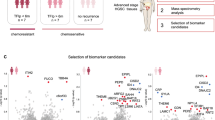
Abstract
Background
Therapeutic response predictors like age, nodal status, and tumor grade and markers, like ER/PR, HER2, and Ki67, are not reliable in predicting the response to a specific form of chemotherapy. The current study aims to identify and validate reliable markers that can predict pathological complete response (pCR) in fluorouracil, epirubicin, and cyclophosphamide (FEC)-based neoadjuvant therapy with (NACT/RT) and without concurrent radiation (NACT).
Materials and methods
Tandem mass tag (TMT) quantitative liquid chromatography-tandem mass spectrometry (LC–MS/MS) was used to identify differentially expressed proteins from core needle breast biopsy between pCR (n = 4) and no-pCR (n = 4). Immunoblotting of shortlisted proteins with the tissue lysates confirmed the differential expression of the markers. Further, immunohistochemistry (IHC) was performed on formalin-fixed paraffin-embedded sections of treatment-naive core needle biopsies. In the NACT, 29 pCR and 130 no-pCR and in NACT/RT, 32 pCR and 71 no-pCR were used.
Results
733 and 807 proteins were identified in NACT and NACT/RT groups, respectively. Ten proteins were shortlisted for validation as potential pCR-predictive markers. THBS1, TNC, and DCN were significantly overexpressed in no-pCR in both the groups. In NACT, CPA3 was significantly upregulated in the no-pCR. In NACT/RT, HnRNPAB was significantly upregulated and HMGB1 significantly downregulated in the no-pCR. HMGB1 was the only marker to show prognostic significance.
Conclusion
Quantitative proteomics followed by IHC identified and validated potential biomarkers for predicting patient response to therapy. These markers can be used, following larger-scale validation, in combination with routine histological analysis providing vital indications of treatment response.








Similar content being viewed by others
Data availability
The authors confirm that the data supporting the findings of this study are available within the article [and/or] its supplementary materials.
References
Gupta S, Rai R (2020) Breast cancer:—risk factors, incidence, mortality and social functioning in India. Int J Biotechnol Biomed Sci 6:1–4
Giordano SH (2003) Update on locally advanced breast cancer. Oncologist 8:521–530. https://doi.org/10.1634/theoncologist.8-6-521
Esteva FJ, Yu D, Hung MC, Hortobagyi GN (2010) Molecular predictors of response to trastuzumab and lapatinib in breast cancer. Nat Rev Clin Oncol 7:98–107
Das U, Lakshmaiah KC, Govind Babu K et al (2014) The actual scenario of neoadjuvant chemotherapy of breast cancer in developing country: a report of 80 cases of breast cancer from a tertiary cancer center in India. J Cancer Res Clin Oncol 140:1777–1782. https://doi.org/10.1007/s00432-014-1724-1
Londero V, Bazzocchi M, del Frate C et al (2004) Locally advanced breast cancer: comparison of mammography, sonography and MR imaging in evaluation of residual disease in women receiving neoadjuvant chemotherapy. Eur Radiol 14:1371–1379. https://doi.org/10.1007/s00330-004-2246-z
Martín M, Rodríguez-Lescure Á, Ruiz A et al (2008) Randomized phase 3 trial of fluorouracil, epirubicin, and cyclophosphamide alone or followed by paclitaxel for early breast cancer. J Natl Cancer Inst 100:805–814. https://doi.org/10.1093/jnci/djn151
Baumann S, Ceglarek U, Fiedler GM et al (2005) Standardized approach to proteome profiling of human serum based on magnetic bead separation and matrix-assisted laser desorption/ionization time-of-flight mass spectrometry. Clin Chem 51:973–980. https://doi.org/10.1373/clinchem.2004.047308
Tewari M, Krishnamurthy A, Shukla HS (2008) Predictive markers of response to neoadjuvant chemotherapy in breast cancer. Surg Oncol 17:301–311. https://doi.org/10.1016/j.suronc.2008.03.003
Duffy MJ (2013) Tumor markers in clinical practice: A review focusing on common solid cancers. Med Princ Pract 22:4–11. https://doi.org/10.1159/000338393
Devita V, Lawren T, Rosenberg S (2015) Cancer: Principles & practice of oncology, 10th edn. Wolters Kluwer Health Adis (EPS), Riverwoods
