Abstract
Purpose
Sacituzumab govitecan (SG) is an antibody–drug conjugate composed of an anti–Trop-2 antibody coupled to SN-38 via a proprietary hydrolyzable linker. In the ASCENT study, SG improved survival versus single-agent treatment of physician’s choice (TPC) in pre-treated metastatic triple-negative breast cancer (mTNBC). Hormone/HER2 receptor changes are common, particularly at relapse/metastasis. This subanalysis assessed outcomes in patients who did/did not have TNBC at initial diagnosis, before enrollment.
Methods
TNBC diagnosis was only required at study entry. Patients with mTNBC refractory/relapsing after ≥ 2 prior chemotherapies were randomized 1:1 to receive SG or TPC. Primary endpoint was progression-free survival (PFS) in patients without brain metastases.
Results
Overall, 70/235 (30%) and 76/233 (33%) patients who received SG and TPC, respectively, did not have TNBC at initial diagnosis. Clinical benefit with SG versus TPC was observed in this subset. Median PFS was 4.6 versus 2.3 months (HR 0.48; 95% CI 0.32–0.72), median overall survival was 12.4 versus 6.7 months (HR 0.44; 95% CI 0.30–0.64), and objective response rate (ORR) was 31% versus 4%; those who also received prior CDK4/6 inhibitors had ORRs of 21% versus 5%. Efficacy and safety for patients with TNBC at initial diagnosis were generally similar to those who did not present with TNBC at initial diagnosis.
Conclusion
Patients without TNBC at initial diagnosis had improved clinical outcomes and a manageable safety profile with SG, supporting SG as a treatment option for mTNBC regardless of subtype at initial diagnosis. Subtype reassessment in advanced breast cancer allows for optimal treatment.
Clinical trial registration number NCT02574455, registered October 12, 2015.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Approximately 15% of breast cancers diagnosed each year are categorized as triple-negative [1]. This subtype is defined by its combined lack of human epidermal growth factor receptor 2 (HER2) amplification, estrogen-receptor (ER) expression, and progesterone-receptor (PR) expression [2,3,4]. Challenges in treating TNBC include its aggressive behavior and heterogeneity, and limited viable targets and effective targeted therapies [2,3,4,5]. Standard of care for pre-treated metastatic TNBC (mTNBC) remains single-agent chemotherapy, such as eribulin, and most patients receive multiple lines of therapy in the metastatic setting [6]. However, progression-free survival (PFS) and response rates to later-line therapies are low and associated with significant toxicity, underscoring the need for novel therapies [7,8,9,10].
A barrier toward optimizing clinical outcomes for breast cancer is receptor status discordance. Although the majority (88%) of patients with breast cancer are initially diagnosed with hormone receptor (Hr)-positive and/or human epidermal growth factor receptor 2 (HER2)-positive disease [11, 12], receptor status discordance from breast cancer diagnosis through relapses and disease progression occurs frequently, most commonly involving changes in Hr status [13,14,15,16,17]. One retrospective analysis of 993 intraindividual tissue samples from primary breast tumors and relapses found alterations in estrogen receptor (ER), progesterone receptor (PR), and HER2 status in 32%, 41%, and 15% of patients, respectively [15]. Positive-to-negative changes in receptor status occur more frequently than negative-to-positive changes [13, 15], with implications for clinical outcomes. Loss of ER, PR, or HER2 expression between primary and recurrent tumors is associated with poorer overall survival (OS) and post-recurrence survival compared with receptor stability between primary and recurrent tumors [14,15,16]. Recognizing the impact on prognosis, tissue confirmation of recurrent/metastatic breast cancer subtype is included in breast cancer management guidelines [6, 18]. However, definitive, evidence-based guidance on treatment decision-making in the setting of discordant receptor status is lacking.
Sacituzumab govitecan is a Trop-2–directed antibody–drug conjugate composed of a humanized anti-Trop-2 IgG1 kappa antibody coupled to SN-38, the active metabolite of the topoisomerase inhibitor irinotecan, via a proprietary, hydrolyzable linker [19,20,21]. Following SG administration, the anti–Trop-2 monoclonal antibody binds to Trop-2 expressed on the tumor cell surface, enabling SN-38 internalization and targeted delivery to tumor cells [19, 22]. Its proprietary linker allows SN-38 to be liberated in the tumor microenvironment, eliciting antitumor effects (bystander effect) without prerequisite internalization and enzymatic cleavage of SN-38 from the anti-Trop-2 antibody [19, 22, 23].
In a phase 1/2, single-arm, basket study (IMMU-132-01; NCT01631552), SG was evaluated for patients with metastatic, epithelial cancers. In this study, a cohort of 108 patients with heavily pre-treated mTNBC treated with SG reported an ORR of 33%, a clinical benefit rate (CBR) of 45%, a median PFS of 5.5 months, a median OS of 13.0 months, and a manageable safety profile [24]. These results led to accelerated approval of SG by the United States Food and Drug Administration (FDA), with full approval received based on results of the randomized phase 3 ASCENT study [25].
The phase 3 ASCENT study evaluated the efficacy and safety of SG compared with single-agent treatment of physician’s choice (TPC; eribulin, vinorelbine, gemcitabine, or capecitabine) in 529 patients with pre-treated mTNBC. Results from this trial confirmed the initial findings from the phase 1/2 study. In the primary efficacy population of 468 patients without known brain metastasis, SG significantly improved survival compared with TPC, with a median PFS of 5.6 months versus 1.7 months (hazard ratio [HR] 0.41; [95% confidence interval [CI] 0.3–0.5]; p < 0.001) and a median OS of 12.1 months versus 6.7 months (HR 0.48; 95% CI 0.38–0.59; p < 0.001) [26]. PFS and OS benefit for SG was consistently observed across all predefined subgroups, and SG demonstrated a manageable safety profile [26].
