Abstract
Social interactions require both the rapid processing of multifaceted socio-affective signals (e.g., eye gaze, facial expressions, gestures) and their integration with evaluations, social knowledge, and expectations. Researchers interested in understanding complex social cognition and behavior face a “black box” problem: What are the underlying mental processes rapidly occurring between perception and action and why are there such vast individual differences? In this review, we promote electroencephalography (EEG) microstates as a powerful tool for both examining socio-affective states (e.g., processing whether someone is in need in a given situation) and identifying the sources of heterogeneity in socio-affective traits (e.g., general willingness to help others). EEG microstates are identified by analyzing scalp field maps (i.e., the distribution of the electrical field on the scalp) over time. This data-driven, reference-independent approach allows for identifying, timing, sequencing, and quantifying the activation of large-scale brain networks relevant to our socio-affective mind. In light of these benefits, EEG microstates should become an indispensable part of the methodological toolkit of laboratories working in the field of social and affective neuroscience.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Goal of the Review
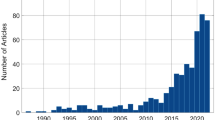
Imagine that you are in a line to board the bus to go pick up your child from kindergarten. As the bus pulls up, you notice an elderly woman down the sidewalk, burdened with shopping bags, preparing to cross the busy street. She asks you for help. You face a difficult choice. Do you help the elderly woman and miss the bus to pick up your child on time? Or do you board the bus and hope that someone else will come to her aid? This example demonstrates how social interaction requires the rapid processing of a variety of multifaceted socio-affective signals, such as eye gaze, facial expressions, gestures, physical contact, postures, and speech. Further, these signals must then be integrated with evaluations, social knowledge, and expectations stored in the brain. Researchers interested in understanding such complex social cognition and behavior are facing a “black box”: What are these distinct and rapid mental processes occurring between perceiving the bus situation described above and making the decision to help or not (e.g., conflict monitoring, neediness evaluation, planning behavior)? In addition, why might some people make the decision to help and others not? In this review, we aim to promote the analysis of electroencephalography (EEG) microstates (see Box 1) as an ideal approach for “opening the black box” and answering essential questions in the field of social and affective neuroscience. The microstate approach can be used to analyze both averaged EEG data (i.e., event-related potentials: ERPs; see Box 1) and non-averaged EEG data (i.e., continuous EEG; see Box 1), recorded at rest or during specific (socio-affective) states. EEG microstates provide unique information on the temporal dynamics of our socio-affective mind at a milliseconds timescale. Specifically, and in contrast to other approaches, the microstate approach allows for identifying, timing, sequencing, and quantifying the activation of large-scale brain networks that are associated with distinct socio-affective states (e.g., being stressed, because you might not be able to pick up your child on time in the example above) and that explain individual differences in socio-affective traits (e.g., general willingness to help others). The recent increase in publications indicates that more and more researchers recognize the potential of the EEG microstate approach in the fields of cognitive and clinical neuroscience (see Fig. 1; for reviews, see Galderisi and Mucci 2002; Khanna et al. 2015; Michel and Koenig 2018). However, we still see large, untapped potential for this analysis approach to better understand our socio-affective mind.
Number of microstate publications per year. The y-axis displays the number of publications as indexed in PubMed from 1987 to 2022 including the terms “EEG OR ERP AND microstates” (black bars) or the terms “(EEG OR ERP) AND (microstates AND (social OR affective))” (red bars) in the title, keywords, or abstract. Introduced by Dietrich Lehmann in 1987 (Lehmann et al. 1987), microstates initially drew limited attention within the EEG community as indexed by 126 publications over the next 25 years (1987–2012). In the last decade (2013–2022), more than 400 publications have followed. Note that only a minority (around 1/8) of all publications focused on applying microstates to ERP data
The Basis of EEG Microstates
The EEG microstate approach originates from the “brain mapping approach,” which analyzes the series of scalp field maps (see Box 1) over time. Scalp field maps represent the momentary spatial distribution of brain electrical fields (i.e., scalp topographies; for a more detailed description of the brain mapping approach, see Box 2). In analyzing scalp topographies in broad-band EEG,Footnote 1 Dietrich Lehmann and colleagues made a fundamental observation (Lehmann et al. 1987). Rather than randomly shifting over time, the scalp topographies tend to remain quasi-stable for a short period (during which the strength of the electric fields increases and decreases) and then change very quickly into a new topography, which then remains quasi-stable for another period. As different scalp topographies must have been produced by different configurations of generators in the brain (e.g., Vaughan 1982; Michel et al. 2004), it has been assumed that periods of quasi-stable topographies correspond to periods of synchronized activation of large-scale neural networks (e.g., Michel et al. 2009; Michel and Koenig 2018). Researchers reasoned that these periods may represent basic building blocks of information processing and named them “microstates”. This idea aligns with the contemporary view that brain functions involve information processing in widespread neural networks (e.g., Bressler and Menon 2010; He 2014; Fries 2015).
There are two basic steps to identify and analyze microstates, which are, despite slight methodological differences, similar for continuous EEG and ERPs (for a more detailed description, refer to Murray et al. 2008; Michel et al. 2009; Michel and Koenig 2018). First, one needs to reveal the most dominant topographies both within and across individuals by applying some sort of cluster or factor analysis (for details, see Murray et al. 2008; Michel et al. 2009). The optimal number of clusters are selected based on criteria that evaluate the quality of clustering (for an overview, see Murray 2009; Custo et al. 2017). The majority of continuous EEG studies have investigated four prototypical topographies (i.e., microstate classes) that are highly similar across studies. Though these microstate classes have been associated with activity of circumscribed neural networks, the specific function of each microstate class is still an active area of research (Michel and Koenig 2018; see Box 3 for an overview of the four prototypical microstate classes). Also note that recent research has trended to report cluster solutions with more than four classes (for a recent review, see Tarailis et al. 2023). Regarding ERPs, the number and topographies of the microstate classes depend on the task and the time window of analysis (for examples, see ERP studies listed in Table 1).
