Abstract
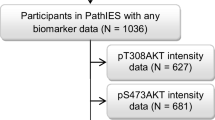
Results from the NSABP B-28 trial suggest AKT activation may predict reduced benefit from taxanes following standard anthracycline therapy. Pre-clinical data support a link between PI3 K/AKT signalling and taxane resistance. Using the UK taxotere as adjuvant chemotherapy trial (TACT), we tested the hypothesis that activation of AKT or downstream markers, p70S6K or p90RSK, identifies patients with reduced benefit from taxane chemotherapy. TACT is a multi-centre open-label phase III trial comparing four cycles of standard FEC (fluorouracil, epirubicin, cyclophosphamide) followed by four cycles of docetaxel versus eight cycles of anthracycline-based chemotherapy. Samples from 3,596 patients were available for the current study. We performed immunohistochemical analysis of activation of AKT, p70S6 K and p90RSK. Using a training set with multiple cut-offs for predictive values (10 % increments in expression), we found no evidence for a treatment by marker interaction for pAKT473, pS6 or p90RSK. pAKT473, pS6 and p90RSK expression levels were weakly correlated. A robust, preplanned statistical analysis in the TACT trial found no evidence that pAKT473, pS6 or p90RSK identifies patients deriving reduced benefit from adjuvant docetaxel. This result is consistent with the recent NASBP B28 study where the pAKT473 effect is not statistically significant for the treatment interaction test. Therefore, neither TACT nor NASBP-B28 provides statistically robust evidence of a treatment by marker interaction between pAKT473 and taxane treatment. Alternative methods for selecting patients benefitting from taxanes should be explored.


Similar content being viewed by others
References
Peto R (2002) Update of the worldwide evidence on the adjuvant treatment of breast cancer. Eur J Cancer 38:E11
Ellis P, Barrett-Lee P, Johnson L et al (2009) Sequential docetaxel as adjuvant chemotherapy for early breast cancer (TACT): an open-label, phase III, randomised controlled trial. Lancet 373(9676):1681–1692
Hayes DF, Thor AD, Dressler LG et al (2007) HER2 and response to paclitaxel in node-positive breast cancer. New Engl J Med 357(15):1496–1506
Yang SX, Costantino JP, Kim C et al (2010) Akt Phosphorylation at Ser473 Predicts Benefit of Paclitaxel Chemotherapy in Node-Positive Breast Cancer. J Clin Oncol 28(18):2974–2981
Hu LM, Hofmann J, Lu YL, Mills GB, Jaffe RB (2002) Inhibition of phosphatidylinositol 3 ‘-kinase increases efficacy of paclitaxel in in vitro and in vivo ovarian cancer models. Cancer Res 62(4):1087–1092
Calastretti A, Bevilacqua A, Ceriani C et al (2001) Damaged microtubules can inactivate BCL-2 by means of the mTOR kinase. Oncogene 20(43):6172–6180
Calastretti A, Rancati F, Vigano S et al (1999) Taxol-induced apoptosis and BCL-2 degradation inhibited by rapamycin. Clin Cancer Res 5:3814S
Page C, Lin HJ, Jin Y et al (2000) Overexpression of Akt/AKT can modulate chemotherapy-induced apoptosis. Anticancer Res 20(1A):407–416
Dillon RL, Marcotte R, Hennessy BT, Woodgett JR, Mills GB, Muller WJ (2009) Akt1 and Akt2 play distinct roles in the initiation and metastatic phases of mammary tumor progression. Cancer Res 69(12):5057–5064
Vanhaesebroeck B, Guillermet-Guibert J, Graupera M, Bilanges B (2010) The emerging mechanisms of isoform-specific PI3 K signalling. Nat Rev Mol Cell Biol 11(5):329–341
Hennessy BT, Smith DL, Ram PT, Lu Y, Mills GB (2005) Exploiting the PI3 K/AKT pathway for cancer drug discovery. Nat Rev Drug Discov 4(12):988–1004
Kirkegaard T, Witton CJ, Edwards J et al (2010) Molecular alterations in AKT1, AKT2 and AKT3 detected in breast and prostatic cancer by FISH. Histopathology 56(2):203–211
Stemke-Hale K, Gonzalez-Angulo AM, Lluch A et al (2008) Integrative genomic and proteomic analysis of PIK3CA, PTEN, and AKT mutations in breast cancer. Cancer Res 68(15):6084–6091
Ellis MJ, Tao Y, Lin L et al (2006) High nuclear p27(kip1) levels predicts sensitivity to neoadjuvant letrozole in ER plus breast cancer independent of pAKT levels, PIK3CA mutation status and cell cycle effects: a potential role for p27(kip1) in predicting enhanced autophagocytosis in response to aromatase inhibitor therapy. Breast Cancer Res Treat 100:S187
Tokunaga E, Kimura Y, Oki E et al (2006) Akt is frequently activated in HER2/neu-positive breast cancers and associated with poor prognosis among hormone-treated patients. Int J Cancer 118(2):284–289
Panigrahi AR, Pinder SE, Chan SY, Paish EC, Robertson JFR, Ellis IO (2004) The role of PTEN and its signalling pathways, including AKT, in breast cancer; an assessment of relationships with other prognostic factors and with outcome. J Pathol 204(1):93–100
MacKeigan JP, Taxman DJ, Hunter D, Earp HS, Graves LM, Ting JPY (2002) Inactivation of the antiapoptotic phosphatidylinositol 3- kinase-Akt pathway by the combined treatment of taxol and mitogen-activated protein kinase kinase inhibition. Clin Cancer Res 8(7):2091–2099
Ling X, Bernacki RJ, Brattain MG, Li FZ (2004) Induction of survivin expression by taxol (paclitaxel) is an early event, which is independent of taxol-mediated G(2)/M arrest. J Biol Chem 279(15):15196–15203
Le XF, Hittelman WN, Liu JX et al (2003) Paclitaxel induces inactivation of p70 S6 kinase and phosphorylation of Thr(421) and Ser(424) via multiple signaling pathways in mitosis. Oncogene 22(4):484–497
Workman P, Clarke PA, Raynaud FI, van Montfort RLM (2010) Drugging the PI3 kinome: from chemical tools to drugs in the clinic. Cancer Res 70(6):2146–2157
Copp J, Manning G, Hunter T (2009) TORC-specific phosphorylation of mammalian target of rapamycin (mTOR): phospho-Ser2481 is a marker for intact mTOR signaling complex 2. Cancer Res 69(5):1821–1827
Sabine VS, Sims AH, Macaskill EJ et al (2009) Transcriptional profiling of patient-matched ER-positive breast cancer tissue from post-menopausal women treated with neoadjuvant RAD001. Cancer Res 69(2):166S–167S
Kirkegaard T, Witton CJ, McGlynn LM et al (2005) AKT activation predicts outcome in breast cancer patients treated with tamoxifen. J Pathol 207(2):139–146
Ellis IO, Dowsett M, Bartlett J et al (2000) Recommendations for HER2 testing in the UK. J Clin Pathol 53(12):890–892
Dowsett M, Bartlett J, Ellis IO et al (2003) Correlation between immunohistochemistry (HercepTest) and fluorescence in situ hybridization (FISH) for HER-2 in 426 breast carcinomas from 37 centres. J Pathol 199(4):418–423
Ellis IO, Bartlett J, Dowsett M et al (2004) Updated recommendations for HER2 testing in the UK. J Clin Pathol 57(3):233–237
Walker RA, Bartlett JMS, Dowsett M et al (2008) HER2 testing in the UK: further update to recommendations. J Clin Pathol 61(7):818–824
Bartlett JMS, Ellis IO, Dowsett M et al (2007) Human Epidermal Growth Factor Receptor 2 Status Correlates With Lymph Node Involvement in Patients With Estrogen Receptor (ER) Negative, but With Grade in Those With ER-Positive Early-Stage Breast Cancer Suitable for Cytotoxic Chemotherapy. J Clin Oncol 25(28):4423–4430
McShane LM, Altman DG, Sauerbrei W, Taube SE, Gion M, Clark GM (2005) REporting recommendations for tumour MARKer prognostic studies (REMARK). Br J Cancer 93(4):387–391
Kirkegaard T, Edwards J, Tovey S et al (2006) Observer variation in immunohistochemical analysis of protein expression, time for a change? Histopathology 48(7):787–794
Royston P, Sauerbrei W (2008) Multivariable model-building. A pragmatic approach to regression analysis based on fractional polynomials for modeling continuous variables
Sarbassov DD, Ali SM, Sengupta S et al (2006) Prolonged rapamycin treatment inhibits mTORC2 assembly and AKT/PKB. Mol Cell 22(2):159–168
Sarbassov DD, Guertin DA, Ali SM, Sabatini DM (2005) Phosphorylation and Regulation of Akt/PKB by the Rictor-mTOR Complex. Science 307(5712):1098–1101
Buttrick GJ, Wakefield JG (2008) PI3-K and GSK-3 AKT-ing together with microtubules. Cell Cycle 7(17):2621–2625
Pinhel I, MacNeill F, Hills M et al (2010) Extreme loss of immunoreactive p-Akt and p-Erk1/2 during routine fixation of primary breast cancer. Breast Cancer Res 12(5):R76
Generali D, Fox SB, Brizzi MP et al (2008) Down-regulation of phosphatidylinositol 3 ‘-kinase/AKT/molecular target of rapamycin metabolic pathway by primary letrozole-based therapy in human breast cancer. Clin Cancer Res 14(9):2673–2680
Bandyopadhyay S, Sengupta TK, Fernandes DJ, Spicer EK (2003) Taxol- and okadaic acid-induced destabilization of bcl-2 mRNA is associated with decreased binding of proteins to a bcl-2 instability element. Biochem Pharmacol 66(7):1151–1162
Montgomery RB, Guzman J, O’Rourke DM, Stahl WL (2000) Expression of oncogenic epidermal growth factor receptor family kinases induces paclitaxel resistance and alters beta-tubulin isotype expression. J Biol Chem 275(23):17358–17363
Kavallaris M, Annereau JP, Barret JM (2008) Potential mechanisms of resistance to microtubule inhibitors. Semin Oncol 35(3):S22–S27
Chien AJ, Moasser MM (2008) Cellular mechanisms of resistance to anthracyclines and taxanes in cancer: intrinsic and acquired. Semin Oncol 35(2):S1–S14
Acknowledgements
This study was conducted with the support of the Ontario Institute for Cancer Research through funding provided by the Government of Ontario. The TACT trial is jointly funded by Cancer Research UK (CRUK/01/001) and educational Grants from Aventis, Roche and Pfizer. Storage, construction of microarrays and tissue HER2 testing were funded by an educational grant from Roche. This analysis was funded by a Cancer Research UK grant. Grateful thanks is due to all the patients who agreed to enter the TACT trial and donated their tumour tissue for this research. We also thank the many UK pathologists who sent tumour tissue samples to the central laboratories for storage and future biological research.
Conflict of interest
In the last year, Judith M Bliss has received support from Roche towards travel to attend the San Antonio Breast Cancer Symposium. Peter J Barrett-Lee has received honoraria in the past for advisory boards for Sanofi-Aventis and Pfizer. John MS Bartlett, Roger A’Hern, Tammy Piper, Ian O Ellis, Mitch Dowsett, Elizabeth A Mallon, David A Cameron, Stephen Johnston and Paul Ellis have no conflicts of interest.
Ethical standards
The experiments comply with the current laws of the United Kingdom.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
10549_2013_2489_MOESM1_ESM.doc
Supplementary Table 1: Table comparing patient characteristics of the overall trial and biomarker subset: N = number of cases per category; percent–percentage. All population = all patients eligible for the TACT trial. Biomarker sub-group = patients with available biomarker data. Non-biomarker sub-group = patients with no biomarker data either due to exclusion from tissue collection or missing data on biomarker analysis.(DOC 59 kb)
10549_2013_2489_MOESM2_ESM.doc
Supplementary Table 2: Treatment by marker interaction terms for breast cancer sub-groups: Interaction (treatment by marker) hazard ratios for tumour sub-groups defined as ER+/HER2−ve (ER positive and HER2 negative), HER2+ (HER2 positive and either ER positive or negative) and ER-/HER2-ve (ER negative and HER2 negative). A ratio of less than 1 indicates that the hazard ratio for taxane benefit is less in patients with biomarkers scores above the median. None of the effects reach statistical significance, but a majority of confidence intervals include values that would be of clinical interest (e.g. below 0.7 or above 1.4) if they represented the real effect size.(DOC 36 kb)
Rights and permissions
About this article
Cite this article
Bartlett, J.M.S., A’Hern, R., Piper, T. et al. Phosphorylation of AKT pathway proteins is not predictive of benefit of taxane therapy in early breast cancer. Breast Cancer Res Treat 138, 773–781 (2013). https://doi.org/10.1007/s10549-013-2489-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-013-2489-y