Patients in ASCENT were required to have TNBC only at study entry; therefore, ASCENT included patients who may have had an initial diagnosis of another breast cancer subtype, such as Hr/HER2-positive disease. Because the ASCENT study population is heavily pre-treated, altered receptor status over the disease course may have been common among these patients. In this exploratory subgroup analysis of data from ASCENT, we assess the clinical impact of SG in the subgroup of patients who did not have TNBC at initial diagnosis.
Patients and methods
Study design
Full details of the study design for ASCENT (NCT02574455) have been described previously [26]. Briefly, patients with pre-treated mTNBC were randomized 1:1 to receive SG (10 mg/kg on days 1 and 8 of 21-day cycles) or TPC (eribulin, vinorelbine, gemcitabine, or capecitabine) until progression, unacceptable toxicity, study withdrawal, or death. The primary endpoint was PFS by blinded independent central review (BICR) in patients without known baseline brain metastases (BMNeg) per Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1. Secondary endpoints included investigator-assessed PFS, OS, ORR (per RECIST 1.1), duration of response (DOR), and safety.
The ASCENT trial was conducted and approved by each investigational site’s institutional review board/ethics committee prior to initiation, and in accordance with the Declaration of Helsinki, International Council for Harmonisation Good Clinical Practice Guidelines, FDA Code of Federal Regulations, national and local drug and data protection laws, and other applicable regulatory requirements. All patients provided written informed consent before enrollment.
Patients
Patients had mTNBC that had progressed following ≥ 2 prior standard chemotherapy regimens (no upper limit) for unresectable, locally advanced, or metastatic disease, and included a taxane (any setting). Per protocol, patients were also eligible after only one prior regimen in the metastatic setting if their disease recurred within 12 months of completing (neo)adjuvant therapy. TNBC status at initial diagnosis was determined from patient histories; biopsies at initial diagnosis were not centrally assessed for this study. TNBC/receptor status prior to enrollment in ASCENT was determined by local assessment of most recent biopsy or other pathology specimen per American Society of Clinical Oncology/College of American Pathologists criteria [27, 28]. Negativity for ER and PR was defined as < 1% of cells expressing ER or PR by immunohistochemistry (IHC). Negativity for HER2 was defined as IHC0 or IHC1+, or if IHC2+, then fluorescence in situ hybridization negative. Collection of new tissue samples after disease metastasis was not required for ASCENT.
Statistical analysis
This post hoc subanalysis evaluated efficacy and safety outcomes for the subpopulations of patients with and without TNBC at their initial breast cancer diagnosis. Efficacy outcomes in these subgroups were assessed in BMNeg patients. Median PFS and ORR were assessed by BICR per RECIST 1.1. The Kaplan–Meier method was used to analyze median PFS and OS. Hazard ratios and 95% confidence intervals were estimated using an unstratified Cox regression model. Safety outcomes were assessed in all patients (with and without brain metastases) who received one or more doses of study treatment. Adverse events (AEs) were coded using the Medical Dictionary for Regulatory Activities v22.1, and AE severity was graded per National Cancer Institute Common Terminology Criteria v4.03. Data cutoff for this analysis was March 11, 2020.
Results
Patients
Between November 2017 and September 2019, 529 patients with TNBC were enrolled in ASCENT; 468 had no evidence of brain metastases at baseline. A total of 146 BMNeg patients did not have TNBC at initial diagnosis (70/235 [30%] patients in the SG arm and 76/233 [33%] in the TPC arm). The disposition of patients without and with TNBC at initial diagnosis in ASCENT is summarized in Online Resource 1. Demographic and baseline characteristics of patients without and with TNBC at initial diagnosis (Table 1) were generally balanced across treatment arms and comparable in patients without and with TNBC at initial diagnosis. The median age in patients without TNBC at initial diagnosis was 56 years (range 31–74) and 55 years (range 27–80) for patients in the SG and TPC arms, respectively. Patients without TNBC at initial diagnosis received a median of 5 prior anticancer regimens (defined as any treatment regimen used to treat breast cancer in any setting, including endocrine therapy and any targeted treatment), whereas patients with TNBC at initial diagnosis received a median of four prior anticancer regimens.
In patients without TNBC at initial diagnosis, 24% and 27% received prior immune checkpoint inhibitor and cyclin-dependent kinase (CDK) 4/6 inhibitor therapy in the SG arm, respectively; in the TPC arm, 30% and 29% of patients received prior immune checkpoint inhibitor and CDK4/6 inhibitor therapy, respectively (Table 1). In the SG versus TPC arms, 20% versus 17%, 3% versus 0%, and 6% versus 7% of patients received prior anti-HER2, phosphoinositide 3 kinase (PI3K) inhibitor, and poly (ADP-ribose) polymerase (PARP) inhibitor therapy, respectively. Compared with patients without TNBC at initial diagnosis, those with TNBC at initial diagnosis had similar frequencies of prior immune checkpoint, PI3K, and PARP inhibitor use, but lower frequencies of prior CDK4/6 inhibitor (2% and 1%) and anti-HER2 therapy (4% and 4%) use in both the SG and TPC arms, respectively.