Aydiner A, İgci A, Soran A (2019) Breast Disease. Springer International Publishing, Cham
Duffy MJ, Harbeck N, Nap M et al (2017) Clinical use of biomarkers in breast cancer: Updated guidelines from the European Group on Tumor Markers (EGTM). Eur J Cancer 75:284–298. https://doi.org/10.1016/j.ejca.2017.01.017
Liao Y, Wang J, Jaehnig EJ et al (2019) WebGestalt 2019: gene set analysis toolkit with revamped UIs and APIs. Nucleic Acids Res 47:W199–W205. https://doi.org/10.1093/nar/gkz401
Wang J, Vasaikar S, Shi Z et al (2017) WebGestalt 2017: a more comprehensive, powerful, flexible and interactive gene set enrichment analysis toolkit. Nucleic Acids Res 45:W130–W137. https://doi.org/10.1093/nar/gkx356
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9:671–675. https://doi.org/10.1038/nmeth.2089
Pu RT, Schott AF, Sturtz DE et al (2005) Pathologic features of breast cancer associated with complete response to neoadjuvant chemotherapy: importance of tumor necrosis. Am J Surg Pathol 29:354–358. https://doi.org/10.1097/01.pas.0000152138.89395.fb
Sasanpour P, Sandoughdaran S, Mosavi-Jarrahi A, Malekzadeh M (2018) Predictors of pathological complete response to neoadjuvant chemotherapy in Iranian breast cancer patients. Asian Pac J Cancer Prev 19:2423–2427. https://doi.org/10.22034/APJCP.2018.19.9.2423
Hayashi Y, Takei H, Nozu S et al (2012) Analysis of complete response by MRI following neoadjuvant chemotherapy predicts pathological tumor responses differently for molecular subtypes of breast cancer. Oncol Lett 5:83–89. https://doi.org/10.3892/ol.2012.1004
Zhao Y, Dong X, Li R et al (2015) Evaluation of the pathological response and prognosis following neoadjuvant chemotherapy in molecular subtypes of breast cancer. Onco Targets Ther 8:1511–1521. https://doi.org/10.2147/OTT.S83243
Llombart-Cussac A, Cortés J, Paré L et al (2017) HER2-enriched subtype as a predictor of pathological complete response following trastuzumab and lapatinib without chemotherapy in early-stage HER2-positive breast cancer (PAMELA): an open-label, single-group, multicentre, phase 2 trial. Lancet Oncol 18:545–554. https://doi.org/10.1016/S1470-2045(17)30021-9
Dhanushkodi M, Sridevi V, Shanta V et al (2021) Locally advanced breast cancer (LABC): real-world outcome of patients from cancer institute, Chennai. JCO Glob Oncol 7:767–781. https://doi.org/10.1200/GO.21.00001
Hortobagyi GN, Blumenschein GR, Spanos W et al (1983) Multimodal treatment of locoregionally advanced breast cancer. Cancer 51:763–768. https://doi.org/10.1002/1097-0142(19830301)51:5%3c763::AID-CNCR2820510502%3e3.0.CO;2-C
Shanta V, Swaminathan R, Rama R, Radhika R (2008) Retrospective analysis of locally advanced noninflammatory breast cancer from Chennai, South India, 1990–1999. Int J Radiat Oncol Biol Phys 70:51–58. https://doi.org/10.1016/J.IJROBP.2007.05.050
Shanta V, Krishnamurthi S (1991) Preoperative multimodal therapy for locally advanced non-inflammatory breast cancer. Clin Oncol 3:137–140. https://doi.org/10.1016/S0936-6555(05)80833-7
Shanta V, Krishnamurthi S (1976) Combined therapy in carcinomas of the female breast. J Surg Oncol 8:113–120
Sandström M, Lindman H, Nygren P et al (2006) Population analysis of the pharmacokinetics and the haematological toxicity of the fluorouracil-epirubicin-cyclophosphamide regimen in breast cancer patients. Cancer Chemother Pharmacol 58:143–156. https://doi.org/10.1007/s00280-005-0140-2
Brackstone M, Palma D, Tuck AB et al (2017) Concurrent neoadjuvant chemotherapy and radiation therapy in locally advanced breast cancer. Int J Radiat Oncol Biol Phys 99:769–776. https://doi.org/10.1016/J.IJROBP.2017.06.005
Levine MN, Bramwell VH, Pritchard KI et al (1998) Randomized trial of intensive cyclophosphamide, epirubicin, and fluorouracil chemotherapy compared with cyclophosphamide, methotrexate, and fluorouracil in premenopausal women with node-positive breast cancer. J Clin Oncol 16:2651–2658. https://doi.org/10.1200/JCO.1998.16.8.2651
