Abstract
The present exploratory analysis examined the efficacy, safety, and quality-of-life effects of everolimus (EVE) + exemestane (EXE) in the subgroup of patients in BOLERO-2 whose last treatment before study entry was in the (neo)adjuvant setting. In BOLERO-2, patients with hormone-receptor-positive (HR+), human epidermal growth factor receptor-2-negative (HER2−) advanced breast cancer recurring/progressing after a nonsteroidal aromatase inhibitor (NSAI) were randomly assigned (2:1) to receive EVE (10 mg/day) + EXE (25 mg/day) or placebo (PBO) + EXE. The primary endpoint was progression-free survival (PFS) by local assessment. Overall, 137 patients received first-line EVE + EXE (n = 100) or PBO + EXE (n = 37). Median PFS by local investigator assessment nearly tripled to 11.5 months with EVE + EXE from 4.1 months with PBO + EXE (hazard ratio = 0.39; 95 % CI 0.25–0.62), while maintaining quality of life. This was confirmed by central assessment (15.2 vs 4.2 months; hazard ratio = 0.32; 95 % CI 0.18–0.57). The marked PFS improvement in patients receiving EVE + EXE as first-line therapy for disease recurrence during or after (neo)adjuvant NSAI therapy supports the efficacy of this combination in the first-line setting. Furthermore, the results highlight the potential benefit of early introduction of EVE + EXE in the management of HR+, HER2− advanced breast cancer in postmenopausal patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The majority of breast cancers are hormone-receptor-positive (HR+), with up to 75 % expressing estrogen receptors and/or progesterone receptors [1, 2], whereas human epidermal growth factor receptor-2 (HER2) is overexpressed in approximately 15–23 % of breast cancers [3]. Thus, the majority of breast cancers are HR+ and HER2-negative (HER2−). Endocrine therapy, particularly aromatase inhibitors (AIs), represents the principal systemic therapy for postmenopausal women with HR+, HER2− breast cancer in both the adjuvant and advanced settings [4, 5]. Nonetheless, disease progression may occur despite continued endocrine therapy (also referred to clinically as endocrine resistance) [6, 7], and has been attributed, among other mechanisms, to the cross-talk between estrogen receptor signaling and the phosphatidylinositol 3-kinase (PI3K)/protein kinase B (AKT)/mammalian target of rapamycin (mTOR) pathway, which is essential for breast cancer growth, progression, and insensitivity to endocrine interventions [7–9]. Thus, co-targeting both signaling pathways may enhance the effectiveness of endocrine therapy and improve outcomes in patients with HR+, HER2− advanced breast cancer [7, 10].
The mTOR inhibitor everolimus (EVE) in combination with exemestane (EXE) was recently approved for the treatment of postmenopausal women with HR+, HER2− advanced breast cancer whose disease recurred or progressed during or after nonsteroidal AI (NSAI) therapy [11]. This approval was based on the results of the pivotal BOLERO-2 trial, wherein EVE + EXE more than doubled median progression-free survival (PFS) versus placebo (PBO) plus EXE at 18 months median follow-up [12], while maintaining health-related quality of life (HRQoL) [13]. Moreover, consistent efficacy results were observed in all predefined subgroups [12], including patients with visceral metastases, patients with bone disease, elderly patients, and Asian patients [14–17].
The present exploratory analyses evaluated the efficacy, safety, and HRQoL effects of EVE + EXE in the subgroup of patients in the BOLERO-2 trial who received this regimen as first-line therapy for advanced disease.
Methods
Study design and patient population
The BOLERO-2 trial is an international, phase 3, multicenter, randomized, double-blind, PBO-controlled trial (ClinicalTrials.gov identifier NCT00863655) that has been described in detail previously [18]. In brief, postmenopausal women with HR+, HER2−, unresectable, locally advanced or metastatic breast cancer recurring or progressing during or after letrozole or anastrozole were randomized at a 2:1 ratio to receive either EVE (10 mg daily) or matching PBO in a blinded manner, with open-label EXE (25 mg daily). For simplicity, the PBO + EXE arm will henceforth be referred to as the control arm. Randomization was stratified by the presence or absence of visceral metastases and sensitivity to prior endocrine therapy [18]. Treatment continued until disease progression, development of unacceptable toxicity, or withdrawal of consent. Written informed consent was obtained from all patients before enrollment; trial-related approvals were obtained from the institutional review boards of participating centers, and the trial was conducted in accordance with the Good Clinical Practice guidelines, Declaration of Helsinki, and local regulations. The present analyses included patients who received EVE + EXE as first-line therapy for advanced disease. Patients in this subset may have received (neo)adjuvant and adjuvant therapy, or adjuvant therapy only as last therapy before study entry.