In patients without TNBC at initial diagnosis, 4 patients (6%) in the SG arm remained on treatment at data cutoff, whereas no patients remained on treatment in the TPC arm. Most patients in both the SG (84%) and TPC (72%) arms discontinued due to progressive disease. In the SG arm, 3 patients each (4% each) discontinued due to AEs and physician decision. In the TPC arm, 5 (7%), 2 (3%), and 1 (1%) patient(s) discontinued due to withdrawal of consent, AEs, and death, respectively. Patients without TNBC at initial diagnosis had a median treatment duration of 5.1 months with SG and 1.2 months with TPC.
Efficacy outcomes
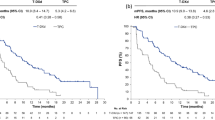
As previously reported, efficacy outcomes were consistently improved in the SG versus TPC arms for all predefined subgroups [26]. At a median follow-up of 8.2 months (range 0.0–23.0), the median PFS by BICR for patients without TNBC at initial diagnosis was 4.6 months for SG versus 2.3 months for TPC (HR 0.48; 95% CI 0.32–0.72; Fig. 1a). In this subgroup, the 12-month PFS rate for patients treated with SG versus TPC was 13% (95% CI 5.7–22.8) versus 3% (95% CI 0.2–13.2). In patients with TNBC at initial diagnosis, median PFS was 5.7 versus 1.6 months for SG versus TPC (HR 0.38; 95% CI 0.29–0.51; Fig. 1b); the 12-month PFS rate was 20% (95% CI 12.5–27.8) versus 9% (95% CI 3.8–15.9). Improvements in PFS for patients without TNBC at initial diagnosis were similar to those observed in the total population of randomized patients, who had a median PFS of 4.8 versus 1.7 months (HR 0.43; 95% CI 0.35–0.54) [26], and a 12-month PFS rate of 16% (95% CI 11.2–22.0) versus 6% (95% CI 2.7–11.2).
Kaplan–Meier estimates of progression-free survival are shown for patients without TNBC at initial diagnosis (a) and with TNBC at initial diagnosis (b). Assessments were in the brain metastases-negative population. BICR blinded independent central review; CI confidence interval; HR hazard ratio; PFS progression-free survival; SG sacituzumab govitecan; TNBC triple-negative breast cancer; TPC treatment of physician’s choice
In patients without TNBC at initial diagnosis, the median OS was 12.4 months for SG versus 6.7 months for TPC (HR 0.44; 95% CI 0.30–0.64; Fig. 2a); the 12- and 18-month OS rates were 52% (95% CI 39.3–62.9) versus 18% (9.6–27.7) and 27% (95% CI 16.6–39.1) versus 8% (95% CI 2.9–17.1), respectively. In patients with TNBC at initial diagnosis, median OS was 12.1 versus 6.9 months for SG versus TPC (HR 0.50; 95% CI 0.38–0.65; Fig. 2b); the 12- and 18-month OS rates were 50% (95% CI 42.2–57.7) versus 24% (95% CI 17.6–31.6) and 32% (95% CI 24.4–40.1) versus 15% (95% CI 8.9–21.4), respectively. OS improvements for patients without TNBC at initial diagnosis with SG versus TPC were similar to those observed in the total population of randomized patients, who had a median OS of 11.8 versus 6.9 months (HR 0.51; 95% CI 0.41–0.62) [26]; 12- and 18-month OS rates were 49% (95% CI 42.5–54.8) versus 23% (95% CI 17.8–28.5) and 29% (95% CI 22.6–34.8) versus 13% (95% CI 8.7–18.0) in the total population of randomized patients.
Kaplan–Meier estimates of overall survival are shown for patients without TNBC at initial diagnosis (a) and with TNBC at initial diagnosis (b). Assessments were in the brain metastases-negative population. CI confidence interval; HR hazard ratio; OS overall survival; SG sacituzumab govitecan; TNBC triple-negative breast cancer; TPC treatment of physician’s choice
In patients without TNBC at initial diagnosis, the ORR was 31% in the SG arm versus 4% in the TPC arm. In the SG arm, 1 patient (1%) had a complete response (CR); 21 patients (30%) had a partial response (PR; Table 2). In the TPC arm, 1 patient (1%) had a CR, and 2 patients (3%) had a PR. In the SG versus TPC arms, the CBR was 44% versus 7%, and median DOR was 5.6 versus 3.5 months, respectively. Response outcomes were similar in patients with TNBC at initial diagnosis; in the SG versus TPC arms, 9 patients (5%) versus 1 patient (1%) had a CR, and 51 patients (31%) versus 7 patients (4%) had a PR.
Among patients without TNBC at initial diagnosis and who had received a prior CDK4/6 inhibitor, patients who received SG (n = 19) had numerically higher response rates versus those who received TPC (n = 22; 21% vs. 5%; Table 3). In the SG and TPC arms, 4 patients (21%) and 1 patient (5%) had a PR as the best overall response, respectively. In the SG versus TPC arms, the CBR was 32% versus 5%.
