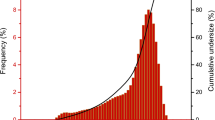
The behavior of cementite (Fe3C) in aqueous environments was investigated in the thermobaric range of 180–950°C and 2–6 GPa. When interacting with water, cementite was transformed into wüstite and magnetite. The gaseous reaction products were represented mainly by saturated hydrocarbons with linear and branched structures up to C7. The composition of the hydrocarbon products synthesized from cementite and water at extreme thermobaric parameters varied from light mixtures similar to «dry» natural gas to complex hydrocarbon systems similar to «wet» natural gas and gas condensate. During the investigation, it was discovered that the chemical reaction between iron carbide and water begins at 220°C under extreme pressure, which is significantly lower than the temperature at which the reaction of cementite with water begins at ambient pressure.



Similar content being viewed by others
References
C. Prescher, L. Dubrovinsky, E. Bykova, et al., Nature Geosci., 8(3), 220-223 (2015).
B. J. Wood, Earth and Planetary Science Letters, 117(3-4), 593-607 (1993).
F. V. Kaminsky, The Canadian Mineralogist, 49(2), 555-572 (2011).
D. E. Jacob, A. Kronz, and K. S. Viljoen, Contributions to Mineralogy and Petrology, 146(5), 566-576 (2004).
A. Y. Serovaiskii, A. Y. Kolesnikov, and V. G. Kutcherov, Geochemistry International, 57(9), 1008-1014 (2019).
A. Serovaiskii, E. Mukhina, L. Dubrovinsky, et al., Minerals, 9(11), 355 (2019).
R. Dasgupta and M. M. Hirschmann, Earth and Planetary Science Letters, 298(1), 1-13 (2010).
V. Kutcherov, K. Ivanov, E. Mukhina, et al., Carbon in Earth’s Interior, 329-339 (2020).
V. G. Kutcherov, A. N. Dmitrievskii, K. S. Ivanov, et al., Doklady Rossiiskoi Akademii Nauk, 492(1), 61-65 (2020).
Y. T. Zhou, X. H. Shao, S. J. Zheng, et al., Journal of Materials Science & Technology, 101, 28-36 (2022).
A. F. Baye, D.-H. Han, S. K. Kassahun, et al., Electrochimica Acta, 398, 139343 (2021).
W. Yang, X. Liu, X. Yue, et al., Journal of the American Chemical Society, 137(4), 1436-1439 (2015).
J. Ågren, Metallurgical Transactions A, 10(12), 1847-1852 (1979).
W. Arabczyk, E. Ekiert, and R. Jędrzejewski, The Journal of Physical Chemistry A, 113(17), 4947-4953 (2009).
L. Xiao, Z. Wen, S. Ci, et al., Nano Energy, 1(5), 751-756 (2012).
T. Akiyama, A. Miyazaki, H. Nakanishi, et al., International Journal of Hydrogen Energy, 29(7), 721-724 (2004).
M. Hisa, A. Tsutsumi, and T. Akiyama, Materials Transactions, 45(6), 1911-1914 (2004).
S. Takahashi, E. Ohtani, T. Sakai, et al., Carbon in Earth’s Interior, 25-26 (2020).
S. Ono and K. Mibe, Physics of the Earth and Planetary Interiors, 180(1), 1-6 (2010).
A. Serovaiskii and V. Kutcherov, Scientific Reports, 10(1), 4559 (2020).
A. Y. Serovaiskii, V. G. Kutcherov, V. A. Vinokurov, et al., Crystallography Reports, 66(6), 1060-1065 (2021).
A. Serovaiskii and V. Kutcherov, Geosciences, 11(4), 163 (2021).
H. P. Scott, R. J. Hemley, H.-K. Mao, et al., Proceedings of the National Academy of Sciences of the United States of America, 101(39), 14023-14026 (2004).
V. M. Sonin, T. A. Bul’bak, E. I. Zhumilev, et al., Doklady Rossiiskoi Akademii Nauk, 454(1), 84-88 (2014).
F. Domine, Organic Geochemistry, 17(5), 619-634 (1991).
J. F. Kenney, V. A. Kutcherov, N. A. Bendeliani, et al., Proceedings of the National Academy of Sciences of the United States of America, 99, 10976-10981 (2002).
H. Khurshid, W. Li, S. Chandra, M.-H. Phan, et al., Nanoscale, 5(17), 7942-7952 (2013).
D. J. Frost and C. A. McCammon, Annual Review of Earth and Planetary Sciences, 36(1), 389-420 (2008).
R. J. Arculus, Annual Review of Earth and Planetary Sciences, 13(1), 75-95 (1985).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya i Tekhnologiya Topliv i Masel, No. 4, pp. 39–42 July –August, 2022.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Serovaiskii, A.Y., Kutcherov, V.G. The Stability of Cementite in the Presence of Water at Extreme Temperatures and Pressures. Chem Technol Fuels Oils 58, 615–619 (2022). https://doi.org/10.1007/s10553-022-01427-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10553-022-01427-y