survival in patients with heart failure. Heart 94:284–289. doi:10.1136/hrt.2006.106294
Hu K, Liu D, Herrmann S et al (2013) Clinical implication of mitral annular plane systolic excursion for patients with cardiovascular disease. Eur Heart J Cardiovasc Imaging 14:205–212. doi:10.1093/ehjci/jes240
Saba SG, Chung S, Bhagavatula S et al (2014) A novel and practical cardiovascular magnetic resonance method to quantify mitral annular excursion and recoil applied to hypertrophic cardiomyopathy. J Cardiovasc Magn Reson 16:35. doi:10.1186/1532-429X-16-35
Maceira AM, Prasad SK, Khan M, Pennell DJ (2009) Normalized left ventricular systolic and diastolic function by steady state free precession cardiovascular magnetic resonance. J Cardiovasc Magn Reson. doi:10.1080/10976640600572889;page:string:Article/Chapter
Garg P, Kidambi A, Ripley DP et al (2015) Mitral annular plane systolic excursion and intra-myocardial haemorrhage in acute myocardial infarction. J Cardiovasc Magn Reson 17:1. doi:10.1186/1532-429X-17-S1-P163
Maffessanti F, Gripari P, Pontone G et al (2013) Three-dimensional dynamic assessment of tricuspid and mitral annuli using cardiovascular magnetic resonance. Eur Heart J Cardiovasc Imaging 14:986–995. doi:10.1093/ehjci/jet004
Bulluck H, Ngamkasem H, Sado D et al (2014) A simple technique to measure TAPSE and MAPSE on CMR and normal values. J Cardiovasc Magn Reson 16:1. doi:10.1186/1532-429X-16-S1-P22
Abdel-Aty H, Katus HA, Lehrke S, Steen H (2012) CMR derived MAPSE and TAPSE Measurements in hypertrophic cardiomyopathy: comparison to healthy volunteers. J Cardiovasc Magn Reson 14:1. doi:10.1186/1532-429X-14-S1-P168
Graham I, Atar D, Borch-Johnsen K et al (2007) European guidelines on cardiovascular disease prevention in clinical practice: executive summary. Eur Heart J 28:2375–2414. doi:10.1093/eurheartj/ehm316
Andre F, Steen H, Matheis P et al (2015) Age- and gender-related normal left ventricular deformation assessed by cardiovascular magnetic resonance feature tracking. J Cardiovasc Magn Reson 17:1. doi:10.1186/s12968-015-0123-3
Mattace-Raso FUS, van der Cammen TJM, Hofman A et al (2006) Arterial stiffness and risk of coronary heart disease and stroke. Circulation 113:657–663. doi:10.1161/CIRCULATIONAHA.105.555235
Najjar SS, Scuteri A, Shetty V et al (2008) Pulse wave velocity is an independent predictor of the longitudinal increase in systolic blood pressure and of incident hypertension in the baltimore longitudinal study of aging. J Am Coll Cardiol 51:1377–1383. doi:10.1016/j.jacc.2007.10.065
Mitchell GF, Hwang SJ, Vasan RS et al (2010) Arterial stiffness and cardiovascular events: the Framingham heart study. Circulation 121:505–511. doi:10.1161/CIRCULATIONAHA.109.886655
Laurent S, Boutouyrie P, Asmar R et al (2001) Aortic stiffness is an independent predictor of all-cause and cardiovascular mortality in hypertensive patients. Hypertension 37:1236–1241
Laurent S, Katsahian S, Fassot C et al (2003) Aortic stiffness is an independent predictor of fatal stroke in essential hypertension. Stroke 34:1203–1206. doi:10.1161/01.STR.0000065428.03209.64
Vlachopoulos C, Aznaouridis K, Stefanadis C (2010) Prediction of cardiovascular events and all-cause mortality with arterial stiffness: a systematic review and meta-analysis. J Am Coll Cardiol 55:1318–1327. doi:10.1016/j.jacc.2009.10.061
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical statement
For this type of study formal consent is not required.
Rights and permissions
About this article
Cite this article
Ochs, M.M., Fritz, T., André, F. et al. A comprehensive analysis of cardiac valve plane displacement in healthy adults: age-stratified normal values by cardiac magnetic resonance. Int J Cardiovasc Imaging 33, 721–729 (2017). https://doi.org/10.1007/s10554-016-1058-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-016-1058-y