Safety outcomes
In patients without TNBC at initial diagnosis, the most common treatment-related AEs (TRAE) of any grade for SG versus TPC were neutropenia (73% vs. 47%), diarrhea (62% vs. 12%), nausea (62% vs. 26%), alopecia (47% vs. 9%), fatigue (50% vs. 32%), and anemia (31% vs. 25%), respectively (Table 4). The most common grade ≥ 3 TRAEs in the SG versus TPC arms were neutropenia (59% vs. 40%), leukopenia (12% vs. 9%), anemia (8% vs. 7%), and diarrhea (7% vs. 0%) in patients without TNBC at initial diagnosis. Key TRAEs were generally similar for patients with TNBC at initial diagnosis. In patients without TNBC at initial diagnosis, 2 patients in each arm (each 3%) experienced grade ≥ 3 treatment-related febrile neutropenia; in those with TNBC at initial diagnosis, 13 (7%) and three (2%) patients had grade ≥ 3 treatment-related febrile neutropenia in the SG versus TPC arms, respectively. In patients without and with TNBC at initial diagnosis, treatment-related peripheral neuropathy of any grade was observed in 3 (4%) versus 9 (13%) patients and 6 (3%) versus 18 (12%) patients in the SG versus TPC arms, respectively; grade ≥ 3 peripheral neuropathy was observed in zero versus 2 (3%) patients in the group without TNBC at initial diagnosis and zero versus 2 (1%) patients in the group with TNBC at initial diagnosis, respectively. In patients without and with TNBC at initial diagnosis, 2 events (3%) versus no events and 4 events (2%) versus 1 event (1%) of grade ≤ 2 treatment-related electrocardiogram QT prolonged (by preferred term) occurred in the SG versus TPC arms, respectively; no grade ≥ 3 treatment-related events of electrocardiogram QT prolonged occurred in either treatment arm. In patients without TNBC at initial diagnosis, no events of treatment-related interstitial lung disease occurred in either arm; in those with TNBC at initial diagnosis, 1 pneumonitis event occurred (grade 3, 1%) in the SG arm that resolved after drug withdrawal.
In patients without TNBC at initial diagnosis, 16% and 25% of patients in the SG and TPC arms, respectively, had dose reductions due to TRAEs; the most common reasons for dose reduction were neutropenia (9% and 25%) and diarrhea (4% and 0%). Discontinuations due to treatment-emergent AEs were low for SG and TPC (5% and 7%, respectively), and no treatment-related deaths occurred in either arm in this subgroup. In patients with TNBC at initial diagnosis, the frequency of dose reductions due to TRAEs in the SG versus TPC arms was similar (21% vs. 22%); the most common reason for dose reduction was neutropenia (11% vs. 17%, including both neutropenia and febrile neutropenia). Discontinuations due to treatment-emergent AEs were low for both arms (4% for both) in this subgroup. One treatment-related death occurred in the TPC arm for this subgroup.
Discussion
The pivotal phase 3 randomized ASCENT trial demonstrated improvement in PFS, OS, and ORR with SG compared with TPC (eribulin, vinorelbine, gemcitabine, or capecitabine) in patients with heavily pre-treated metastatic TNBC [26]. Due to the eligibility criteria, the overall study population of ASCENT included patients without TNBC at initial diagnosis. In the current subanalysis of ASCENT, the clinical benefit of SG over TPC was confirmed in patients who did not have TNBC at initial breast cancer diagnosis; this benefit was similar to that observed for the ASCENT primary analysis population of all randomized BMNeg patients and the total ASCENT study population [26]. Key efficacy outcomes with the use of SG versus TPC for this subgroup were a median PFS of 4.6 versus 2.3 months, median OS of 12.4 versus 6.7 months, and ORR of 31% versus 4%. Responses were durable with SG versus TPC, with a median DOR of 5.6 versus 3.5 months. SG also had a manageable safety profile in patients without TNBC at initial diagnosis, which was generally similar to that of patients with TNBC at initial diagnosis and the overall study population, with key SG-related AEs being hematologic toxicities and diarrhea [26].
Approximately one-third of patients in the ASCENT trial did not have TNBC at their initial breast cancer diagnosis. This finding is consistent with previous reports documenting changes in HER2 and Hr status over the course of disease, particularly at disease relapse or metastasis [13,14,15]. Loss of Hr expression following relapse is particularly common, occurring in approximately 25–45% of patients who have relapse of their primary tumor [13, 15]. The underlying reasons for changes in receptor status between primary and recurrent lesions may include intratumoral heterogeneity, changes in tumor biology, and selective pressure from previous therapies [14, 29, 30]. In patients who received trastuzumab as part of neoadjuvant therapy for HER2-positive breast cancer and did not achieve a pathogenic CR, approximately one-third of assessable residual tumors lost HER2 amplification [29]. Similarly, loss of PD-1/PD-L1 expression from primary to metastatic tumors is frequent, and resistance to immune checkpoint inhibitors is a concern [31, 32]. These studies indicate that residual or metastatic tumors should be reassessed for biomarker status, and novel treatment strategies like SG are needed in populations with altered biomarker status.
The subset of patients without TNBC at initial diagnosis in ASCENT represent a particularly heavily pre-treated population; these patients received a median of 5 prior anticancer regimens in any treatment setting for breast cancer, including endocrine therapy and everolimus, numerically higher than the four median prior regimens observed for patients with TNBC at initial diagnosis [26]. However, the clinical benefit with SG over TPC in patients without TNBC at initial diagnosis was similar to that observed for patients with TNBC at initial diagnosis and the overall ASCENT primary analysis population [26]. Although patients without TNBC at initial diagnosis who received prior CDK4/6 inhibitors and received SG had a numerically lower ORR (21%) compared with all patients without TNBC at initial diagnosis who received SG (31%) and the overall ASCENT primary analysis population (35%) [26], the numerically higher ORR in the SG versus TPC arms (21% vs. 5%) suggests that SG may have a clinical benefit in patients without TNBC who previously received CDK4/6 inhibitors.