Primary and secondary endpoints
Here we report the results of a retrospective and exploratory analysis from the BOLERO-2 trial. The primary endpoint of BOLERO-2 was investigator-assessed PFS (defined as time from randomization to first documented progression or death from any cause) per Response Evaluation Criteria in Solid Tumors (RECIST), v1.0. Secondary endpoints included safety, and HRQoL using European Organization for Research and Treatment of Cancer QLQ-C30 and BR23 questionnaires [18]. The present analysis evaluated investigator-assessed and centrally assessed PFS according to RECIST, v1.0. As with the overall study, adverse events (AEs) were recorded throughout the trial and were graded according to the Common Terminology Criteria for Adverse Events, v3.0.
Statistical analyses
The subset analyses reported in this manuscript are retrospective and exploratory. Estimates of PFS were obtained by Kaplan–Meier method, and hazard ratios and 95 % confidence intervals (CIs) were calculated using an unstratified Cox proportional hazards model. Time to definitive deterioration (TDD) of the Global Health Status was defined as a 5 % decrease in HRQoL relative to baseline, with no subsequent increase above this threshold, and was estimated by Kaplan–Meier method; hazard ratios and 95 % CIs were calculated using an unstratified Cox proportional hazards model. All analyses were conducted using SAS® for Windows, v9.2 (SAS Institute, Cary, NC, USA). All analyses were based on data at 18 months median follow-up.
Results
Demographics and disposition
Between June 2009 and January 2011, 724 women across 189 centers in 24 countries were randomized to the BOLERO-2 trial treatment arms (EVE + EXE, n = 485; control, n = 239) [18]. Patient baseline characteristics were well balanced between treatment groups and have been reported previously [18]. Overall, 19 % of patients (137 of 724) entered the trial having received (neo)adjuvant therapy as their last systemic treatment before study entry. These patients received EVE + EXE (100 of 485) or control (37 of 239) as first-line treatment for advanced breast cancer. Baseline characteristics were well balanced between the two treatment arms in this subset, including the presence of visceral metastases (EVE + EXE, 50 %; control, 43 %), bone metastases (EVE + EXE, 65 %; control, 70 %), and bone-only metastases (EVE + EXE, 29 %; control, 24 %) at baseline (Table 1).
Efficacy
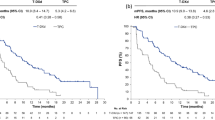
The efficacy data from this subset were consistent with outcomes in the overall trial population [12, 13]. Median PFS was nearly tripled with EVE + EXE versus control by local investigator assessment (11.5 vs 4.1 months, respectively; hazard ratio = 0.39; 95 % CI 0.25–0.62) in patients whose disease recurred during or after (neo)adjuvant therapy. Median PFS assessed by independent central review (15.2 vs 4.2 months, respectively; hazard ratio = 0.32; 95 % CI 0.18–0.57) was consistent with local assessment (Fig. 1a, b).
Kaplan–Meier curves of PFS in patients whose disease recurred during/after adjuvant therapy [includes patients who also received (neo)adjuvant therapy] by a local assessment and b central assessment. CI Confidence interval, EVE everolimus, EXE exemestane, HR hazard ratio, mo month(s), PBO placebo, wk week(s)
Quality of life
In patients who received EVE + EXE as first-line therapy for advanced disease, baseline mean Global Health Status scores were similar between treatment arms (62.8 vs 63.4). Median TDD in Global Health Status was numerically longer with EVE + EXE versus control (11.1 vs 7.2 months, respectively; hazard ratio = 0.69; 95 % CI 0.39–1.22; Fig. 2).
Kaplan–Meier curves of TDD (5 % decrease in HRQoL relative to baseline) in Global Health Status in patients whose disease recurred during/after adjuvant therapy [includes patients who also received (neo)adjuvant therapy]. CI Confidence interval, EVE everolimus, EXE exemestane, HR hazard ratio, mo month(s), PBO placebo, wk week(s)
Safety
The safety profile of EVE + EXE in this subset analysis was consistent with that of the overall patient population [12]. The majority of AEs were mild to moderate in intensity (i.e., grade 1 or 2) and manageable. The most frequently reported AEs of any grade with EVE + EXE were stomatitis (68 %), diarrhea (40 %), and rash (37 %; Table 2). Among the most frequently reported any grade AEs, the most common AEs of grade 3 or 4 intensity with EVE + EXE were hyperglycemia (8 %), stomatitis (4 %), diarrhea (4 %), and fatigue (3 %). Treatment discontinuation due to AEs was slightly higher with EVE + EXE (10 %) compared with control (8 %). In the EVE + EXE arm, 74 % of patients had one or more EVE dose reductions or interruptions and 23 % had one or more EXE dose reductions or interruptions. In the control arm, 32 % of patients had one or more PBO dose reductions or interruptions and 19 % had one or more EXE dose reductions or interruptions. The majority of dose reductions or interruptions for both EVE and EXE were due to AEs.