Lopez M, Papaldo P, di Lauro L et al (1989) 5–fluorouracil, adriamycin, cyclophosphamide (FAC) vs. 5–fluorouracil, epirubicin, cyclophosphamide (FEC) in metastatic breast cancer. Oncology 46:1–5. https://doi.org/10.1159/000226671
Yan PW, Huang XE, Jiang Y et al (2010) Clinical comparison of safety and efficacy of vinorelbine/epirubicin (NE) with fluorouracil/epirubicin/cyclophosphamide (FEC). Asian Pac J Cancer Prev 11:1115–1118
Anderson BO, Braun S, Lim S et al (2003) Early detection of breast cancer in countries with limited resources. Breast Journal. https://doi.org/10.1046/j.1524-4741.9.s2.4.x
Farmer P, Bonnefoi H, Anderle P et al (2009) A stroma-related gene signature predicts resistance to neoadjuvant chemotherapy in breast cancer. Nat Med 15:68–74. https://doi.org/10.1038/nm.1908
Wang T, Srivastava S, Hartman M et al (2016) High expression of intratumoral stromal proteins is associated with chemotherapy resistance in breast cancer. Oncotarget 7:55155–55168. https://doi.org/10.18632/oncotarget.10894
Bai Z, Zhang Z, Qu X et al (2012) Sensitization of breast cancer cells to taxol by inhibition of taxol resistance gene 1. Oncol Lett 3:135–140. https://doi.org/10.3892/ol.2011.416
Jayachandran A, Anaka M, Prithviraj P et al (2014) Thrombospondin 1 promotes an aggressive phenotype through epithelial-to-mesenchymal transition in human melanoma. Oncotarget 5:5782–5797. https://doi.org/10.18632/oncotarget.2164
Rofstad EK, Henriksen K, Galappathi K, Mathiesen B (2003) Antiangiogenic treatment with thrombospondin-1 enhances primary tumor radiation response and prevents growth of dormant pulmonary micrometastases after curative radiation therapy in human melanoma xenografts | cancer research. Can Res 63:4055–4061
Liu L, Bai Z, Ma X et al (2016) Effects of taxol resistance gene 1 expression on the chemosensitivity of SGC-7901 cells to oxaliplatin. Exp Ther Med 11:846–852. https://doi.org/10.3892/etm.2016.2994
Lin Y, Sun D, Xu Q et al (2020) miR-338-3p attenuates oxaliplatin resistance in gastric cancer cells via targeting thrombospondin-1. Arch Med Sci. https://doi.org/10.5114/aoms.2020.98039
Buraschi S, Neill T, Owens RT et al (2012) Decorin protein core affects the global gene expression profile of the tumor microenvironment in a triple-negative orthotopic breast carcinoma xenograft model. PLoS ONE. https://doi.org/10.1371/journal.pone.0045559
Boström P, Sainio A, Kakko T et al (2013) Localization of decorin gene expression in normal human breast tissue and in benign and malignant tumors of the human breast. Histochem Cell Biol 139:161–171. https://doi.org/10.1007/s00418-012-1026-0
Klintman M, Buus R, Cheang MCU et al (2016) Changes in expression of genes representing key biologic processes after neoadjuvant chemotherapy in breast cancer, and prognostic implications in residual disease. Clin Cancer Res 22:2405–2416. https://doi.org/10.1158/1078-0432.CCR-15-1488
Lanceta L, O’Neill C, Lypova N et al (2020) Transcriptomic profiling identifies differentially expressed genes in palbociclib-resistant ER+ MCF7 breast cancer cells. Genes (Basel) 11:467. https://doi.org/10.3390/genes11040467
Mavrogonatou E, Papadopoulou A, Fotopoulou A et al (2021) Down-regulation of the proteoglycan decorin fills in the tumor-promoting phenotype of ionizing radiation-induced senescent human breast stromal fibroblasts. Cancers (Basel). https://doi.org/10.3390/cancers13081987
Teicher BA, Holden SA, Ara G, Chen G (1996) Reversal of in vivo drug resistance by the transforming growth factor-beta inhibitor decorin. Cancer Chemother Pharmacol 37:601–609. https://doi.org/10.1007/s002800050435
Teicher BA, Ikebe M, Keyes SR, Herbst RS (1997) Transforming growth factor-beta 1 overexpression produces drug resistance in vivo: reversal by decorin. In Vivo 11:463–472
Helleman J, Jansen MPHM, Ruigrok-Ritstier K et al (2008) Association of an extracellular matrix gene cluster with breast cancer prognosis and endocrine therapy response. Clin Cancer Res 14:5555–5564. https://doi.org/10.1158/1078-0432.CCR-08-0555
Naik A, Al-Yahyaee A, Abdullah N et al (2018) Neuropilin-1 promotes the oncogenic Tenascin-C/integrin β3 pathway and modulates chemoresistance in breast cancer cells. BMC Cancer. https://doi.org/10.1186/s12885-018-4446-y