The results of the current analysis are similar to those of the phase 1/2 IMMU-132-01 basket trial of SG for patients with breast cancer subtypes other than TNBC [33]. Like patients in ASCENT who did not have TNBC at initial diagnosis, the 54 patients in the earlier trial with Hr-positive, HER2-negative metastatic breast cancer were heavily pre-treated, and included CDK4/6 inhibition (59%). The ORRs and CBRs seen with SG in the phase 1/2 trial were 31% and 44%, respectively, with a median PFS of 5.5 months and median OS of 12 months, in line with the results observed in this analysis [33].
This subgroup analysis had several limitations. Primarily, the ASCENT trial was not designed to assess the efficacy of SG in patients without TNBC at initial diagnosis. Further, tumor phenotyping was not performed centrally on the initial breast cancer diagnostic tissue, or on the trial-qualifying tissue. As a result, information on specific changes in receptor status prior to enrollment in ASCENT are not available, limiting our interpretation of the efficacy and safety of SG for different subtypes of breast cancer. However, 27% versus 29% and 20% versus 17% of patients without TNBC at initial diagnosis in the SG versus TPC arms received prior CDK4/6 inhibitor and anti-HER2 therapy, respectively, suggesting a substantial proportion of patients in ASCENT may have had HER2-positive or Hr-positive disease prior to TNBC diagnosis. Additionally, the limited number of patients without TNBC at initial diagnosis enrolled in the ASCENT study, particularly those who also received prior CDK4/6 inhibitor therapy (SG, n = 19; TPC, n = 22), limits interpretability of these results.
In conclusion, this subanalysis from the ASCENT study showed that SG provides clinical benefit for patients with TNBC regardless of subtype at initial diagnosis, with a manageable safety profile. With the advent of new systemic treatment options for advanced TNBC, such as SG, patients with advanced disease should be reassessed for changes in breast cancer subtype to determine the optimal treatment. The results provide evidence for further evaluating SG as a treatment option for patients with subtypes other than TNBC, including those who previously received CDK4/6 inhibitors. However, additional studies are needed to further determine the efficacy and safety profile of SG in breast cancer subtypes other than TNBC. Ongoing studies include a phase 3 trial for Hr-positive, HER2-negative metastatic breast cancer (TROPiCS-02, NCT03901339) and multiple trials evaluating SG as a single-agent or in combination with other therapies for TNBC and HER2-negative breast cancer, including in the curative setting.
Data availability
Immunomedics, Inc., a subsidiary of Gilead Sciences, Inc. will provide the study protocol and statistical analysis plan with publication of this manuscript as well as post results on clinicaltrials.gov, as required.
References
Kohler BA, Sherman RL, Howlader N, Jemal A, Ryerson AB, Henry KA et al (2015) Annual report to the nation on the status of cancer, 1975–2011, featuring incidence of breast cancer subtypes by race/ethnicity, poverty, and state. J Natl Cancer Inst 107(6):djv048. https://doi.org/10.1093/jnci/djv048
Plasilova ML, Hayse B, Killelea BK, Horowitz NR, Chagpar AB, Lannin DR (2016) Features of triple-negative breast cancer: analysis of 38,813 cases from the national cancer database. Medicine (Baltimore) 95(35):e4614. https://doi.org/10.1097/MD.0000000000004614
Newman LA, Reis-Filho JS, Morrow M, Carey LA, King TA (2015) The 2014 Society of Surgical Oncology Susan G. Komen for the cure symposium: triple-negative breast cancer. Ann Surg Oncol 22(3):874–882. https://doi.org/10.1245/s10434-014-4279-0
Lee A, Djamgoz MBA (2018) Triple negative breast cancer: emerging therapeutic modalities and novel combination therapies. Cancer Treat Rev 62:110–122. https://doi.org/10.1016/j.ctrv.2017.11.003
Anders CK, Abramson V, Tan T, Dent R (2016) The evolution of triple-negative breast cancer: from biology to novel therapeutics. Am Soc Clin Oncol Educ Book 35:34–42. https://doi.org/10.14694/EDBK_159135
Cardoso F, Paluch-Shimon S, Senkus E, Curigliano G, Aapro MS, Andre F et al (2020) 5th ESO-ESMO international consensus guidelines for advanced breast cancer (ABC 5). Ann Oncol 31(12):1623–1649. https://doi.org/10.1016/j.annonc.2020.09.010
Park IH, Im SA, Jung KH, Sohn JH, Park YH, Lee KS et al (2019) Randomized open label phase III trial of irinotecan plus capecitabine versus capecitabine monotherapy in patients with metastatic breast cancer previously treated with anthracycline and taxane: PROCEED Trial (KCSG BR 11-01). Cancer Res Treat 51(1):43–52. https://doi.org/10.4143/crt.2017.562