Notably, the median duration of exposure to EVE in this subset was 31.1 weeks (range 1.0–109.4 weeks), which is substantially longer than the median duration of exposure of 23.9 weeks in the overall patient population (range 1.0–123.3 weeks) [12]. However, the median relative dose intensity of EVE in this subset (85 %) was comparable to that in the overall population (86 %).
Discussion
In the overall BOLERO-2 trial population, EVE + EXE more than doubled the median PFS compared with control (local assessment 7.8 vs 3.2 months, respectively; hazard ratio = 0.45; log-rank P < 0.0001) without compromising HRQoL (confirmed by central assessment 11.0 vs 4.1 months, respectively; hazard ratio = 0.38; log-rank P < 0.0001) [12]. The present subset analysis from BOLERO-2 demonstrated that EVE + EXE as first-line therapy for advanced breast cancer nearly tripled PFS in patients with HR+, HER2− advanced breast cancer previously treated with (neo)adjuvant NSAIs.
Notably, guidelines from the 1st International Consensus Conference for Advanced Breast Cancer (ABC1), European Society for Medical Oncology (ESMO), and National Comprehensive Cancer Network (NCCN) generally recommend multiple lines of endocrine therapy before switching to chemotherapy for patients with HR+ advanced breast cancer [4, 5, 19]. The German Arbeitsgemeinschaft Gynäkologische Onkologie e.V. (AGO) and Canadian consensus guidelines specifically recommend EVE + EXE, fulvestrant, tamoxifen, or EXE for patients who have recurred during or after adjuvant AI therapy, with EVE + EXE reserved for shorter treatment-free intervals (i.e., no longer endocrine sensitive) [20, 21].
First-line treatment of advanced breast cancer with AIs (EXE, letrozole, or anastrozole) has demonstrated superior efficacy compared with tamoxifen, substantially prolonging median time to progression (TTP)/PFS durations (9.9/10.7 vs 5.8/6.4 months) in postmenopausal women with HR+ or hormone-receptor status unknown, locally advanced or recurrent disease without prior exposure to NSAIs [22–24]. However, endocrine therapy in patients with HR+, HER2− advanced breast cancer previously exposed to NSAIs, whether in the adjuvant or advanced disease setting, generally results in shorter median TTP or PFS. For example, in the EFECT trial, median TTP was 3.7 months for both fulvestrant (250 mg q 4 weeks) and EXE (P = 0.65) [6]. In addition, median PFS was 3–5 months in the SoFEA trial (4.4 months for fulvestrant 250 mg q 4 weeks + anastrozole vs 4.8 months for fulvestrant 250 mg q 4 weeks vs 3.4 months for EXE; P = 0.56–0.98) and 6–7 months in CONFIRM (6.5 months for fulvestrant 500 mg q 4 weeks and 5.5 months for fulvestrant 250 mg q 4 weeks; P = 0.006) [25, 26]. Increasingly, NSAIs have become standard of care in the adjuvant setting. Thus, although both EXE and fulvestrant may be used for breast cancer recurring after adjuvant NSAI therapy, the efficacy of these treatments is limited, and additional effective first-line treatment options are needed for these patients, especially those whose disease progressed during/after NSAI therapy. In this context, data from the present subset analysis provide support for the efficacy of EVE + EXE as first-line therapy in HR+, HER2− breast cancer progressing on adjuvant NSAIs, and suggest that earlier use of this strategy in the treatment course may lead to greater benefit.
