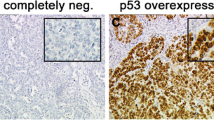
Abstract
Purpose
The TF (Thomsen–Friedenreich, CD176, Galβ1-3GalNAc) carbohydrate moiety is known as a specific oncofetal carbohydrate epitope present in fetal and neoplastic tissue as well as in stem cells. TF was demonstrated to mediate tumor-promoting features and to be highly immunogenic. The current study aimed to evaluate whether presence of the TF antigen is associated with clinico-pathological parameters and prognosis of early breast cancer (BC).
Methods
Primary BC tissue (n = 226) was stained for TF using two monoclonal anti-TF antibodies (Nemod-TF1, Nemod-TF2). Staining results were correlated to clinical data including survival.
Results
Nemod-TF1 staining was positively correlated to lymph node metastasis (p = 0.03) and the presence of tumor-associated MUC1 (TA-MUC1; p = 0.003). Further, the presence of the Nemod-TF1 epitope predicted worse prognosis in TA-MUC1 positive (overall survival: p = 0.026) as well as in triple negative (overall survival: p = 0.002; distant metastasis-free survival: p = 0.012) BC.
Conclusions
The data presented here further support a role of TF in BC tumor biology. Whether anti-TF directed treatment approaches may gain clinical relevance in those cases determined as triple negative or TA-MUC1 positive remains to be determined.


Similar content being viewed by others
Abbreviations
- BC:
-
Breast cancer
- CIS:
-
Carcinoma in situ
- CSC:
-
Cancer stem cell
- DFS:
-
Disease-free survival
- DMFS:
-
Distant metastasis-free survival
- ER:
-
Estrogen receptor
- FFPE:
-
Formalin-fixed, paraffin-embedded
- IRS:
-
Immunoreactive score
- LRFS:
-
Local recurrence-free survival
- OS:
-
Overall survival
- PR:
-
Progesterone receptor
- REMARK:
-
REporting recommendations for tumor MARKer prognostic studies
- TA-MUC1:
-
Tumor-associated mucin-1
- TF:
-
Thomsen–Friedenreich, CD176, Galβ1-3GalNAc
- TNBC:
-
Triple negative BC
- WHO:
-
World Health Organization
References
Karsten U, Goletz S (2015) What controls the expression of the core-1 (Thomsen–Friedenreich) glycotope on tumor cells? Biochemistry (Mosc) 80(7):801–807. https://doi.org/10.1134/S0006297915070019
Dippold W, Steinborn A, Meyer zum Buschenfelde KH (1990) The role of the Thomsen–Friedenreich antigen as a tumor-associated molecule. Environ Health Perspect 88:255–257
Satyanarayana J, Gururaja TL, Narasimhamurthy S, Naganagowda GA, Levine MJ (2001) Synthesis and conformational features of human salivary mucin C-terminal derived peptide epitope carrying Thomsen–Friedenreich antigen: implications for its role in self-association. Biopolymers 58(5):500–510. https://doi.org/10.1002/1097-0282(20010415)58:5%3c500:AID-BIP1025%3e3.0.CO;2-5
Karsten U, Goletz S (2013) What makes cancer stem cell markers different? SpringerPlus 2(1):301. https://doi.org/10.1186/2193-1801-2-301
Axford SE (2003) Glycobiology and Medicine, 1st edn. Springer, New York
Yu LG, Andrews N, Zhao Q, McKean D, Williams JF, Connor LJ, Gerasimenko OV, Hilkens J, Hirabayashi J, Kasai K, Rhodes JM (2007) Galectin-3 interaction with Thomsen–Friedenreich disaccharide on cancer-associated MUC1 causes increased cancer cell endothelial adhesion. J Biol Chem 282(1):773–781. https://doi.org/10.1074/jbc.M606862200
Geiger P, Mayer B, Wiest I, Schulze S, Jeschke U, Weissenbacher T (2016) Binding of galectin-1 to breast cancer cells MCF7 induces apoptosis and inhibition of proliferation in vitro in a 2D- and 3D- cell culture model. BMC Cancer 16(1):870. https://doi.org/10.1186/s12885-016-2915-8
Rittenhouse-Olson K (2007) Jaa-f11: extending the life of mice with breast cancer. Expert Opin Biol Ther 7(7):923–928. https://doi.org/10.1517/14712598.7.7.923