Perez EA, Patel T, Moreno-Aspitia A (2010) Efficacy of ixabepilone in ER/PR/HER2-negative (triple-negative) breast cancer. Breast Cancer Res Treat 121(2):261–271. https://doi.org/10.1007/s10549-010-0824-0
Pivot X, Marme F, Koenigsberg R, Guo M, Berrak E, Wolfer A (2016) Pooled analyses of eribulin in metastatic breast cancer patients with at least one prior chemotherapy. Ann Oncol 27(8):1525–1531. https://doi.org/10.1093/annonc/mdw203
Li CH, Karantza V, Aktan G, Lala M (2019) Current treatment landscape for patients with locally recurrent inoperable or metastatic triple-negative breast cancer: a systematic literature review. Breast Cancer Res 21(1):143. https://doi.org/10.1186/s13058-019-1210-4
American Cancer Society (2019) Breast cancer facts & figures 2019–2020. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/breast-cancer-facts-and-figures/breast-cancer-facts-and-figures-2019-2020.pdf. Accessed 30 Sept 2021
Howlader N, Altekruse SF, Li CI, Chen VW, Clarke CA, Ries LA et al (2014) US incidence of breast cancer subtypes defined by joint hormone receptor and HER2 status. J Natl Cancer Inst. https://doi.org/10.1093/jnci/dju055
Aurilio G, Disalvatore D, Pruneri G, Bagnardi V, Viale G, Curigliano G et al (2014) A meta-analysis of oestrogen receptor, progesterone receptor and human epidermal growth factor receptor 2 discordance between primary breast cancer and metastases. Eur J Cancer 50(2):277–289. https://doi.org/10.1016/j.ejca.2013.10.004
Dieci MV, Barbieri E, Piacentini F, Ficarra G, Bettelli S, Dominici M et al (2013) Discordance in receptor status between primary and recurrent breast cancer has a prognostic impact: a single-institution analysis. Ann Oncol 24(1):101–108. https://doi.org/10.1093/annonc/mds248
Lindstrom LS, Karlsson E, Wilking UM, Johansson U, Hartman J, Lidbrink EK et al (2012) Clinically used breast cancer markers such as estrogen receptor, progesterone receptor, and human epidermal growth factor receptor 2 are unstable throughout tumor progression. J Clin Oncol 30(21):2601–2608. https://doi.org/10.1200/JCO.2011.37.2482
Liedtke C, Broglio K, Moulder S, Hsu L, Kau SW, Symmans WF et al (2009) Prognostic impact of discordance between triple-receptor measurements in primary and recurrent breast cancer. Ann Oncol 20(12):1953–1958. https://doi.org/10.1093/annonc/mdp263
Niikura N, Liu J, Hayashi N, Mittendorf EA, Gong Y, Palla SL et al (2012) Loss of human epidermal growth factor receptor 2 (HER2) expression in metastatic sites of HER2-overexpressing primary breast tumors. J Clin Oncol 30(6):593–599. https://doi.org/10.1200/JCO.2010.33.8889
Buchholz TA, Ali S, Hunt KK (2020) Multidisciplinary management of locoregional recurrent breast cancer. J Clin Oncol 38(20):2321–2328. https://doi.org/10.1200/JCO.19.02806
Lopez S, Perrone E, Bellone S, Bonazzoli E, Zeybek B, Han C et al (2020) Preclinical activity of sacituzumab govitecan (IMMU-132) in uterine and ovarian carcinosarcomas. Oncotarget 11(5):560–570. https://doi.org/10.18632/oncotarget.27342
Goldenberg DM, Sharkey RM (2020) Sacituzumab govitecan, a novel, third-generation, antibody-drug conjugate (ADC) for cancer therapy. Expert Opin Biol Ther 20(8):871–885. https://doi.org/10.1080/14712598.2020.1757067
Govindan SV, Cardillo TM, Sharkey RM, Tat F, Gold DV, Goldenberg DM (2013) Milatuzumab-SN-38 conjugates for the treatment of CD74+ cancers. Mol Cancer Ther 12(6):968–978. https://doi.org/10.1158/1535-7163.MCT-12-1170
Goldenberg DM, Cardillo TM, Govindan SV, Rossi EA, Sharkey RM (2015) Trop-2 is a novel target for solid cancer therapy with sacituzumab govitecan (IMMU-132), an antibody-drug conjugate (ADC). Oncotarget 6(26):22496–22512. https://doi.org/10.18632/oncotarget.4318
Goldenberg DM, Sharkey RM (2019) Antibody-drug conjugates targeting TROP-2 and incorporating SN-38: a case study of anti-TROP-2 sacituzumab govitecan. mAbs 11(6):987–995. https://doi.org/10.1080/19420862.2019.1632115
Bardia A, Mayer IA, Vahdat LT, Tolaney SM, Isakoff SJ, Diamond JR et al (2019) Sacituzumab govitecan-hziy in refractory metastatic triple-negative breast cancer. N Engl J Med 380(8):741–751. https://doi.org/10.1056/NEJMoa1814213
Food and Drug Administration (2021) FDA grants regular approval to sacituzumab govitecan for triple-negative breast cancer. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-regular-approval-sacituzumab-govitecan-triple-negative-breast-cancer. Accessed 28 Sept 2021
Bardia A, Hurvitz SA, Tolaney SM, Loirat D, Punie K, Oliveira M et al (2021) Sacituzumab govitecan in metastatic triple-negative breast cancer. N Engl J Med 384(16):1529–1541. https://doi.org/10.1056/NEJMoa2028485