Combinations of endocrine therapies with targeted agents in the first-line setting for advanced breast cancer progressing on NSAIs have shown mixed results. The histone deacetylate inhibitor entinostat plus EXE increased median PFS by 2 months versus EXE alone in patients whose disease recurred after adjuvant NSAI treatment or progressed after first-line NSAI (4.3 vs 2.3 months, P = 0.055) [27]. In the phase 3 HORIZON trial, temsirolimus, an mTOR inhibitor, plus letrozole versus letrozole alone failed to improve PFS as first-line therapy in patients with AI-naive advanced breast cancer [28]. It was suggested that the cyclic dosing regimen (30 mg/day for 5 days/2 weeks) used for oral temsirolimus [28] may not have achieved adequate suppression of mTOR activity as observed with a continuous dosing regimen [29]. In addition, the study populations in the BOLERO-2 and HORIZON trials had a different prior AI exposure. In fact, approximately 60 % of patients in HORIZON were endocrine therapy-naive at study entry [29]. In contrast, the TAMRAD (a phase 2 study of EVE plus tamoxifen vs tamoxifen alone in patients with HR+, HER2− advanced breast cancer after AI therapy) and BOLERO-2 [8, 18] study populations only included patients who progressed on a prior AI.
The data presented here demonstrate that HRQoL is maintained even though a higher rate of AEs was observed with EVE + EXE compared with control. In addition, proactive monitoring and management of AEs are recommended in patients treated with EVE + EXE to help further optimize clinical benefit. Data from the present exploratory analysis indicate that EVE + EXE may be an effective and tolerable first-line therapy for advanced breast cancer after (neo)adjuvant NSAI treatment. Moreover, EVE + EXE could offer an additional first-line treatment option, providing PFS prolongation beyond the duration that would be expected with endocrine therapy alone, which is currently the recommended standard of care in this patient population [4, 5]. Limitations of this subset analysis include its retrospective and exploratory nature and modest sample size. Analysis of outcomes based on prior use of endocrine therapy only versus endocrine therapy plus chemotherapy in this subset also was not considered feasible because of the small sample size and the risk of creating an imbalance between treatment arms in further subanalyses.
These data raise a possible hypothesis that EVE + EXE combination therapy may be more effective in patients exposed to multiple lines of endocrine therapy, including those whose only prior therapy was an NSAI in the adjuvant setting. Specifically, the disease may be using alternative pathways, such as the mTOR pathway, to continue cancer cell growth and proliferation in the presence of endocrine interventions. These alternative pathways are less likely to be active in patients with minimal or no prior exposure to systemic endocrine therapy [29, 30]. Nonetheless, disease relapse during standard adjuvant endocrine therapy remains a concern, and might involve similar signaling pathways. Several prospective trials are also evaluating the effectiveness of EVE in improving disease-free survival and overall survival when used in combination with endocrine therapy in the adjuvant setting. For example, the SWOG/NSABP S1207 study is a phase 3 randomized trial that is evaluating the efficacy of 1 year of EVE plus endocrine therapy in patients with high-risk early HR+, HER2− breast cancer [31, 32]. The UNIRAD study by the UNICANCER group is evaluating the efficacy of EVE when administered concurrently with endocrine therapy after 3 years of adjuvant endocrine therapy in a similar study population [33, 34].
Based on data from BOLERO-2, recent NCCN and Canadian Consensus clinical practice guidelines have recommended EVE + EXE therapy for HR+, HER2− advanced breast cancer recurring/progressing during or after an NSAI, even in the presence of visceral metastases [19, 21]; however, it should be noted that these guidelines are not specific to progression during or after adjuvant therapy. Furthermore, an ongoing multicenter, open-label, single-arm, phase 2 trial (BOLERO-4) is prospectively evaluating the efficacy of EVE plus letrozole as first-line therapy in patients with HR+, HER2− advanced breast cancer [35], and may provide additional insight into the efficacy of EVE plus an AI as first-line treatment for advanced disease. Notably, BOLERO-4 also is evaluating measures to proactively manage common AEs (e.g., stomatitis) during EVE treatment to maintain continuity of therapy. In addition, the promising efficacy of EVE in managing advanced breast cancer has resulted in the ongoing SWOG and UNIRAD trials (discussed earlier) evaluating the efficacy of adding EVE to adjuvant endocrine therapy for reducing the risk of disease recurrence in patients with high-risk early breast cancer [31–34].
Conclusions
In the BOLERO-2 trial, the benefit of adding EVE to EXE observed in the subset of patients whose disease progressed during or after (neo)adjuvant NSAI therapy was consistent with that observed in the overall population. Furthermore, the substantial improvement in PFS in this subset was accomplished while maintaining HRQoL. The safety and tolerability profile of EVE in this subset analysis also was similar to that observed in the overall BOLERO-2 population and with prior experience in the oncology setting. These data support the efficacy of EVE + EXE as first-line therapy for advanced breast cancer in patients with recurrence on adjuvant NSAI therapy.