Ferguson K, Yadav A, Morey S, Abdullah J, Hrysenko G, Eng JY, Sajjad M, Koury S, Rittenhouse-Olson K (2014) Preclinical studies with JAA-F11 anti-Thomsen–Friedenreich monoclonal antibody for human breast cancer. Future Oncol 10(3):385–399. https://doi.org/10.2217/fon.13.209
Goletz S, Cao Y, Danielczyk A, Ravn P, Schoeber U, Karsten U (2003) Thomsen–Friedenreich antigen: the “hidden” tumor antigen. Adv Exp Med Biol 535:147–162
He ZY, Wu SG, Zhou J, Li FY, Lin Q, Lin HX, Sun JY (2015) Postmastectomy radiotherapy improves disease-free survival of high risk of locoregional recurrence breast cancer patients with T1-2 and 1 to 3 positive nodes. PLoS ONE 10(3):e0119105. https://doi.org/10.1371/journal.pone.0119105
Heublein S, Page SK, Mayr D, Ditsch N, Jeschke U (2016) p53 determines prognostic significance of the carbohydrate stem cell marker TF1 (CD176) in ovarian cancer. J Cancer Res Clin Oncol 142(6):1163–1170. https://doi.org/10.1007/s00432-016-2126-3
Jeschke U, Walzel H, Mylonas I, Papadopoulos P, Shabani N, Kuhn C, Schulze S, Friese K, Karsten U, Anz D, Kupka MS (2009) The human endometrium expresses the glycoprotein mucin-1 and shows positive correlation for Thomsen–Friedenreich epitope expression and galectin-1 binding. J Histochem Cytochem 57(9):871–881. https://doi.org/10.1369/jhc.2009.952085
Engelstaedter V, Fluegel B, Kunze S, Mayr D, Friese K, Jeschke U, Bergauer F (2012) Expression of the carbohydrate tumour marker Sialyl Lewis A, Sialyl Lewis X, Lewis Y and Thomsen–Friedenreich antigen in normal squamous epithelium of the uterine cervix, cervical dysplasia and cervical cancer. Histol Histopathol 27(4):507–514
Dian D, Janni W, Kuhn C, Mayr D, Karsten U, Mylonas I, Friese K, Jeschke U (2009) Evaluation of a novel anti-mucin 1 (MUC1) antibody (PankoMab) as a potential diagnostic tool in human ductal breast cancer; comparison with two established antibodies. Onkologie 32(5):238–244. https://doi.org/10.1159/000209280
Dian D, Lenhard M, Mayr D, Heublein S, Karsten U, Goletz S, Kuhn C, Wiest I, Friese K, Weissenbacher T, Jeschke U (2013) Staining of MUC1 in ovarian cancer tissues with PankoMab-GEX detecting the tumour-associated epitope, TA-MUC1, as compared to antibodies HMFG-1 and 115D8. Histol Histopathol 28(2):239–244
Scholz C, Toth B, Barthell E, Mylonas I, Weissenbacher T, Friese K, Jeschke U (2009) Immunohistochemical expression of glycodelin in breast cancer correlates with estrogen-receptor alpha and progesterone-receptor a positivity. Histol Histopathol 24(4):467–471
Heublein S, Mayr D, Egger M, Karsten U, Goletz S, Angele M, Gallwas J, Jeschke U, Ditsch N (2015) Immunoreactivity of the fully humanized therapeutic antibody PankoMab-GEX is an independent prognostic marker for breast cancer patients. J. Exp. Clin. Cancer Res 34:50. https://doi.org/10.1186/s13046-015-0152-7
McShane LM, Altman DG, Sauerbrei W, Taube SE, Gion M, Clark GM (2005) Reporting recommendations for tumor marker prognostic studies. J Clin Oncol 23(36):9067–9072. https://doi.org/10.1200/JCO.2004.01.0454
Cao Y, Karsten UR, Liebrich W, Haensch W, Springer GF, Schlag PM (1995) Expression of Thomsen–Friedenreich-related antigens in primary and metastatic colorectal carcinomas. A reevaluation. Cancer 76(10):1700–1708
Imai J, Ghazizadeh M, Naito Z, Asano G (2001) Immunohistochemical expression of T, Tn and sialyl-Tn antigens and clinical outcome in human breast carcinoma. Anticancer Res 21(2B):1327–1334
Lin WM, Karsten U, Goletz S, Cheng RC, Cao Y (2011) Expression of CD176 (Thomsen–Friedenreich antigen) on lung, breast and liver cancer-initiating cells. Int J Exp Pathol 92(2):97–105. https://doi.org/10.1111/j.1365-2613.2010.00747.x
Schindlbeck C, Jeschke U, Schulze S, Karsten U, Janni W, Rack B, Krajewski S, Sommer H, Friese K (2007) Prognostic impact of Thomsen–Friedenreich tumor antigen and disseminated tumor cells in the bone marrow of breast cancer patients. Breast Cancer Res Treat 101(1):17–25. https://doi.org/10.1007/s10549-006-9271-3
Gunkel L, Mylonas I, Richter DU, Makovitzky J (2005) Immunohistochemical studies of mucinous mammary carcinomas and their metastases. Anticancer Res 25(3A):1755–1759
Danielczyk A, Stahn R, Faulstich D, Loffler A, Marten A, Karsten U, Goletz S (2006) PankoMab: a potent new generation anti-tumour MUC1 antibody. Cancer Immunol Immunother 55(11):1337–1347. https://doi.org/10.1007/s00262-006-0135-9
Masuzawa Y, Miyauchi T, Hamanoue M, Ando S, Yoshida J, Takao S, Shimazu H, Adachi M, Muramatsu T (1992) A novel core protein as well as polymorphic epithelial mucin carry peanut agglutinin binding sites in human gastric carcinoma cells: sequence analysis and examination of gene expression. J Biochem 112(5):609–615
Lloyd KO, Burchell J, Kudryashov V, Yin BW, Taylor-Papadimitriou J (1996) Comparison of O-linked carbohydrate chains in MUC-1 mucin from normal breast epithelial cell lines and breast carcinoma cell lines. Demonstration of simpler and fewer glycan chains in tumor cells. J Biol Chem 271(52):33325–33334
Quin RJ, McGuckin MA (2000) Phosphorylation of the cytoplasmic domain of the MUC1 mucin correlates with changes in cell-cell adhesion. Int J Cancer 87(4):499–506
Raina D, Kharbanda S, Kufe D (2004) The MUC1 oncoprotein activates the anti-apoptotic phosphoinositide 3-kinase/Akt and Bcl-xL pathways in rat 3Y1 fibroblasts. J Biol Chem 279(20):20607–20612. https://doi.org/10.1074/jbc.M310538200
Baldus SE, Zirbes TK, Glossmann J, Fromm S, Hanisch FG, Monig SP, Schroder W, Schneider PM, Flucke U, Karsten U, Thiele J, Holscher AH, Dienes HP (2001) Immunoreactivity of monoclonal antibody BW835 represents a marker of progression and prognosis in early gastric cancer. Oncology 61(2):147–155. https://doi.org/10.1159/000055366
Baldus SE, Zirbes TK, Hanisch FG, Kunze D, Shafizadeh ST, Nolden S, Monig SP, Schneider PM, Karsten U, Thiele J, Holscher AH, Dienes HP (2000) Thomsen–Friedenreich antigen presents as a prognostic factor in colorectal carcinoma: a clinicopathologic study of 264 patients. Cancer 88(7):1536–1543
Takanami I (1999) Expression of Thomsen–Friedenreich antigen as a marker of poor prognosis in pulmonary adenocarcinoma. Oncol Rep 6(2):341–344
Hirao T, Sakamoto Y, Kamada M, Hamada S, Aono T (1993) Tn antigen, a marker of potential for metastasis of uterine cervix cancer cells. Cancer 72(1):154–159
Choufani G, Nagy N, Saussez S, Marchant H, Bisschop P, Burchert M, Danguy A, Louryan S, Salmon I, Gabius HJ, Kiss R, Hassid S (1999) The levels of expression of galectin-1, galectin-3, and the Thomsen–Friedenreich antigen and their binding sites decrease as clinical aggressiveness increases in head and neck cancers. Cancer 86(11):2353–2363
Heimburg J, Yan J, Morey S, Glinskii OV, Huxley VH, Wild L, Klick R, Roy R, Glinsky VV, Rittenhouse-Olson K (2006) Inhibition of spontaneous breast cancer metastasis by anti-Thomsen–Friedenreich antigen monoclonal antibody JAA-F11. Neoplasia 8(11):939–948. https://doi.org/10.1593/neo.06493
Egger M (2014) Untersuchungen zur Expression von Mucin1 im Tumorgewebe von Brustkrebspatientinnen: eine Studie zur Korrelation mit dem Thomsen–Friedenreich Antigen und mit Her4 sowie Phospho-Her4. Dissertation. LMU München: Medizinische Fakultät
Acknowledgements
The authors thank Irmgard Wiest and Susanne Kunze (†) for their excellent technical assistance. We would further like to acknowledge Jutta Engel and Max Wiedemann for their help with collecting follow-up data from the Munich Cancer Registry. Parts of this work are part of ME’s MD thesis obtained at the Department of Obstetrics and Gynecology, LMU Munich, Germany [36].