Hammond ME, Hayes DF, Dowsett M, Allred DC, Hagerty KL, Badve S et al (2010) American Society of Clinical Oncology/College of American Pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. J Clin Oncol 28(16):2784–2795. https://doi.org/10.1200/JCO.2009.25.6529
Wolff AC, Hammond MEH, Allison KH, Harvey BE, Mangu PB, Bartlett JMS et al (2018) Human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline focused update. Arch Pathol Lab Med 142(11):1364–1382. https://doi.org/10.5858/arpa.2018-0902-SA
Mittendorf EA, Wu Y, Scaltriti M, Meric-Bernstam F, Hunt KK, Dawood S et al (2009) Loss of HER2 amplification following trastuzumab-based neoadjuvant systemic therapy and survival outcomes. Clin Cancer Res 15(23):7381–7388. https://doi.org/10.1158/1078-0432.CCR-09-1735
Bertos NR, Park M (2011) Breast cancer—one term, many entities? J Clin Invest 121(10):3789–3796. https://doi.org/10.1172/JCI57100
Manson QF, Schrijver W, Ter Hoeve ND, Moelans CB, van Diest PJ (2019) Frequent discordance in PD-1 and PD-L1 expression between primary breast tumors and their matched distant metastases. Clin Exp Metastasis 36(1):29–37. https://doi.org/10.1007/s10585-018-9950-6
Sun JY, Zhang D, Wu S, Xu M, Zhou X, Lu XJ et al (2020) Resistance to PD-1/PD-L1 blockade cancer immunotherapy: mechanisms, predictive factors, and future perspectives. Biomark Res 8:35. https://doi.org/10.1186/s40364-020-00212-5
Kalinsky K, Diamond JR, Vahdat LT, Tolaney SM, Juric D, O’Shaughnessy J et al (2020) Sacituzumab govitecan in previously treated hormone receptor-positive/HER2-negative metastatic breast cancer: final results from a phase I/II, single-arm, basket trial. Ann Oncol 31(12):1709–1718. https://doi.org/10.1016/j.annonc.2020.09.004
Acknowledgements
We thank the patients and their caregivers for helping us realize the possibilities of this research. We thank the dedicated clinical trial investigators and their devoted team members participating in the ASCENT trial. We thank Loretta M. Itri, MD, Martin S. Olivo, MD, and Quan Hong, PhD, for their work in data analysis of this subgroup. Medical writing and editorial assistance were provided by Kim Farina, PhD, and Yao Bian, PhD, of Team 9 Science, and was funded by Gilead Sciences, Inc.
Funding
This study is funded by Gilead Sciences, Inc.
Author information
Authors and Affiliations
Contributions
Conceptualization of the study and writing of the manuscript was performed by JOS, HSR, YZ, SP, and JC. Acquisition of data was performed by JOS, Adam Brufsky, HSR, SMT, KP, SS, EH, DL, TT, RL-F, SAH, KK, Aditya Bardia, SH, IM, and JC. Data analysis and interpretation was performed by YZ and SP. Review and revision of the manuscript and reading and approval of the final manuscript version was performed by all authors.
Corresponding author
Ethics declarations
Conflict of interest
Joyce O’Shaughnessy reports consultancy/advisory roles with AbbVie, Agendia, Amgen, Aptitude Health, AstraZeneca, Bristol Myers Squibb, Celgene, Eisai, G1 Therapeutics, Genentech/Roche, Gilead, Ipsen, Eli Lilly, Merck, Myriad, Novartis, Odonate, Pfizer, Puma, Prime Oncology, Seattle Genetics, and Syndax; honoraria from Gilead. Adam Brufsky reports consultancy roles with Pfizer, Gilead, Roche, Eli Lilly, Novartis, Eisai, Genentech, and Merck. Hope S. Rugo reports research funding from AstraZeneca, Pfizer, Novartis, Eli Lilly, Genentech, Gilead, MacroGenics, OBI, Merck, Gilead, Daiichi Sankyo, Seattle Genetics, Odonate, Ayala, Sanofi, and Sermonix; honoraria from Mylan, Samsung, and Puma. Sara M. Tolaney reports research funding from Bristol Myers Squibb, Eisai, Gilead, Genentech/Roche, Pfizer, Novartis, Nektar, Merck, AstraZeneca, Eli Lilly, and Exelixis. Kevin Punie reports research funding from Sanofi and MSD; consultancy/advisory roles with AstraZeneca, Eli Lilly, Gilead, Medscape, Novartis, Pfizer, Pierre Fabre, Roche, Roularta, and Vifor Pharma; honoraria from Eli Lilly, McCann Health, Mundi Pharma, MSD, Novartis, Pfizer, and Roche; travel/accommodations/expenses from AstraZeneca, Novartis, PharmaMar, Roche, and Pfizer. Sagar Sardesai reports consultancy roles with Gilead, Novartis, and Biotheranostics. Erika Hamilton reports institutional research funding from OncoMed, Genentech/Roche, Zymeworks, Rgenix, ArQule, Clovis, Silverback Therapeutics, Millenium, Acerta Pharma, Sermonix Pharmaceuticals, Torque, Black Diamond, Karyopharm, Infinity Pharmaceuticals, Curis, Syndax, Novartis, Boehringer Ingelheim, Immunomedics, FujiFilm, Taiho, Deciphera, Fochon, Molecular Templates, Onconova Therapeutics, Dana Farber Cancer Hospital, Hutchinson MediPharma, MedImmune, SeaGen, Puma Biotechnology, Compugen, TapImmune, Lilly, Pfizer, H3 Biomedicine, Takeda, Merus, Regeneron, Arvinas, StemCentRx, Verastem, eFFECTOR Therapeutics, CytomX, InventisBio, Lycera, Mersana, Radius Health, AbbVie, Nucana, Leap Therapeutics, Zenith Epigenetics, Harpoon, Orinove, AstraZeneca, Tesaro, Macrogenics, EMD Serono, Daiichi Sankyo, Syros, Sutro, G1 Therapeutics, Merck, PharmaMar, Olema, Polyphor, Immunogen, Plexxicon, Amgen, Akesobio Australia, and Shattuck Labs; consultancy/advisory roles with Genentech/Roche, Boehringer Ingelheim, Novartis, Dantari, Eli Lilly, Merck, Puma, Silverback Therapeutics, CytomX, Pfizer, Mersana, Black Diamond, H3 Biomedicine, Daiichi Sankyo, AstraZeneca, Arvinas, Deciphera Pharmaceuticals, Eisai, and Seattle Genetics. Delphine Loirat reports consultancy/advisory roles with Novartis, MSD, and Roche. Tiffany Traina reports consultancy/advisory roles with Genentech/Roche, Pfizer, AstraZeneca, Merck, Puma, Advaxis, Celgene, Innocrin, Genomic Health, Bristol Myers Squibb, Athenex, Aduro, Halozyme, Daiichi Sankyo, Ionis, Seattle Genetics, and Gilead; institutional research funding from Eisai, Pfizer, Novartis, Innocrin, AstraZeneca, Astellas, Gilead, Genentech/Roche, Daiichi Sankyo, and Carrick. Roberto Leon-Ferre reports consultancy/advisory roles with AstraZeneca and Gilead; research funding from the National Center for Advancing Translational Science and the Mayo Clinic Breast Cancer SPORE Career Enhancement Program. Sara A. Hurvitz reports research funding from Ambrx, Amgen, Arvinas, Bayer, Daiichi Sankyo, Genentech/Roche, GSK, Gilead, Eli Lilly, Macrogenics, Novartis, Pfizer, OBI Pharma, Pieris, PUMA, Phoenix Molecular Designs, Radius, Sanofi, Seattle Genetics, and Dignitana; stock from NK Max; stock (spouse) from ROMTech and Ideal Implant. Kevin Kalinsky reports research funding from Gilead, Novartis, Incyte, Genentech, Eli Lilly, Pfizer, Calithera, Acetylon, Seattle Genetics, Amgen, Zeno, and CytomX Therapeutics; advisory roles with Gilead, AstraZeneca, and Genentech; consulting fees from Gilead, Pfizer, Eisai, Eli Lilly, Novartis, Amgen, and AstraZeneca, Genentech, Merck, Seattle Genetics, Cyclacel, and OncoSec; honoraria from Eli Lilly; travel/accommodations/expenses from Eli Lilly, Pfizer, and AstraZeneca; employment (spouse) with Array Biopharma, Pfizer, and Grail. Aditya Bardia reports research funding from Genentech, Novartis, Pfizer, Merck, Sanofi, Radius Health, Gilead, and Biotheranostics; personal fees from Pfizer, Novartis, Genentech, Merck, Radius Health, Gilead, Taiho, Sanofi, Daiichi Sankyo/AstraZeneca, Puma, Phillips, Eli Lilly, and Foundation Medicine. Stephanie Henry reports consultancy/advisory roles with AstraZeneca, BMS, Merck, Gilead, Novartis, MSD, and Sanofi; travel/accommodations/expenses from MSD, Novartis, and Roche. Ingrid Mayer reports consultancy/advisory roles with Novartis, Pfizer, Genentech, Eli Lilly, Puma, AbbVie, Gilead, Macrogenics, Seattle Genetics, AstraZeneca, GSK, Cyclacel, Blueprint; research funding from Pfizer and Genentech. Yanni Zhu and See Phan report employment with Gilead and may own stock or stock options. Javier Cortés reports consultancy/advisory roles with Roche, Celgene, Cellestia, AstraZeneca, Seattle Genetics, Daiichi Sankyo, Erytech, Athenex, Polyphor, Lilly, Merck, GSK, Leuko, Bioasis, Clovis Oncology, Boehringer Ingelheim, Ellipses, Hibercell, BioInvent, GEMoaB, and Gilead; honoraria from Roche, Novartis, Celgene, Eisai, Pfizer, Samsung Bioepis, Lilly, Merck, and Daiichi Sankyo; institutional research funding from Roche, ARIAD Pharmaceuticals, AstraZeneca, Baxalta GMBH/Servier Affaires, Bayer, Eisai, Roche, Guardant Health, Merck, Pfizer, Piqur Therapeutics, Puma, Queen Mary University of London; stock, patents, and intellectual property with MedSIR; travel, accommodation, and expenses with Roche, Novartis, Eisai, Pfizer, and Daiichi Sankyo.
Ethical approval
The ASCENT trial was conducted and approved by each investigational site’s institutional review board/ethics committee prior to initiation, and in accordance with the Declaration of Helsinki, International Council for Harmonisation Guidelines for Good Clinical Practice, FDA Code of Federal Regulations, national and local drug and data protection laws, and other applicable regulatory requirements.
Consent to participate
All patients provided written informed consent prior to enrollment in the study.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
O’Shaughnessy, J., Brufsky, A., Rugo, H.S. et al. Analysis of patients without and with an initial triple-negative breast cancer diagnosis in the phase 3 randomized ASCENT study of sacituzumab govitecan in metastatic triple-negative breast cancer. Breast Cancer Res Treat 195, 127–139 (2022). https://doi.org/10.1007/s10549-022-06602-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-022-06602-7