Abbreviations
- ABC1:
-
1st International Consensus Conference for Advanced Breast Cancer
- AE:
-
Adverse event
- AGO:
-
Arbeitsgemeinschaft Gynäkologische Onkologie e.V
- AI:
-
Aromatase inhibitor
- AKT:
-
Protein kinase B
- ANA:
-
Anastrozole
- CI:
-
Confidence interval
- ECOG:
-
Eastern Cooperative Oncology Group
- ESMO:
-
European Society for Medical Oncology
- EVE:
-
Everolimus
- EXE:
-
Exemestane
- HER2− :
-
Human epidermal growth factor receptor-2-negative
- LET:
-
Letrozole
- HR:
-
Hazard ratio
- HR+ :
-
hormone-receptor-positive
- HRQoL:
-
Health-related quality of life
- LHRHa:
-
Luteinizing hormone-releasing hormone analogue
- mTOR:
-
Mammalian target of rapamycin
- NCCN:
-
National Comprehensive Cancer Network
- NSAI:
-
Nonsteroidal aromatase inhibitor
- PBO:
-
Placebo
- PFS:
-
Progression-free survival
- PI3K:
-
Phosphatidylinositol 3-kinase
- RECIST:
-
Response Evaluation Criteria in Solid Tumors
- TDD:
-
Time to definitive deterioration
- TTP:
-
Time to progression
References
Chumsri S, Howes T, Bao T, Sabnis G, Brodie A (2011) Aromatase, aromatase inhibitors, and breast cancer. J Steroid Biochem Mol Biol 125(1–2):13–22. doi:10.1016/j.jsbmb.2011.02.001
Nadji M, Gomez-Fernandez C, Ganjei-Azar P, Morales AR (2005) Immunohistochemistry of estrogen and progesterone receptors reconsidered: experience with 5,993 breast cancers. Am J Clin Pathol 123(1):21–27. doi:10.1309/4WV79N2GHJ3X1841
Mohd Sharial MS, Crown J, Hennessy BT (2012) Overcoming resistance and restoring sensitivity to HER2-targeted therapies in breast cancer. Ann Oncol 23(12):3007–3016. doi:10.1093/annonc/mds200
Cardoso F, Costa A, Norton L, Cameron D, Cufer T, Fallowfield L, Francis P, Gligorov J, Kyriakides S, Lin N, Pagani O, Senkus E, Thomssen C, Aapro M, Bergh J, Di Leo A, El Saghir N, Ganz PA, Gelmon K, Goldhirsch A, Harbeck N, Houssami N, Hudis C, Kaufman B, Leadbeater M, Mayer M, Rodger A, Rugo H, Sacchini V, Sledge G, van’t Veer L, Viale G, Krop I, Winer E (2012) 1st International consensus guidelines for advanced breast cancer (ABC 1). Breast 21(3):242–252. doi:10.1016/j.breast.2012.03.003
Cardoso F, Harbeck N, Fallowfield L, Kyriakides S, Senkus E (2012) Locally recurrent or metastatic breast cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 23(Suppl 7):vii11–19. doi:10.1093/annonc/mds232
Chia S, Gradishar W, Mauriac L, Bines J, Amant F, Federico M, Fein L, Romieu G, Buzdar A, Robertson JF, Brufsky A, Possinger K, Rennie P, Sapunar F, Lowe E, Piccart M (2008) Double-blind, randomized placebo controlled trial of fulvestrant compared with exemestane after prior nonsteroidal aromatase inhibitor therapy in postmenopausal women with hormone receptor-positive, advanced breast cancer: results from EFECT. J Clin Oncol 26(10):1664–1670. doi:10.1200/JCO.2007.13.5822
Gnant M (2013) The role of mammalian target of rapamycin (mTOR) inhibition in the treatment of advanced breast cancer. Curr Oncol Rep 15(1):14–23. doi:10.1007/s11912-012-0277-1
Bachelot T, Bourgier C, Cropet C, Ray-Coquard I, Ferrero JM, Freyer G, Abadie-Lacourtoisie S, Eymard JC, Debled M, Spaeth D, Legouffe E, Allouache D, El Kouri C, Pujade-Lauraine E (2012) Randomized phase II trial of everolimus in combination with tamoxifen in patients with hormone receptor-positive, human epidermal growth factor receptor 2-negative metastatic breast cancer with prior exposure to aromatase inhibitors: a GINECO study. J Clin Oncol 30(22):2718–2724. doi:10.1200/JCO.2011.39.0708
Boulay A, Rudloff J, Ye J, Zumstein-Mecker S, O’Reilly T, Evans DB, Chen S, Lane HA (2005) Dual inhibition of mTOR and estrogen receptor signaling in vitro induces cell death in models of breast cancer. Clin Cancer Res 11(14):5319–5328. doi:10.1158/1078-0432.CCR-04-2402
Gnant M (2012) Overcoming endocrine resistance in breast cancer: importance of mTOR inhibition. Expert Rev Anticancer Ther 12(12):1579–1589. doi:10.1586/era.12.138
Novartis (2012) Afinitor (everolimus) prescribing information. Novartis Pharmaceuticals Corporation, East Hanover, NJ
Yardley DA, Noguchi S, Pritchard KI, Burris HA III, Baselga J, Gnant M, Hortobagyi GN, Campone M, Pistilli B, Piccart M, Melichar B, Petrakova K, Arena FP, Erdkamp F, Harb WA, Feng W, Cahana A, Taran T, Lebwohl D, Rugo HS (2013) Everolimus plus exemestane in postmenopausal patients with HR+ breast cancer: BOLERO-2 final progression-free survival analysis. Adv Ther 30(10):870–884. doi:10.1007/s12325-013-0060-1
Burris HA 3rd, Lebrun F, Rugo HS, Beck JT, Piccart M, Neven P, Baselga J, Petrakova K, Hortobagyi GN, Komorowski A, Chouinard E, Young R, Gnant M, Pritchard KI, Bennett L, Ricci JF, Bauly H, Taran T, Sahmoud T, Noguchi S (2013) Health-related quality of life of patients with advanced breast cancer treated with everolimus plus exemestane versus placebo plus exemestane in the phase 3, randomized, controlled, BOLERO-2 trial. Cancer 119(10):1908–1915. doi:10.1002/cncr.28010
Campone M, Bachelot T, Gnant M, Deleu I, Rugo HS, Pistilli B, Noguchi S, Shtivelband M, Pritchard KI, Provencher L, Burris HA 3rd, Hart L, Melichar B, Hortobagyi GN, Arena F, Baselga J, Panneerselvam A, Heniquez A, El-Hashimy M, Taran T, Sahmoud T, Piccart M (2013) Effect of visceral metastases on the efficacy and safety of everolimus in postmenopausal women with advanced breast cancer: subgroup analysis from the BOLERO-2 study. Eur J Cancer 49(12):2621–2632. doi:10.1016/j.ejca.2013.04.011
Gnant M, Baselga J, Rugo HS, Noguchi S, Burris HA, Piccart M, Hortobagyi GN, Eakle J, Mukai H, Iwata H, Geberth M, Hart LL, Hadji P, El-Hashimy M, Rao S, Taran T, Sahmoud T, Lebwohl D, Campone M, Pritchard KI (2013) Effect of everolimus on bone marker levels and progressive disease in bone in BOLERO-2. J Natl Cancer Inst 105(9):654–663. doi:10.1093/jnci/djt026
Noguchi S, Masuda N, Iwata H, Mukai H, Horiguchi J, Puttawibul P, Srimuninnimit V, Tokuda Y, Kuroi K, Iwase H, Inaji H, Ohsumi S, Noh WC, Nakayama T, Ohno S, Rai Y, Park BW, Panneerselvam A, El-Hashimy M, Taran T, Sahmoud T, Ito Y (2013) Efficacy of everolimus with exemestane versus exemestane alone in Asian patients with HER2-negative, hormone-receptor-positive breast cancer in BOLERO-2. Breast Cancer. doi:10.1007/s12282-013-0444-8
Pritchard KI, Burris HA III, Ito Y, Rugo HS, Dakhil S, Hortobagyi GN, Campone M, Csöszi T, Baselga J, Puttawibul P, Piccart M, Heng D, Noguchi S, Srimuninnimit V, Bourgeois H, Gonzalez Martin A, Osborne K, Paneerselvam A, Taran T, Sahmoud T, Gnant M (2013) Safety and efficacy of everolimus with exemestane vs. exemestane alone in elderly patients with HER2-negative, hormone receptor-positive breast cancer in BOLERO-2. Clin Breast Cancer 13(6):421–432. doi:10.1016/j.clbc.2013.08.011
Baselga J, Campone M, Piccart M, Burris HA 3rd, Rugo HS, Sahmoud T, Noguchi S, Gnant M, Pritchard KI, Lebrun F, Beck JT, Ito Y, Yardley D, Deleu I, Perez A, Bachelot T, Vittori L, Xu Z, Mukhopadhyay P, Lebwohl D, Hortobagyi GN (2012) Everolimus in postmenopausal hormone-receptor-positive advanced breast cancer. N Engl J Med 366(6):520–529. doi:10.1056/NEJMoa1109653
National Comprehensive Cancer Network (2013) NCCN Clinical Practice Guidelines in Oncology: breast cancer screening and diagnosis. Version 3.2013. http://www.nccn.org/professionals/physician_gls/pdf/breast-screening.pdf. Accessed 26 Sep 2013
Arbeitsgemeinschaft Gynäkologische Onkologie (2013) Diagnosis and treatment of patients with primary and metastatic breast cancer: guidelines of the AGO Breast Committee. www.ago-online.de. Accessed 26 Sep 2013
Pritchard KI, Gelmon KA, Rayson D, Provencher L, Webster M, McLeod D, Verma S (2013) Endocrine therapy for postmenopausal women with hormone receptor-positive her2-negative advanced breast cancer after progression or recurrence on nonsteroidal aromatase inhibitor therapy: a Canadian consensus statement. Curr Oncol 20(1):48–61. doi:10.3747/co.20.1316
Bonneterre J, Buzdar A, Nabholtz JM, Robertson JF, Thurlimann B, von Euler M, Sahmoud T, Webster A, Steinberg M (2001) Anastrozole is superior to tamoxifen as first-line therapy in hormone receptor positive advanced breast carcinoma. Cancer 92(9):2247–2258
Cohen MH, Johnson JR, Li N, Chen G, Pazdur R (2002) Approval summary: letrozole in the treatment of postmenopausal women with advanced breast cancer. Clin Cancer Res 8(3):665–669
Paridaens RJ, Dirix LY, Beex LV, Nooij M, Cameron DA, Cufer T, Piccart MJ, Bogaerts J, Therasse P (2008) Phase III study comparing exemestane with tamoxifen as first-line hormonal treatment of metastatic breast cancer in postmenopausal women: the European Organisation for Research and Treatment of Cancer Breast Cancer Cooperative Group. J Clin Oncol 26(30):4883–4890. doi:10.1200/JCO.2007.14.4659
Di Leo A, Jerusalem G, Petruzelka L, Torres R, Bondarenko IN, Khasanov R, Verhoeven D, Pedrini JL, Smirnova I, Lichinitser MR, Pendergrass K, Garnett S, Lindemann JP, Sapunar F, Martin M (2010) Results of the CONFIRM phase III trial comparing fulvestrant 250 mg with fulvestrant 500 mg in postmenopausal women with estrogen receptor-positive advanced breast cancer. J Clin Oncol 28(30):4594–4600. doi:10.1200/JCO.2010.28.8415
Johnston SR, Kilburn LS, Ellis P, Dodwell D, Cameron D, Hayward L, Im YH, Braybrooke JP, Brunt AM, Cheung KL, Jyothirmayi R, Robinson A, Wardley AM, Wheatley D, Howell A, Coombes G, Sergenson N, Sin HJ, Folkerd E, Dowsett M, Bliss JM (2013) Fulvestrant plus anastrozole or placebo versus exemestane alone after progression on non-steroidal aromatase inhibitors in postmenopausal patients with hormone-receptor-positive locally advanced or metastatic breast cancer (SoFEA): a composite, multicentre, phase 3 randomised trial. Lancet Oncol 14(10):989–998. doi:10.1016/S1470-2045(13)70322-X
Yardley DA, Ismail-Khan RR, Melichar B, Lichinitser M, Munster PN, Klein PM, Cruickshank S, Miller KD, Lee MJ, Trepel JB (2013) Randomized phase II, double-blind, placebo-controlled study of exemestane with or without entinostat in postmenopausal women with locally recurrent or metastatic estrogen receptor-positive breast cancer progressing on treatment with a nonsteroidal aromatase inhibitor. J Clin Oncol 31(17):2128–2135. doi:10.1200/JCO.2012.43.7251
Wolff AC, Lazar AA, Bondarenko I, Garin AM, Brincat S, Chow L, Sun Y, Neskovic-Konstantinovic Z, Guimaraes RC, Fumoleau P, Chan A, Hachemi S, Strahs A, Cincotta M, Berkenblit A, Krygowski M, Kang LL, Moore L, Hayes DF (2013) Randomized phase III placebo-controlled trial of letrozole plus oral temsirolimus as first-line endocrine therapy in postmenopausal women with locally advanced or metastatic breast cancer. J Clin Oncol 31(2):195–202. doi:10.1200/JCO.2011.38.3331
Tabernero J, Rojo F, Calvo E, Burris H, Judson I, Hazell K, Martinelli E, Ramon y Cajal S, Jones S, Vidal L, Shand N, Macarulla T, Ramos FJ, Dimitrijevic S, Zoellner U, Tang P, Stumm M, Lane HA, Lebwohl D, Baselga J (2008) Dose- and schedule-dependent inhibition of the mammalian target of rapamycin pathway with everolimus: a phase I tumor pharmacodynamic study in patients with advanced solid tumors. J Clin Oncol 26(10):1603–1610. doi:10.1200/JCO.2007.14.5482
Dees EC, Carey LA (2013) Improving endocrine therapy for breast cancer: it’s not that simple. J Clin Oncol 31(2):171–173. doi:10.1200/JCO.2012.46.2655
ClinicalTrials.gov. S1207 hormone therapy with or without everolimus in treating patients with breast cancer. http://www.clinicaltrials.gov/ct2/show/record/NCT01674140. Accessed 26 Sep 2013
Chavez-Mac Gregor M, Barlow WE, Gonzalez-Angulo AM, Rastogi P, Mamounas EP, Ganz PA, Schott AF, Paik S, Lew DL, Bandos H, Hortobagyi GN (2012) S1207: phase III randomized, placebo-controlled clinical trial evaluating the use of adjuvant endocrine therapy ± one year of everolimus in patients with high-risk, hormone receptor-positive and HER2-neu negative breast cancer (NCT01674140). Presented at San Antonio Breast Cancer Symposium; December 4–8, 2012; San Antonio, TX, USA. Poster OT2-2-04
ClinicalTrials.gov. Safety study of adding everolimus to adjuvant hormone therapy in women with poor prognosis, ER+ and HER2− primary breast cancer, free of disease after receiving 3 years of adjuvant hormone therapy. http://www.clinicaltrials.gov/ct2/show/NCT01805271. Accessed 26 Sep 2013
Bachelot T, Chabaud S, Martin A-L, Lemonnier J, Bliss J, Cameron D, Hardy Bessard A-C, Campone M, Andre F (2013) Everolimus plus adjuvant endocrine therapy in high risk breast cancer: the UNIRAD study. Presented at American Society of Clinical Oncology (ASCO); May 31–June 4, 2013; Chicago, IL, USA. Poster/abstract TPS653
ClinicalTrials.gov. Open-label, phase II, study of everolimus plus letrozole in postmenopausal women with ER+ metastatic breast cancer (BOLERO-4). http://clinicaltrials.gov/ct2/show/NCT01698918?term=NCT+01698918&rank=1. Accessed 26 Sep 2013
Acknowledgments
We thank the patients who participated in the BOLERO-2 trial and the investigators, study nurses, and clinical research associates from the individual trial centers who provided ongoing support. We thank Duprane Pedaci Young, PhD, ProEd Communications, Inc., and Avishek Pal, MSc, Novartis Healthcare Pvt. Ltd., for providing medical editorial assistance with this manuscript. Financial support for these analyses and for medical editorial assistance was provided by Novartis Pharmaceuticals.
Conflict of interest
T. Brechenmacher, M. El-Hashimy, S. Douma, and F. Ringeisen are employees of Novartis. M. Campone reports a consultant/advisory role with and remuneration/honoraria from Novartis. I. Deleu reports clinical trial funding from Novartis. L. Hart reports research funding to his institution from Novartis. D. Heng reports a consultant/advisory role with Novartis. G. Hortobagyi reports a consultant/advisory role with and research funding from Novartis. B. Melichar reports a consultant/advisory role with and remuneration/honoraria from Novartis. M. Piccart reports research funding from Novartis. B. Pistilli reports a consultant/advisory role with Novartis. The other authors report no competing interests.
Ethical Standards
The BOLERO-2 trial was conducted in accordance with the Good Clinical Practice guidelines, the Declaration of Helsinki, and each country’s laws and regulations.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Beck, J.T., Hortobagyi, G.N., Campone, M. et al. Everolimus plus exemestane as first-line therapy in HR+, HER2− advanced breast cancer in BOLERO-2. Breast Cancer Res Treat 143, 459–467 (2014). https://doi.org/10.1007/s10549-013-2814-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-013-2814-5