Li ZL, Zhang HL, Huang Y et al (2020) Autophagy deficiency promotes triple-negative breast cancer resistance to T cell-mediated cytotoxicity by blocking tenascin-C degradation. Nat Commun 11:1–19. https://doi.org/10.1038/s41467-020-17395-y
Popova OP, Bogomazova SY, Ivanov AA (2017) Role of tenascin C in triple-negative breast cancer. Arkh Patol 79:10–15. https://doi.org/10.17116/patol201779510-15
Li X, Warren S, Pelekanou V et al (2019) Immune profiling of pre- and post-treatment breast cancer tissues from the SWOG S0800 neoadjuvant trial. J Immunother Cancer 7:1–9. https://doi.org/10.1186/s40425-019-0563-7
Moriggi M, Giussani M, Torretta E et al (2018) ECM remodeling in breast cancer with different grade: contribution of 2D-DIGE proteomics. Proteomics. https://doi.org/10.1002/pmic.201800278
Teicher BA, Maehara Y, Kakeji Y et al (1997) Reversal of in vivo drug resistance by the transforming growth factor-β inhibitor decorin. Int J Cancer 71:49–58. https://doi.org/10.1002/(SICI)1097-0215(19970328)71:1%3c49::AID-IJC10%3e3.0.CO;2-4
Zarzynska JM (2014) Two Faces of TGF-Beta1 in Breast Cancer. Mediators Inflamm 2014:1–16. https://doi.org/10.1155/2014/141747
Ning X, Chen Y, Wang X et al (2016) The potential role of CacyBP / SIP in tumorigenesis. Tumour Biol. https://doi.org/10.1007/s13277-016-4871-y
Shi Y, Hu W, Yin F et al (2004) Regulation of drug sensitivity of gastric cancer cells by human calcyclin-binding protein (CacyBP). Gastric Cancer 7:160–166. https://doi.org/10.1007/s10120-004-0286-3
Fucikova J, Kralikova P, Fialova A et al (2011) Human tumor cells killed by anthracyclines induce a tumor-specific immune response. Can Res 71:4821–4833. https://doi.org/10.1158/0008-5472.CAN-11-0950
Jiao Y, Wang H, Fan S (2007) Growth suppression and radiosensitivity increase by HMGB1 in breast cancer. Acta Pharmacol Sin 28:1957–1967. https://doi.org/10.1111/j.1745-7254.2007.00669.x
An J, Luo Z, An W et al (2021) Identification of spliceosome components pivotal to breast cancer survival. RNA Biol 18:833–842. https://doi.org/10.1080/15476286.2020.1822636
Acknowledgements
We acknowledge the Department of Science, Government of India for the funding of the project (No. SERB/F/2369/2014-15, dt. 29-06-2014). We also thank the Indian Council of Medical Research for the financial support of Ms. Prarthana Gopinath, through their Senior Research Fellowship Program (5/3/8/37/ITR-F/2018-ITR dt. 07-06-2018).
Funding
We acknowledge the Department of Science, Government of India for the funding of the project (No. SERB/F/2369/2014–15, dt. 29–06-2014). We also thank the Indian Council of Medical Research for the financial support of Ms. Prarthana Gopinath, through their Senior Research Fellowship program (5/3/8/37/ITR-F/2018-ITR dt. 07–06-2018).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. PG, SV, SS, and RS: contributed to resource and sample collection. PG, GG, and TR: contributed in the methodology, data curation, formal analysis, investigation, and validation. Visualization of the histology results was performed by SS. The first draft of the manuscript was written by PG and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
The study was approved by the Institutional Ethical Committee. Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.

10549_2022_6617_MOESM2_ESM.jpg
Supplementary file2 (JPG 188 KB) Prognostic significance of the candidate markers Kaplan–Meier plots for DFS and OS for low (green) vs. high protein expression (red). p-value ≤ 0.05 was considered statistically significant. Graphs were generated using GraphPad Prism v5.
Rights and permissions
About this article
Cite this article
Gopinath, P., Veluswami, S., Gopisetty, G. et al. Identification of tumor biomarkers for pathological complete response to neoadjuvant treatment in locally advanced breast cancer. Breast Cancer Res Treat 194, 207–220 (2022). https://doi.org/10.1007/s10549-022-06617-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-022-06617-0