Second, the dominant topographies, which have been identified on the group-level, are then fit back to the original, individual EEG data for labeling each time point as the topography it correlates best with (in terms of GMD, see Box 1). Once the periods of quasi-stable topographies (i.e., microstates) are defined (usually lasting 60-120 ms for continuous EEG data: Brandeis et al. 1995, Michel and Koenig 2018; the duration of an ERP microstate is more variable and depends on the specific ERP component, ranging from very brief [< 100 ms] to several hundreds milliseconds in duration: Sur and Sinha 2009; Luck 2012), one can extract different parameters or features from each microstate class (see Box 4) to analyze associations with socio-affective traits or differences across experimental treatments or groups.
Benefits of EEG Microstates
Compared to EEG waveform analysis, the EEG microstate approach has three key benefits. First, the microstate approach is reference-independent by analyzing temporal dynamics of scalp field maps whose topographies do not depend on the location of the reference electrode. Thereby, microstate analysis avoids a potential source of bias (i.e., the choice of the “appropriate” reference) that critically affects the findings and replicability of EEG waveform analysis. Second, the microstate approach offers unique quantifications of the EEG data with potential neurophysiological relevance that are not available in typical waveform analyses (see the parameters listed in Box 4). Third, by considering information from all available electrodes and time points, it allows a comprehensive, data-driven approach. In waveform analysis, effects may go unnoticed (e.g., during periods of low amplitude) because analyses are restricted to a few specific time windows, ERP components, and electrode positions. Moreover, as these decisions are often flexibly based on self-selected “prior research” or subjective parameters discovered via the “visual inspection of the data,” they involve a high number of researcher degrees of freedom (Murray et al. 2008; Keil et al. 2014). In comparison, the data-driven approach of EEG microstates reduces these degrees of freedom, although deciding about the cluster solution can yet affect the reported results.
Applications of EEG Microstates
Applying the microstate approach to ERPs and to continuous EEG offers insights into the rapid neural network dynamics underlying our socio-affective mind regarding two main research goals: i) mapping the mind in action by examining socio-affective states; ii) mapping the individual mind by examining socio-affective traits. Table 1 provides an overview of studies applying the microstate approach in social and affective neuroscience. To delineate the scope of our review, we decided to focus on studies analyzing healthy participants. Overall, there are four main applications of EEG microstates, which are illustrated in Fig. 2.
Overview of the four main applications of EEG microstates. As the first application, one can “map the mind in action” by analyzing ERP microstates evoked by particular socio-affective information. Consider the bus example described at the beginning of this review. ERP microstate analysis opens the “black box” by identifying, timing, and sequencing mental processes occurring between hearing the request by the elderly woman and making the decision to help or not (e.g., neediness evaluation, conflict monitoring, planning behavior). One can then compare these processes’ characteristics across different socio-affective states, for instance, a non-stressed state vs. a stressed state. The second application is to reveal the “socio-affective mind in action” by comparing neural network dynamics extracted from continuous EEG data across a variety of socio-affective states, for instance, during discrete emotions, or during social exclusion vs. social inclusion. The third and fourth application of EEG microstates can be subsumed under the “neural trait approach,” i.e., “mapping the individual mind” by examining socio-affective traits (e.g., Schiller et al., 2019a; for a review, see Nash et al. 2014). Briefly, this approach indexes objective information from stable brain-based characteristics to reveal the sources of individual differences in socio-affective traits (e.g., behavioral intergroup bias, emotion detection ability, prosociality, tendency to deceive others, theory of mind). Microstate parameters from both ERP data (third application) and continuous EEG data (fourth application) are promising “neural trait candidates” as they possess high retest-reliabilities (for ERP: Jouen et al. 2021; for continuous EEG: Khanna et al. 2014; Liu et al. 2020; Schiller et al. 2020b; Antonova et al. 2022; Kleinert et al. 2023) and heritability (continuous EEG: da Cruz et al. 2020)
Mapping the Mind in Action: Socio-affective States
Socio-affective States and Microstate Analysis of ERPs (Application 1)
Microstate analysis of ERPs enables the researcher to identify, time, and sequence neurophysiological processes across distinct experimental conditions or trial types, such as anxiety vs. no-anxiety (Nash et al. 2023), conditioned fear vs. safety stimuli (Pizzagalli et al. 2003; Mueller and Pizzagalli 2016), direct vs. averted gaze (Burra et al. 2016), honest vs. dishonest decisions (Globig et al. 2023), ingroup- vs. outgroup-related information (Walker et al. 2008; Schiller et al. 2020a), less vs. more attractive faces (Han et al. 2020, 2022), self- vs. other-voice processing (Iannotti et al. 2022), social vs. non-social stimuli/contexts (Thierry et al. 2006; Ortigue et al. 2009, 2010; Cacioppo et al. 2012, 2015, 2016, 2018; Koban et al. 2012; Decety and Cacioppo 2012; Pegna et al. 2015), stereotype-congruent vs. stereotype-incongruent information (Schiller et al. 2016), stress vs. no stress (Schiller et al. 2023a) and neutral vs. emotional stimuli (Pizzagalli et al. 2000; Gianotti et al. 2007, 2008; Cacioppo et al. 2016; Tanaka et al. 2021; Zerna et al. 2021; Liang et al. 2022; Prete et al. 2022) (Fig. 3).
Mapping the mind in action. Examples of the application of the microstate approach in ERPs (application 1) and in continuous EEG (application 2) are shown on the left and right side, respectively. A ERPs are recorded while participants are confronted with specific socio-affective information (e.g., non-threatening faces vs. threatening faces, non-social vs. social stimuli). B Scalp topographies of 6 clusters in the sequence of their occurrence. C Microstates across time for two conditions plotted over Global Field Power (GFP). D Exemplary quantitative and qualitative differences between the two conditions. E Continuous EEG is recorded while participants are resting with their eyes closed. F Scalp topographies of the 4 prototypical microstate classes during continuous EEG: class A in green, class B in orange, class C in pink, and class D in violet. G Exemplary 2-s of microstates are shown for three individuals in two conditions (e.g., no stress vs. stress; happy mood vs. sad mood). All microstates belonging to class A are highlighted with a black frame. H Participants’ mean durations of microstate class A are shown as box plots for both conditions. The white diamond shape indicates the mean duration in the two conditions, the horizontal line the median. All figure panels are based on simulated data
Schiller et al. (2016) provide an example demonstrating quantitative differences across ERPs in the incongruent and the congruent condition of the Implicit Association Test (IAT), a widely-cited measure of implicit bias (Greenwald et al. 1998). This study addressed whether the IAT effect (i.e., longer response times when confronted with stereotype-incongruent information) is due to additional mental processes in the incongruent condition, or due to longer duration of the same processes. The authors identified seven microstates from stimulus presentation to response production that occurred in the same temporal sequence in both IAT conditions. However, participants showed a longer duration of one early-occurring microstate (starting around 220 ms after stimulus presentation) and of one late-occurring microstate (starting around 450 ms after stimulus presentation) in the incongruent condition, compared to the congruent condition. To shed light on the nature of the mental process underlying each microstate, the authors used source localization, linking the early microstate to lingual gyrus activity and the late microstate to activity in the middle cingulate cortex and posterior parietal areas. Based on these results, the authors suggested that the IAT effect is due to the prolongation of early-occurring perceptual processing as well as the implementation of late-occurring cognitive control, which is needed to select the correct motor response.
Response time differences in socio-affective behaviors, however, can also be due to qualitative processing differences. For example, Globig et al. (2023) analyzed ERPs during honest and dishonest social decision-making. Behaviorally, participants needed more time to lie than to tell the truth about the outcome of an incentivized card game. On the neural level, the authors found that the response time difference between the two conditions (lying and telling the truth) was the result of an additional microstate (occurring between 450 and 540 ms after stimulus presentation), unique to dishonest decisions, interrupting the antecedent microstate. Source localization indicated that the microstate unique to dishonest decisions was characterized by activity in the dorsolateral prefrontal cortex and the orbitofrontal cortex, whereas the antecedent microstate was characterized by activity in the supplementary motor areas. Based on the order of appearance and their pattern of neural activation, the authors speculated that when participants decide to lie, a unique process related to inhibiting the response selection process (i.e., telling the truth) occurred.
The benefit of the comprehensive ERP microstate analysis approach is also illustrated by research using classical conditioning paradigms to study brain mechanisms in the acquisition of social threat. Among the several studies on modulations of ERP components during fear conditioning, the latencies used for computing these components have varied greatly (Miskovic and Keil 2012; Ferreira de Sá et al. 2019; Sperl et al. 2021), with some focusing on early-latency (e.g., C1; Thigpen et al. 2017), others on mid-latency (e.g., N170; Camfield et al. 2016), and still others on late-latency components (e.g., LPP; Panitz et al. 2015, 2018; Bacigalupo and Luck 2018). However, as noted above, effects can easily be overlooked (e.g., during periods of low amplitude) if time windows for statistical ERP analyses are restricted to a few specific components and electrode positions (Murray et al. 2008). To overcome these methodological problems, two ERP studies applied the microstate approach to fear conditioning paradigms to help determine the electrophysiological signatures of responses to social threat (Pizzagalli et al. 2003; Mueller and Pizzagalli 2016). In these studies, the authors investigated ERPs elicited by socially-relevant conditioned stimuli (face stimuli) that had been paired with an unpleasant unconditioned stimulus (aversive noise). Critically, microstate analysis revealed that fear-conditioned faces modulated activity in visual brain regions within 80 ms after stimulus presentation (Mueller and Pizzagalli 2016). These early neural responses were overlooked in many prior studies that limited their analyses to specific (often late-latency) ERP components. This example illustrates how the comprehensive, data-driven nature of the microstate approach minimizes the risk of missing essential neurophysiological effects.
Socio-affective States and Microstate Analysis of Continuous EEG (Application 2)
Though relatively few in number at this point, recent work examining continuous EEG showcases the potential that microstates have for revealing the neural network dynamics of the socio-affective mind in action, at a millisecond scale, in several key ways (see Fig. 3).
First, microstate analysis can be combined with experimental designs (e.g., modulating the brain levels of hormones that regulate socio-affective processing: Schiller et al. 2019b; Zelenina et al. 2022; inducing psychosocial stress: Sikka et al. 2020; Kadier et al. 2021; Hu et al. 2021; performing social imitation tasks: Tomescu et al. 2022) to examine how manipulations causally change microstate parameters extracted from continuous EEG recorded after the experimental manipulation. For example, Schiller et al. (2019b) investigated the effects of experimentally increasing the availability of oxytocin in the nervous system by means of intranasal administration, compared to the administration of a placebo substance. The authors sought to illuminate the neurophysiological mechanisms underlying oxytocin’s well-known effects in the socio-affective domain (e.g., Meyer-Lindenberg et al. 2011; Ma et al. 2016). Globally, oxytocin increased stability (longer durations) across all four canonical resting networks, as recorded 45 min after substance administration (i.e., where most effective oxytocin effects are observed; see Spengler et al. 2017). Furthermore, oxytocin showed some microstate-class specific effects on further parameters, such as increasing the occurrence of microstate D (associated with attentional processing), decreasing the occurrence of microstate C (associated with interoceptive-automatic processing), and decreasing transitions from microstate B (associated with visual processing) to microstate C. The authors suggest that these results are in line with the anxiolytic effect of oxytocin to promote more attentional processing of external or social stimuli. Notably, Zelenina et al. (2022) conducted a similar study, examining the impact of oxytocin on neural network dynamics, but across a different time frame (i.e., continuous EEG was measured at various time windows, ranging from 15 to 100 min after substance administration). The results in this study partially mirror those in the Schiller et al. (2019b) study, demonstrating that, across time windows, oxytocin caused increased coverage and duration of microstates A and D, and decreased coverage of microstates B and C. The authors similarly reasoned that oxytocin seems to promote processes that tune the brain towards social stimuli.
Second, researchers have also manipulated socio-affective states using experimental manipulations administered across multiple sessions. For example, Zanesco and colleagues (2021) examined the impact of a 3-month meditation training on resting neural networks by means of microstate analysis (Zanesco et al. 2021b). Participants were randomly assigned to either the 3-month meditation training program or a wait-list control condition and resting EEG was recorded before and after training. Results showed that meditation increased mental awareness and quiescence, reflecting a kind of mental calm. Microstate analysis revealed six microstate classes in the continuous EEG, with four of these classes matching the prototypical microstate classes. Intriguingly, meditation training was also associated with decreased average duration across these microstates. The authors suggested that this decreased network stability reflected decreased cognitive control and increases in felt attentiveness and serenity. In a similar study, Bréchet et al. (2021) demonstrated significant topographical changes in EEG microstates in a quiet rest period after only 6 weeks of digital meditation training.
Third, microstate analysis can examine if different socio-affective states involve different neural network dynamics, as evidenced by unique changes to microstate features. For example, researchers have analyzed continuous EEG data recorded while participants are processing socio-affective information, such as watching emotional videos (e.g., Chen et al. 2021; Hu et al. 2022, 2023; Liu et al. 2023) or engaging in worship (Walter and Koenig 2022). For example, Hu et al. (2023) examined archival EEG data in which the original authors manipulated emotional states with the presentation of music videos. EEG was recorded during video presentation. The authors identified four microstate classes that had significant overlap with the four prototypical microstate classes. Importantly, while microstate C’s coverage and occurrence (associated with interoceptive-automatic processing) showed a positive relation with emotional arousal, microstate D (associated with attentional processing) occurred more often when watching videos of negative valence.
Fourth, microstate analysis can help examine mediators of subsequent behavior. For example, Pedroni et al. (2017) took the novel step of examining continuous EEG recorded during the inter-trial intervals in a risky decision-making task. On a behavioral level, results revealed that participants took higher risks after a winning trial than after a losing trial. On a neural level, microstate analysis of the inter-trial intervals revealed that two microstate classes (not referring to any of the prototypical resting EEG microstate classes) mediated the influence of outcomes of prior decisions on subsequent risk-taking on a trial-by-trial basis: one microstate class was associated with increased risk-taking, and a second microstate class was associated with decreased risk-taking. In other words, the two mediators act akin to a “gas pedal” and a “brake pedal”, respectively. Notably, the “brake” microstate class was source-localized to a bilateral prefrontal network, consistent with its role in self-regulation and cognitive control. This study demonstrates how microstate analysis can shed light on the socio-affective states that drive subsequent behavior on a single-trial level.
Mapping the Individual Mind: Socio-affective Traits
Socio-affective Traits and Microstate Analysis of ERPs (Application 3)
Another approach to study individual differences in socio-affective traits (e.g., behavioral intergroup bias, implicit intergroup attitudes, narcissism) is to analyze dispositional brain responses to specific socio-affective information, measured by ERPs (e.g., ingroup vs. outgroup words, Schiller et al. 2016; outcomes affecting ingroup vs. outgroup members, Schiller et al. 2020a; face attractiveness judgments, Han et al. 2022, see Fig. 4).
Mapping the individual mind. Examples of the application of the microstate approach in ERPs (application 3) and in continuous EEG (application 4) are shown on the left and right side, respectively. A ERPs are recorded while participants are viewing and responding to some specific socio-affective information (e.g., threatening faces, social stimuli). B Scalp topographies of the 6 clusters in the sequence of their occurrence. C Exemplary individual differences in microstates and response times across time plotted over Global Field Power (GFP). The hand symbols indicate mean response times. Arrows indicate the different durations of the third microstate. D Scatterplot of the association between duration of microstate 3 in milliseconds and the variable of interest (e.g., mean response time, indicated by the hands, in a particular condition of interest). E Continuous EEG is recorded while participants are resting with their eyes closed. F Scalp topographies of the four prototypical microstate classes during resting EEG: class A in green, class B in orange, class C in pink, and class D in violet. G Exemplary 2-s of microstates are shown for three individuals during resting condition. In panel G, all microstates belonging to class A are highlighted with a black frame. H Scatterplot of the association between the occurrence of microstate class A and the variable of interest (e.g., prosocial preferences, behavioral intergroup bias). All figure panels are based on simulated data
For example, recall Schiller et al.’s (2016) analysis of ERPs recorded during the IAT. This study revealed that response time differences in the IAT are mainly due to two microstates that take longer in the incongruent than in the congruent condition. However, what does this tell us about the mental processes underlying individual differences in implicit bias? In a next step, the authors checked whether the individual durations of these two microstates would contribute to individual differences in implicit bias. Indeed, they found that the longer an individual spent in the later occurring microstate when confronted with stereotype-incongruent information, the larger was this individual’s implicit bias. Based on the interpretation that this microstate reflected the implementation of cognitive control to select between the two competing response options, the authors suggest that individual differences in implicit bias are partly due to cognitive control ability. This finding illustrates how ERP microstate analysis can help to identify which specific features of neurophysiological processes drive heterogeneity in socio-affective traits.
Finally, one can also compare groups of people who already vary in socio-affective traits of interest. For example, Cacioppo et al. (2015) prescreened participants using a questionnaire on loneliness and, based on the scores, split participants into either a high or low loneliness group. These two groups then performed a Stroop task containing negative and positive words of either social or non-social relevance. Utilizing ERP microstate analysis, the authors demonstrated that individuals high in loneliness differentiate negative social stimuli from negative nonsocial stimuli more quickly than individuals low in loneliness (200 ms earlier, beginning at 280 ms after stimulus onset). Source localization analysis linked this early differentiation process to activity in brain regions associated with the orienting and executive control aspects of visual attention (e.g., extrastriate cortex, fusiform cortex, frontal eye field, dorsolateral prefrontal cortex, and anterior prefrontal cortex extending to the dorsal anterior cingulate). Relying on ERP microstates, this study demonstrated that loneliness seems to be associated with hypervigilance for social threats. This example illustrates how researchers can study individual differences in socio-affective traits by comparing millisecond brain dynamics across groups of people varying in these traits of interest.
Socio-affective Traits and Microstate Analysis of Continuous EEG (Application 4)
Microstate analysis of continuous EEG has been used to illuminate the sources of individual differences in socio-affective traits, in domains such as aggression (Kleinert and Nash 2022), anxiety (Schiller et al. 2019b; Du et al. 2022; Nash et al. 2023), approach vs. withdrawal tendency (Takehara et al. 2020; Kaur et al. 2020), disgust sensitivity (Li et al. 2021), empathy (Zhang et al. 2021), personality (Zanesco et al. 2020; Guo et al. 2020; Tomescu et al. 2022), prosociality (Schiller et al. 2020b), religious belief (Schlegel et al. 2012; Nash et al. 2022), or somatic awareness (Pipinis et al. 2017; Tarailis et al. 2021; Zanesco et al. 2021a). The majority of these studies have relied on regression or correlation analysis to uncover associations between features of the prototypical microstate classes (e.g., duration, occurrence, coverage, transition probabilities; see Box 4) and socio-affective traits (see Fig. 4).
For example, Schiller et al. (2020b) associated individual differences in trait prosociality with the prototypical resting-state microstates. They found that more prosocial individuals showed a higher coverage of microstate A and more transitions towards this microstate class from microstate C. The authors interpret these findings based on links of microstate A with auditory processing and microstate C with interoceptive-automatic processing (see Box 3). They suggest that more prosocial individuals might show a tendency to engage in bottom-up, sensory processing during rest, as well as a tendency to shift from stimulus-independent, top-down to more stimulus-dependent, bottom-up processing. They further suggest that these findings might be interpreted in light of the hypothesis that humans are intuitively cooperative and prosocial if they do not engage in time-consuming top-down processing during decision-making (e.g., Rand et al. 2014; but also see Kvarven et al. 2020). Overall, by identifying associations of trait prosociality with specific neural network dynamics at rest, this study illustrates how the microstate approach can help gain new insights into the sources of individual differences in socio-affective traits.
Individuals also possess more general and stable millisecond-level neural network dynamics that are independent of specific networks and that could essentially contribute to explain heterogeneity in socio-affective traits. For example, average durations and occurrences of microstates are inversely correlated across microstate classes (Khanna et al. 2014; Kleinert et al. 2022). These correlations suggest that individuals show a general tendency for more (i.e., fewer but longer-lasting network activations) or less (i.e., more but shorter-lasting network activations) network stability, potentially indicating the stability of one’s mental processing at rest. Following up on research (Kleinert et al. 2022) demonstrating a positive association between network stability and trait self-control, Kleinert and Nash (2022) found that individuals with higher levels of trait aggression (which is inversely related to self-control) showed less stable neural networks (indexed by shorter durations and more occurrences of microstates across microstate classes). In a related line of research, Tomescu et al. (2022) demonstrated more stable neural networks in individuals who are less neurotic, more conscientious, more extraverted, and report to have more coherent thoughts.
Finally, though less commonly applied, one can also illuminate heterogeneity in socio-affective traits by comparing neural network dynamics recorded at rest across two or more groups of people. This approach in particular makes sense for socio-affective traits that are more categorical in nature. For example, to illuminate why some individuals believe in deities and others do not, researchers have compared network features extracted from continuous EEG between a group of believers and a group of non-believers. They noted an increased coverage of a neural network associated with top-down processing (i.e., microstate D; Nash et al. 2022) and a decreased coverage of neural networks associated with bottom-up and interoceptive-automatic processing (i.e., microstate B, Schlegel et al. 2012; microstate C, Nash et al. 2022) in non-believers. These findings fit with the notion that believers show more intuitive reasoning and non-believers more analytic reasoning (e.g., Gervais and Norenzayan 2012). Future studies might apply this approach to investigate similarities and differences in milliseconds brain dynamics between other groups, such as people of different gender, conservatives vs. liberals, or single people vs. people in relationships.
Outlook and Future Directions
The research summarized above illustrates how microstates can reveal the temporal dynamics of our socio-affective mind. Taking this approach could help to shed light on a range of long-standing “black box” puzzles in the field of social and affective neuroscience. For example, failed attempts to replicate popular socio-affective phenomena (e.g., ego depletion, social priming) have left large gaps within the theoretical framework of affective and social sciences (e.g., Open Science Collaboration 2015; Vohs et al. 2021). To fill these gaps, it may be necessary to study the underlying neurophysiological and mental processes during these phenomena by means of the microstate approach. For example, do self-control demands lead to prolonged mental processes in subsequent tasks, indicating more cognitive difficulty? Does social priming speed up mental processing during the presentation of target stimuli? Beyond that, we see many more fascinating research questions that could be tackled by microstate research. Do adult minds process socio-affective information more quickly compared to those of adolescents or children? Do our brains—evolutionarily optimized for interacting “face-to-face” (e.g., Kock 2004)—process socio-affective cues differently in digital environments? Are neural networks activated in the same sequence at distinct states of consciousness (e.g., distinct sleep stages; Bréchet et al. 2020; Diezig et al. 2022)? Finally, EEG microstates might not only be useful for understanding our socio-affective mind, but also for modulating it. Preliminary evidence indicates that microstate-neurofeedback training is feasible in healthy participants (Diaz Hernandez et al. 2016; Asai et al. 2022). One could thus experimentally alter microstate features to induce specific socio-affective processes, e.g., experimentally increasing a key microstate’s duration at rest to decrease trait-levels of anxiety.
Conclusion
In sum, EEG microstate analysis offers a powerful tool for opening the “black box” of neurophysiological processing underlying our socio-affective mind. This review has illustrated how studies utilizing the microstate approach have illuminated the sources of socio-affective traits and examined distinct socio-affective states, highlighting issues of prosocial behavior, emotional processing, and social evaluations, as just a few examples. Given the major benefit afforded by EEG microstates for identifying, timing, sequencing, and quantifying the neural network dynamics underlying our socio-affective mind at a milliseconds time scale, the microstate approach has the potential to become an indispensable part of the methodological toolkit of laboratories working in the field of social and affective neuroscience.
Notes
Recent studies have also performed microstate analysis separately for different frequency bands, see, e.g., Férat et al. (2022).
References
Antonova E, Holding M, Suen HC, Sumich A, Maex R, Nehaniv C (2022) EEG microstates: functional significance and short-term test-retest reliability. Neuroimage Rep 2:100089. https://doi.org/10.1016/j.ynirp.2022.100089
Asai T, Hamamoto T, Kashihara S, Imamizu H (2022) Real-time detection and feedback of canonical electroencephalogram microstates: validating a neurofeedback system as a function of delay. Front Syst Neurosci 16:786200. https://doi.org/10.3389/fnsys.2022.786200
Bacigalupo F, Luck SJ (2018) Event-related potential components as measures of aversive conditioning in humans. Psychophysiology 55:e13015. https://doi.org/10.1111/psyp.13015
Brandeis D, Lehmann D, Michel CM, Mingrone W (1995) Mapping event-related brain potential microstates to sentence endings. Brain Topogr 8:145–159. https://doi.org/10.1007/BF01199778
Bréchet L, Brunet D, Perogamvros L, Tononi G, Michel CM (2020) EEG microstates of dreams. Sci Rep 10:17069. https://doi.org/10.1038/s41598-020-74075-z
Bréchet L, Ziegler DA, Simon AJ, Brunet D, Gazzaley A, Michel CM (2021) Reconfiguration of electroencephalography microstate networks after breath-focused, digital meditation training. Brain Connect 11:146–155. https://doi.org/10.1089/brain.2020.0848
Bressler SL, Menon V (2010) Large-scale brain networks in cognition: emerging methods and principles. Trends Cogn Sci 14:277–290. https://doi.org/10.1016/J.TICS.2010.04.004
Britz J, Van De Ville D, Michel CM (2010) BOLD correlates of EEG topography reveal rapid resting-state network dynamics. Neuroimage 52:1162–1170. https://doi.org/10.1016/j.neuroimage.2010.02.052
Burra N, Kerzel D, George N (2016) Early left parietal activity elicited by direct gaze: a high-density EEG study. PLoS ONE 11:e0166430. https://doi.org/10.1371/journal.pone.0166430
Buzsáki G, Anastassiou CA, Koch C (2012) The origin of extracellular fields and currents — EEG, ECoG, LFP and spikes. Nat Rev Neurosci 13:407–420. https://doi.org/10.1038/nrn3241
Cacioppo S, Balogh S, Cacioppo JT (2015) Implicit attention to negative social, in contrast to nonsocial, words in the Stroop task differs between individuals high and low in loneliness: Evidence from event-related brain microstates. Cortex 70:213–233. https://doi.org/10.1016/j.cortex.2015.05.032
Cacioppo S, Bangee M, Balogh S, Cardenas-Iniguez C, Qualter P, Cacioppo JT (2016) Loneliness and implicit attention to social threat: a high-performance electrical neuroimaging study. Cogn Neurosci 7:138–159. https://doi.org/10.1080/17588928.2015.1070136
Cacioppo S, Bolmont M, Monteleone G (2018) Spatio-temporal dynamics of the mirror neuron system during social intentions. Soc Neurosci 13:718–738. https://doi.org/10.1080/17470919.2017.1394911
Cacioppo S, Grafton S, Bianchi-Demicheli F (2012) The speed of passionate love, as a subliminal prime: a high-density electrical neuroimaging study. Neuroquantology 10:715–724. https://doi.org/10.14704/nq.2012.10.4.509
Camfield DA, Mills J, Kornfeld EJ, Croft RJ (2016) Modulation of the N170 with classical conditioning: the use of emotional imagery and acoustic startle in healthy and depressed participants. Front Hum Neurosci 10:337. https://doi.org/10.3389/fnhum.2016.00337
Chen J, Li H, Ma L, Bo H, Soong F, Shi Y (2021) Dual-threshold-based microstate analysis on characterizing temporal dynamics of affective process and emotion recognition from EEG signals. Front Neurosci 15:689791. https://doi.org/10.3389/fnins.2021.689791
Custo A, Van De Ville D, Wells WM, Tomescu MI, Brunet D, Michel CM (2017) Electroencephalographic resting-state networks: source localization of microstates. Brain Connect 7:671–682. https://doi.org/10.1089/brain.2016.0476
da Cruz JR, Favrod O, Roinishvili M, Chkonia E, Brand A, Mohr C, Figueiredo P, Herzog MH (2020) EEG microstates are a candidate endophenotype for schizophrenia. Nat Commun 11:3089. https://doi.org/10.1038/s41467-020-16914-1
Decety J, Cacioppo S (2012) The speed of morality: a high-density electrical neuroimaging study. J Neurophysiol 108:3068–3072. https://doi.org/10.1152/jn.00473.2012
Diaz Hernandez L, Rieger K, Baenninger A, Brandeis D, Koenig T (2016) Towards using microstate-neurofeedback for the treatment of psychotic symptoms in schizophrenia. A feasibility study in healthy participants. Brain Topogr 29:308–321. https://doi.org/10.1007/s10548-015-0460-4
Diezig S, Denzer S, Achermann P, Mast FW, Koenig T (2022) EEG microstate dynamics associated with dream-like experiences during the transition to sleep. Brain Topogr. https://doi.org/10.1007/s10548-022-00923-y
Du M, Peng Y, Li Y, Zhu Y, Yang S, Li J, Zou F, Wang Y, Wu X, Zhang Y, Zhang M (2022) Effect of trait anxiety on cognitive flexibility: evidence from event-related potentials and resting-state EEG. Biol Psychol 170:108319. https://doi.org/10.1016/j.biopsycho.2022.108319
Férat V, Seeber M, Michel CM, Ros T (2022) Beyond broadband: towards a spectral decomposition of electroencephalography microstates. Hum Brain Mapp 43:3047–3061. https://doi.org/10.1002/hbm.25834
Ferreira de Sá DS, Michael T, Wilhelm FH, Peyk P (2019) Learning to see the threat: temporal dynamics of ERPs of motivated attention in fear conditioning. Soc Cogn Affect Neurosci 14:189–203. https://doi.org/10.1093/scan/nsy103
Fries P (2015) Rhythms for cognition: communication through coherence. Neuron 88:220–235. https://doi.org/10.1016/J.NEURON.2015.09.034
Galderisi S, Mucci A (2002) Psychophysiology in psychiatry: new perspectives in the study of mental disorders. World Psychiatry 1:166–168
Gervais WM, Norenzayan A (2012) Analytic thinking promotes religious disbelief. Science 336:493–496. https://doi.org/10.1126/science.1215647
Gianotti LRR, Faber PL, Pascual-Marqui RD, Kochi K, Lehmann D (2007) Processing of positive versus negative emotional words is incorporated in anterior versus posterior brain areas: an ERP microstate Loreta study. Chaos Complexity Lett 2:189–211
Gianotti LRR, Faber PL, Schuler M, Pascual-Marqui RD, Kochi K, Lehmann D (2008) First valence, then arousal: the temporal dynamics of brain electric activity evoked by emotional stimuli. Brain Topogr 20:143–156. https://doi.org/10.1007/s10548-007-0041-2
Globig LK, Gianotti LRR, Ponsi G, Koenig T, Dahinden FM, Knoch D (2023) The path of dishonesty: identification of mental processes with electrical neuroimaging. Cereb Cortex 33:6693–6700. https://doi.org/10.1093/cercor/bhac535
Greenwald AG, McGhee DE, Schwartz JL (1998) Measuring individual differences in implicit cognition: the implicit association test. J Pers Soc Psychol 74:1464–1480. https://doi.org/10.1037/0022-3514
Guo P, Cui J, Wang Y, Zou F, Wu X, Zhang M (2020) Spontaneous microstates related to effects of low socioeconomic status on neuroticism. Sci Rep 10:15710. https://doi.org/10.1038/s41598-020-72590-7
Han S, Liu S, Gan Y, Xu Q (2020) Repeated exposure makes attractive faces more attractive: neural responses in facial attractiveness judgement. Neuropsychologia 139:107365. https://doi.org/10.1016/j.neuropsychologia.2020.107365
Han S, Hu J, Gao J, Fan J, Xu X, Xu P, Luo Y (2022) Comparisons make faces more attractive: An ERP study. Brain Behav 12:e2561. https://doi.org/10.1002/brb3.2561
He BJ (2014) Scale-free brain activity: past, present, and future. Trends Cogn Sci 18:480–487. https://doi.org/10.1016/J.TICS.2014.04.003
Hu N, Long Q, Li Q, Hu X, Li Y, Zhang S, Chen A, Huo R, Liu J, Wang X (2021) The modulation of salience and central executive networks by acute stress in healthy males: an EEG microstates study. Int J Psychophysiol 169:63–70. https://doi.org/10.1016/j.ijpsycho.2021.09.001
Hu W, Zhang Z, Zhang L, Huang G, Li L, Liang Z (2022) Microstate detection in naturalistic electroencephalography data: a systematic comparison of topographical clustering strategies on an emotional database. Front Neurosci 16:812624. https://doi.org/10.3389/fnins.2022.812624
Hu W, Zhang Z, Zhao H, Zhang L, Li L, Huang G, Liang Z (2023) EEG microstate correlates of emotion dynamics and stimulation content during video watching. Cereb Cortex 33:523–542. https://doi.org/10.1093/cercor/bhac082
Iannotti GR, Orepic P, Brunet D, Koenig T, Alcoba-Banqueri S, Garin DFA, Schaller K, Blanke O, Michel CM (2022) EEG spatiotemporal patterns underlying self-other voice discrimination. Cereb Cortex 32:1978–1992. https://doi.org/10.1093/cercor/bhab329
Jackson AF, Bolger DJ (2014) The neurophysiological bases of EEG and EEG measurement: a review for the rest of us. Psychophysiology 51:1061–1071. https://doi.org/10.1111/psyp.12283
Jouen A-L, Lancheros M, Laganaro M (2021) Microstate ERP analyses to pinpoint the articulatory onset in speech production. Brain Topogr 34:29–40. https://doi.org/10.1007/s10548-020-00803-3
Kadier N, Stein M, Koenig T (2021) EEG microstates and psychosocial stress during an exchange year. Brain Topogr 34:117–120. https://doi.org/10.1007/s10548-020-00806-0
Kaur A, Chinnadurai V, Chaujar R (2020) Microstates-based resting frontal alpha asymmetry approach for understanding affect and approach/withdrawal behavior. Sci Rep 10:4228. https://doi.org/10.1038/s41598-020-61119-7
Keil A, Debener S, Gratton G, Junghöfer M, Kappenman ES, Luck SJ, Luu P, Miller GA, Yee CM (2014) Committee report: publication guidelines and recommendations for studies using electroencephalography and magnetoencephalography. Psychophysiology 51:1–21. https://doi.org/10.1111/psyp.12147
Khanna A, Pascual-Leone A, Michel CM, Farzan F (2015) Microstates in resting-state EEG: current status and future directions. Neurosci Biobehav Rev 49:105–113. https://doi.org/10.1016/j.neubiorev.2014.12.010
Khanna A, Pascual-Leone A, Farzan F (2014) Reliability of resting-state microstate features in electroencephalography. PLoS ONE 9:e114163. https://doi.org/10.1371/journal.pone.0114163
Kleinert T, Nash K (2022) Trait aggression is reflected by a lower temporal stability of EEG resting networks. Brain Topogr. https://doi.org/10.1007/s10548-022-00929-6
Kleinert T, Nash K, Leota J, Koenig T, Heinrichs M, Schiller B (2022) A self-controlled mind is reflected by stable mental processing. Psychol Sci 33:2123–2137. https://doi.org/10.1177/09567976221110136
Kleinert T, Koenig T, Nash K, Wascher E (2023) On the reliability of the EEG microstate approach. Brain Topogr. https://doi.org/10.1007/s10548-023-00982-9
Koban L, Pourtois G, Bediou B, Vuilleumier P (2012) Effects of social context and predictive relevance on action outcome monitoring. Cogn Affect Behav Neurosci 12:460–478. https://doi.org/10.3758/s13415-012-0091-0
Kock N (2004) The psychobiological model: towards a new theory of computer-mediated communication based on Darwinian evolution. Organ Sci 15:327–348. https://doi.org/10.1287/orsc.1040.0071
Koenig T, Prichep L, Lehmann D, Sosa PV, Braeker E, Kleinlogel H, Isenhart R, John ER (2002) Millisecond by millisecond, year by year: normative EEG microstates and developmental stages. Neuroimage 16:41–48. https://doi.org/10.1006/nimg.2002.1070
Kvarven A, Strømland E, Johannesson M (2020) Comparing meta-analyses and preregistered multiple-laboratory replication projects. Nature Human Behav 4:423–434. https://doi.org/10.1038/s41562-019-0787-z
Lehmann D, Ozaki H, Pal I (1987) EEG alpha map series: brain micro-states by space-oriented adaptive segmentation. Electroencephalogr Clin Neurophysiol 67:271–288. https://doi.org/10.1016/0013-4694(87)90025-3
Li Z, Li Y, Li X, Zou F, Wang Y, Wu X, Luo Y, Zhang M (2021) The spontaneous brain activity of disgust: perspective from resting state fMRI and resting state EEG. Behav Brain Res 403:113135. https://doi.org/10.1016/j.bbr.2021.113135
Liang J, Li Y, Zhang Z, Luo W (2022) Sound gaps boost emotional audiovisual integration independent of attention: evidence from an ERP study. Biol Psychol 168:108246. https://doi.org/10.1016/j.biopsycho.2021.108246
Liu J, Xu J, Zou G, He Y, Zou Q, Gao J-H (2020) Reliability and individual specificity of EEG microstate characteristics. Brain Topogr 33:438–449. https://doi.org/10.1007/s10548-020-00777-2
Liu J, Hu X, Shen X, Lv Z, Song S, Zhang D (2023) The EEG microstate representation of discrete emotions. Int J Psychophysiol 186:33–41. https://doi.org/10.1016/j.ijpsycho.2023.02.002
Luck SJ (2012) Event-related potentials. In: Cooper H (ed) APA handbook of research methods in psychology, Vol 1: foundations, planning, measures, and psychometrics. American Psychological Association, Washington, DC, US, pp 523–546. https://doi.org/10.1037/13619-000
Ma Y, Shamay-Tsoory S, Han S, Zink CF (2016) Oxytocin and social adaptation: insights from neuroimaging studies of healthy and clinical populations. Trends Cogn Sci 20:133–145. https://doi.org/10.1016/j.tics.2015.10.009
Meyer-Lindenberg A, Domes G, Kirsch P, Heinrichs M (2011) Oxytocin and vasopressin in the human brain: social neuropeptides for translational medicine. Nat Rev Neurosci 12:524–538. https://doi.org/10.1038/nrn3044
Michel CM, Koenig T (2018) EEG microstates as a tool for studying the temporal dynamics of whole-brain neuronal networks: a review. Neuroimage 180:577–593. https://doi.org/10.1016/j.neuroimage.2017.11.062
Michel CM, Murray MM, Lantz G, Gonzalez S, Spinelli L, de Peralta RG (2004) EEG source imaging. Clin Neurophysiol 115:2195–2222. https://doi.org/10.1016/j.clinph.2004.06.001
Michel CM, Koenig T, Brandeis D, Gianotti LRR, Wackermann J (eds) (2009) Electrical neuroimaging. Cambridge University Press, Cambridge, UK. https://doi.org/10.1017/CBO9780511596889
Miskovic V, Keil A (2012) Acquired fears reflected in cortical sensory processing: a review of electrophysiological studies of human classical conditioning. Psychophysiology 49:1230–1241. https://doi.org/10.1111/j.1469-8986.2012.01398.x
Mueller EM, Pizzagalli DA (2016) One-year-old fear memories rapidly activate human fusiform gyrus. Soc Cogn Affect Neurosci 11:308–316. https://doi.org/10.1093/scan/nsv122
Murray MM, Brunet D, Michel CM (2008) Topographic ERP analyses: a step-by-step tutorial review. Brain Topogr 20:249–264. https://doi.org/10.1007/s10548-008-0054-5
Murray MM (2009) Principles of topographic analyses for electrical neuroimaging. In: Handy TC (ed) Brain signal analysis: advances in neuroelectric and neuromagnetic methods. The MIT Press, Cambridge, MA, US, pp 21–54. https://doi.org/10.7551/mitpress/9780262013086.003.0002
Nash K, Gianotti LRR, Knoch D (2014) A neural trait approach to exploring individual differences in social preferences. Front Behav Neurosci 8:458. https://doi.org/10.3389/fnbeh.2014.00458
Nash K, Leota J, Kleinert T, Hayward DA (2023) Anxiety disrupts performance monitoring: integrating behavioral, event-related potential, EEG microstate, and sLORETA evidence. Cereb Cortex 33:3787–3802. https://doi.org/10.1093/cercor/bhac307
Nash K, Schiller B, Gianotti LRR, