Abstract
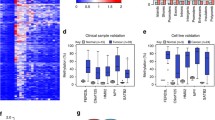
Identification of novel targets for the treatment of basal-like breast cancer is essential for improved outcomes in patients with this disease. This study investigates the association of MMP7 expression and MMP7 promoter methylation with subtype and outcome in breast cancer patient cohorts. Immunohistochemical analysis was performed on a breast cancer tissue microarray and validated in independent histological samples. MMP7 expression significantly correlated with patient age, tumor size, triple-negative (TN) status, and recurrence. Analysis of publically available datasets confirmed MMP7 gene expression as a prognostic marker of breast cancer metastasis, particularly metastasis to the brain and lungs. Methylation of the MMP7 promoter was assessed by methylation-specific PCR in a panel of breast cancer cell lines and patient tumor samples. Hypomethylation of the MMP7 promoter significantly correlated with TN status in DNA from patient tumor samples, and this association was confirmed using The Cancer Genome Atlas (TCGA) dataset. Evaluation of a panel of breast cancer cell lines and data from the Curtis and TCGA breast carcinoma datasets revealed that elevated MMP7 expression and MMP7 promoter hypomethylation are specific biomarkers of the basal-like molecular subtype which shares considerable, but not complete, overlap with the clinical TN subtype. Importantly, MMP7 expression was identified as an independent predictor of pathological complete response in a large breast cancer patient cohort. Combined, these data suggest that MMP7 expression and MMP7 promoter methylation may be useful as prognostic biomarkers. Furthermore, MMP7 expression and promoter methylation analysis may be effective mechanisms to distinguish basal-like breast cancers from other triple-negative subtypes. Finally, these data implicate MMP7 as a potential therapeutic target for the treatment of basal-like breast cancers.




Similar content being viewed by others
References
Hua H, Li M, Luo T, Yin Y, Jiang Y (2011) Matrix metalloproteinases in tumorigenesis: an evolving paradigm. Cell Mol Life Sci 68(23):3853–3868
Kleiner DE, Stetler-Stevenson WG (1999) Matrix metalloproteinases and metastasis. Cancer Chemother Pharmacol 43(Suppl):S42–S51
Gialeli C, Theocharis AD, Kletsas D, Tzanakakis GN, Karamanos NK (2013) Expression of matrix macromolecules and functional properties of EGF-responsive colon cancer cells are inhibited by panitumumab. Invest New Drugs 31(3):516–524
Noe V, Fingleton B, Jacobs K, Crawford HC, Vermeulen S, Steelant W, Bruyneel E, Matrisian LM, Mareel M (2001) Release of an invasion promoter E-cadherin fragment by matrilysin and stromelysin-1. J Cell Sci 114(Pt 1):111–118
Rucci N, Sanita P, Angelucci A (2011) A Roles of metalloproteases in metastatic niche. Curr Mol Med 11(8):609–622
Yu Q, Stamenkovic I (2000) Cell surface-localized matrix metalloproteinase-9 proteolytically activates TGF-beta and promotes tumor invasion and angiogenesis. Genes Dev 14(2):163–176
Yu WH, Woessner JF Jr, McNeish JD, Stamenkovic I (2002) CD44 anchors the assembly of matrilysin/MMP-7 with heparin-binding epidermal growth factor precursor and ErbB4 and regulates female reproductive organ remodeling. Genes Dev 16(3):307–323
Heppner KJ, Matrisian LM, Jensen RA, Rodgers WH (1996) Expression of most matrix metalloproteinase family members in breast cancer represents a tumor-induced host response. Am J Path 149(1):273–282
Fang YJ, Pan ZZ, Li LR, Lu ZH, Zhang LY, Wan DS (2009) MMP7 expression regulated by endocrine therapy in ERbeta-positive colon cancer cells. J Exp Clin Cancer Res 28:132
Fukuda A, Wang SC, Morris JPt, Folias AE, Liou A, Kim GE, Akira S, Boucher KM, Firpo MA, Mulvihill SJ, Hebrok M (2011) Stat3 and MMP7 contribute to pancreatic ductal adenocarcinoma initiation and progression. Cancer Cell 19(4):441–455
Gentner B, Wein A, Croner RS, Zeittraeger I, Wirtz RM, Roedel F, Dimmler A, Dorlaque L, Hohenberger W, Hahn EG, Brueckl WM (2009) Differences in the gene expression profile of matrix metalloproteinases (MMPs) and their inhibitors (TIMPs) in primary colorectal tumors and their synchronous liver metastases. Anticancer Res 29(1):67–74
Tonini G, Pantano F, Vincenzi B, Gabbrielli A, Coppola R, Santini D (2007) Molecular prognostic factors in patients with pancreatic cancer. Expert Opin Ther Targets 11(12):1553–1569
Fang YJ, Lu ZH, Wang F, Wu XJ, Li LR, Zhang LY, Pan ZZ, Wan DS (2010) Prognostic impact of ERβ and MMP7 expression on overall survival in colon cancer. Tumour Biol 31(6):651–658
Huang Y, Yu H, Lei H, Xie C, Zhong Y (2014) Matrix metalloproteinase 7 is a usefulmarker for 5-fluorouracil-based adjuvant chemotherapy in stage II and stage III colorectal cancer patients. Med Oncol 31:824
Gallego R, Codony-Servat J, García-Albéniz X, Carcereny E, Longarón R, Oliveras A, Tosca M, Augé JM, Gascón P, Maurel J (2009) Serum IGF-I, IGFBP-3, and matrix metalloproteinase-7 levels and acquired chemo-resistance in advanced colorectal cancer. Endocr Relat Cancer 16(1):1412–1420
Fang YJ, Lu ZH, Wang GQ, Pan ZZ, Zhou ZW, Yun JP, Zhang MF, Wan DS (2009) Elevated expressions of MMP7, TROP2, and survivin are associated with survival, disease recurrence, and liver metastasis of colon cancer. Int J Colorectal Dis 24(8):875–884
Wu J, Guan X, Zhang K, Li YT, Bai P, Wu J (2013) A/G polymorphism of matrix metalloproteinase 7 gene promoter region and cancer risk: a meta-analysis. Biomed Rep 1(5):792–796
Ke P, Wu ZD, Wen HS, Ying MX, Long HC, Qing LG (2013) Current evidence on associations between the MMP-7 (-181A > G) polymorphism and digestive system cancer risk. Asian Pac J Cancer Prev 14(4):2269–2272
Liu D, Guo H, Li Y, Xu X, Yang K, Bai Yc (2012) Association between polymorphisms in the promoter regions of matrix metalloproteinases (MMPs) and risk of cancer metastasis: a meta-analysis. PLoS ONE 7(2):e31251
Saarialho-Kere UK, Crouch EC, Parks WC (1995) Matrix metalloproteinase matrilysin is constitutively expressed in adult human exocrine epithelium. J Invest Dermatol 105(2):190–196
Yuan G, Qian L, Shi M, Lu F, Li D, Hu M, Yu M, Shen B, Guo N (2008) HER2-dependent MMP-7 expression is mediated by activated STAT3. Cell Signal 20(7):1284–1291
Yuan G, Qian L, Song L, Shi M, Li D, Yu M, Hu M, Shen B, Guo N (2008) Heregulin-beta promotes matrix metalloproteinase-7 expression via HER2-mediated AP-1 activation in MCF-7 cells. Mol Cell Biochem 318(1–2):73–79
Sizemore ST, Keri RA (2012) The forkhead box transcription factor FOXC1 promotes breast cancer invasion by inducing matrix metalloprotease 7 (MMP7) expression. J Biol Chem 287(29):24631–24640
Bertucci F, Finetti P, Cervera N, Esterni B, Hermitte F, Viens P, Birnbaum D (2008) How basal are triple-negative breast cancers? Int J Cancer 123(1):236–240
Valentin MD, da Silva SD, Privat M, Alaoui-Jamali M, Bignon YJ (2012) Molecular insights on basal-like breast cancer. Breast Cancer Res Treat 134(1):21–30
Parker JS, Mullins M, Cheang MC, Leung S, Voduc D, Vickery T, Davies S, Fauron C, He X, Hu Z, Quackenbush JF, Stijleman IJ, Palazzo J, Marron JS, Nobel AB, Mardis E, Nielsen TO, Ellis MJ, Perou CM, Bernard PS (2009) Supervised risk predictor of breast cancer based on intrinsic subtypes. J Clin Oncol 27(8):1160–1167
Sørlie T, Perou CM, Tibshirani R, Aas T, Geisler S, Johnsen H, Hastie T, Eisen MB, van de Rijn M, Jeffrey SS, Thorsen T, Quist H, Matese JC, Brown PO, Botstein D, Lønning PE, Børresen-Dale AL (2001) Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci USA 98(19):10869–10874
Weigelt B, Hu Z, He X, Livasy C, Carey LA, Ewend MG, Glas AM, Perou CM, Van’t-Veer LJ (2005) Molecular portraits and 70-gene prognosis signature are preserved throughout the metastatic process of breast cancer. Cancer Res 65(20):9155–9158
Carey LA, Dees EC, Sawyer L, Gatti L, Moore DT, Collichio F, Ollila DW, Sartor CI, Graham ML, Perou CM (2007) The triple negative paradox: primary tumor chemosensitivity of breast cancer subtypes. Clin Cancer Res 13(8):2329–2334
Sato N, Maehara N, Su GH, Goggins M (2003) Effects of 5-aza-2′-deoxycytidine on matrix metalloproteinase expression and pancreatic cancer cell invasiveness. J Natl Cancer Inst 95(4):327–330
Schneider CA, Rasband WS, Eliceiri KW (2012) Image to ImageJ: 25 years of image analysis. Nat Methods 9(7):671–675
Bos PD, Zhang XH, Nadal C, Shu W, Gomis RR, Nguyen DX, Minn AJ, van de Vijver MJ, Gerald WL, Foekens JA, Massague J (2009) Genes that mediate breast cancer metastasis to the brain. Nature 459(7249):1005–1009
Curtis C, Shah SP, Chin SF, Turashvili G, Rueda OM, Dunning MJ, Speed D, Lynch AG, Samarajiwa S, Yuan Y, Graf S, Ha G, Haffari G, Bashashati A, Russell R, McKinney S, Langerod A, Green A, Provenzano E, Wishart G, Pinder S, Watson P, Markowetz F, Murphy L, Ellis I, Purushotham A, Borresen-Dale AL, Brenton JD, Tavare S, Caldas C, Aparicio S (2012) The genomic and transcriptomic architecture of 2,000 breast tumours reveals novel subgroups. Nature 486(7403):346–352
Hatzis C, Pusztai L, Valero V, Booser DJ, Esserman L, Lluch A, Vidaurre T, Holmes F, Souchon E, Wang H, Martin M, Cotrina J, Gomez H, Hubbard R, Chacon JI, Ferrer-Lozano J, Dyer R, Buxton M, Gong Y, Wu Y, Ibrahim N, Andreopoulou E, Ueno NT, Hunt K, Yang W, Nazario A, DeMichele A, O’Shaughnessy J, Hortobagyi GN, Symmans WF (2011) A genomic predictor of response and survival following taxane-anthracycline chemotherapy for invasive breast cancer. JAMA 305(18):1873–1881
Cancer Genome Atlas Network (2012) Comprehensive molecular portraits of human breast tumours. Nature 490(7418):61–70
Camp RL, Neumeister V, Rimm DL (2008) A decade of tissue microarrays: progress in the discovery and validation of cancer biomarkers. J Clin Oncol 26(34):5630–5637
Hasegawa M, Furuya M, Kasuya Y, Nishiyama M, Sugiura T, Nikaido T, Momota Y, Ichinose M, Kimura S (2007) CD151 dynamics in carcinoma-stroma interaction: integrin expression, adhesion strength and proteolytic activity. Lab Invest 87(9):882–892
Mitsui H, Suarez-Farinas M, Gulati N, Shah KR, Cannizzaro MV, Coats I, Felsen D, Krueger JG, Carucci JA (2013) Gene Expression Profiling of the Leading Edge of Cutaneous Squamous Cell Carcinoma: IL-24-Driven MMP-7. J Invest Dermatol. doi:10.1038/2013.494
Puthenedam M, Wu F, Shetye A, Michaels A, Rhee KJ, Kwon JH (2011) Matrilysin-1 (MMP7) cleaves galectin-3 and inhibits wound healing in intestinal epithelial cells. Inflamm Bowel Dis 17(1):260–267
Billar JA, Dueck AC, Stucky CC, Gray RJ, Wasif N, Northfelt DW, McCullough AE, Pockaj BA (2010) Triple-negative breast cancers: unique clinical presentations and outcomes. Ann Surg Oncol 17(Suppl 3):384–390
Kennecke H, Yerushalmi R, Woods R, Cheang MC, Voduc D, Speers CH, Nielsen TO, Gelmon K (2010) Metastatic behavior of breast cancer subtypes. J Clin Oncol 28(20):3271–3277
Charafe-Jauffret E, Ginestier C, Monville F, Finetti P, Adelaide J, Cervera N, Fekairi S, Xerri L, Jacquemier J, Birnbaum D, Bertucci F (2006) Gene expression profiling of breast cell lines identifies potential new basal markers. Oncogene 25(15):2273–2284
Neve RM, Chin K, Fridlyand J, Yeh J, Baehner FL, Fevr T, Clark L, Bayani N, Coppe JP, Tong F, Speed T, Spellman PT, DeVries S, Lapuk A, Wang NJ, Kuo WL, Stilwell JL, Pinkel D, Albertson DG, Waldman FM, McCormick F, Dickson RB, Johnson MD, Lippman M, Ethier S, Gazdar A, Gray JW (2006) A collection of breast cancer cell lines for the study of functionally distinct cancer subtypes. Cancer Cell 10(6):515–527
Palmieri D, Bronder JL, Herring JM, Yoneda T, Weil RJ, Stark AM, Kurek R, Vega-Valle E, Feigenbaum L, Halverson D, Vortmeyer AO, Steinberg SM, Aldape K, Steeg PS (2007) Her-2 overexpression increases the metastatic outgrowth of breast cancer cells in the brain. Cancer Res 67(9):4190–4198
Perou CM (2011) Molecular stratification of triple-negative breast cancers. Oncologist 16(Suppl 1):61–70
Prat A, Adamo B, Cheang MC, Anders CK, Carey LA, Perou CM (2013) Molecular characterization of basal-like and non-basal-like triple-negative breast cancer. Oncologist 18(2):123–133
Prat A, Parker JS, Karginova O, Fan C, Livasy C, Herschkowitz JI, He X, Perou CM (2010) Phenotypic and molecular characterization of the claudin-low intrinsic subtype of breast cancer. Breast Cancer Res 12(5):R68
Hu Z, Fan C, Oh DS, Marron JS, He X, Qaqish BF, Livasy C, Carey LA, Reynolds E, Dressler L, Nobel A, Parker J, Ewend MG, Sawyer LR, Wu J, Liu Y, Nanda R, Tretiakova M, Ruiz-Orrico A, Dreher D, Palazzo JP, Perreard L, Nelson E, Mone M, Hansen H, Mullins M, Quackenbush JF, Ellis MJ, Olopade OI, Bernard PS, Perou CM (2006) The molecular portraits of breast tumors are conserved across microarray platforms. BMC Genom 7:96
Crawford HC, Fingleton BM, Rudolph-Owen LA, Goss KJ, Rubinfeld B, Polakis P, Matrisian LM (1999) The metalloproteinase matrilysin is a target of beta-catenin transactivation in intestinal tumors. Oncogene 18(18):2883–2891
Brenton JD, Carey LA, Ahmed AA, Caldas C (2005) Molecular classification and molecular forecasting of breast cancer: ready for clinical application? J Clin Oncol 23(29):7350–7360
Cakir A, Gonul II, Uluoglu O (2012) A comprehensive morphological study for basal-like breast carcinomas with comparison to nonbasal-like carcinomas. Diagn Pathol 7:145
Nielsen TO, Hsu FD, Jensen K, Cheang M, Karaca G, Hu Z, Hernandez-Boussard T, Livasy C, Cowan D, Dressler L, Akslen LA, Ragaz J, Gown AM, Gilks CB, van de Rijn M, Perou CM (2004) Immunohistochemical and clinical characterization of the basal-like subtype of invasive breast carcinoma. Clin Cancer Res 10(16):5367–5374
Rodriguez-Pinilla SM, Sarrio D, Honrado E, Moreno-Bueno G, Hardisson D, Calero F, Benitez J, Palacios J (2007) Vimentin and laminin expression is associated with basal-like phenotype in both sporadic and BRCA1-associated breast carcinomas. J Clin Pathol 60(9):1006–1012
Sousa B, Paredes J, Milanezi F, Lopes N, Martins D, Dufloth R, Vieira D, Albergaria A, Veronese L, Carneiro V, Carvalho S, Costa JL, Zeferino L, Schmitt F (2010) P-cadherin, vimentin and CK14 for identification of basal-like phenotype in breast carcinomas: an immunohistochemical study. Histol Histopathol 25(8):963–974
Balak MN, Gong Y, Riely GJ, Somwar R, Li AR, Zakowski MF, Chiang A, Yang G, Ouerfelli O, Kris MG, Ladanyi M, Miller VA, Pao W (2006) Novel D761Y and common secondary T790 M mutations in epidermal growth factor receptor-mutant lung adenocarcinomas with acquired resistance to kinase inhibitors. Clin Cancer Res 12(21):6494–6501
Suda K, Onozato R, Yatabe Y, Mitsudomi T (2009) EGFR T790 M mutation: a double role in lung cancer cell survival? J Thorac Oncol 4(1):1–4
Zucker S, Cao J, Chen WT (2000) Critical appraisal of the use of matrix metalloproteinase inhibitors in cancer treatment. Oncogene 19(56):6642–6650
Zheng G, Chen J, Stefflova K, Jarvi M, Li H, Wilson BC (2007) Photodynamic molecular beacon as an activatable photosensitizer based on protease-controlled singlet oxygen quenching and activation. PNAS 104(21):8989–8994
Liu S, Netzel-Arnett S, Birkedal-Hansen H, Leppla SH (2000) Tumor cell-selective cytotoxicity of matrix metalloproteinase-activated anthrax toxin. Cancer Res 60(21):6061–6067
Liu S, Wang H, Currie BM, Molinolo A, Leung HJ, Moayeri M, Basile JR, Alfano RW, Gutkind JS, Frankel AE, Bugge TH, Leppla SH (2008) Matrix metalloproteinase-activated anthrax lethal toxin demonstrates high potency in targeting tumor vasculature. J Biol Chem 283(1):529–540
Acknowledgments
This work was supported by grants from the Department of Defense W81XWH-09-1-0696 (to STS) and RO1 CA090398 and RO1 CA154384 (to RAK). The Tissue Procurement and Histology Core Facility of the Case Comprehensive Cancer Center (P30 CA043703) was instrumental in the development of the TMA. We thank Darcie Seachrist of the Department of Pharmacology at Case Western Reserve University for her assistance in acquiring images of the pathological samples. We would also like to thank Michael Goggins, M.D. of the Departments of Pathology, Oncology and Medicine at The Johns Hopkins Medical Institute for the primer sequences used to detect the MMP7 promoter methylation status.
Conflict of interest
The authors declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sizemore, S.T., Sizemore, G.M., Booth, C.N. et al. Hypomethylation of the MMP7 promoter and increased expression of MMP7 distinguishes the basal-like breast cancer subtype from other triple-negative tumors. Breast Cancer Res Treat 146, 25–40 (2014). https://doi.org/10.1007/s10549-014-2989-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-014-2989-4