Funding
The study was financed by the Department of Obstetrics and Gynecology, Ludwig-Maximilians-University of Munich, Munich, Germany. No special funding was received.
Author information
Authors and Affiliations
Contributions
SH participated in design and coordination of the study, significantly participated in experimental assays and analysis as well as in statistical analysis, and wrote the manuscript. ME participated in IHC assays and analysis and helped to draft the manuscript. LB participated in data collection and analysis. JZ participated in experimental assays. DM supervised IHC as a gynecologic pathologist and participated in IHC analysis. CS participated in the design of the study and carefully reviewed the manuscript for important intellectual content. CK optimized Nemod-TF staining and participated in immunohistochemistry. SSH participated in cell culture experiment design and conduction. FS helped to draft the manuscript and revised the manuscript for important intellectual content. UJ conceived and coordinated the study, participated in study design, and approved the final version of the manuscript. ND conceived and coordinated the study, participated in study design, and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors read the manuscript and agree to the publication of the manuscript. SH reports grants from the following organizations/companies: FöFoLe LMU Munich Medical Faculty, FERRING, Novartis Oncology, Astra Zeneca, Apceth, Heuer Stiftung, Deutsche Forschungsgemeinschaft. She further reports personal fees from Roche and non-financial support from Addex. ND held honorary speeches for Roche, AstraZeneca, Mentor, Omniamed, TEVA, and MSD. All the support listed here has been received outside the submitted work. Remaining authors have no competing interests to declare.
Ethical approval
This article does not contain any studies with animals performed by any of the authors. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The tissue samples were retrieved from the archive of Obstetrics and Gynecology, Ludwig-Maximilians-University, Munich, Germany. The study was approved by the Ethics Committee of the Ludwig-Maximilians-University, Munich, Germany (approval number: 18-166). The tumor tissue re-used for our analysis had initially been collected for histopathological diagnostics. At the time, the tissue was examined for the current study all diagnostic procedures had already been fully completed and thus the tissue was classified as “left-over” material. Clinical data and tissue specimens were irreversibly anonymized prior to inclusion in the study. Researchers were blinded from patient data during experimental and statistical analysis.
Informed consent
As per declaration of our ethics committee [Ludwig-Maximilians-University, Munich, Germany (approval number: 18-166)], no written informed consent of the participants is needed given the circumstances described above.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.

10549_2019_5503_MOESM1_ESM.jpg
Supplementary material 1 (JPEG 63 kb) Supplementary Figure 1: Disease-free survival (DFS) of Nemod-TF1-positive vs. -negativepatients in the subgroup of TA-MUC1mem-positive (A) and triple negative (B) breast cancers

10549_2019_5503_MOESM2_ESM.jpg
Supplementary material 2 (JPEG 28 kb) Supplementary Figure 2: Distant metastasis-free survival (DMFS) of Nemod-TF1-positive vs. -negative patients in the subgroup of Her2-positive breast cancers
Rights and permissions
About this article
Cite this article
Heublein, S., Egger, M., Zhu, J. et al. Evaluation of the anti-Thomsen–Friedenreich antibodies Nemod-TF1 and Nemod-TF2 as prognostic markers in breast cancer. Breast Cancer Res Treat 179, 643–652 (2020). https://doi.org/10.1007/s10549-019-05503-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-019-05503-6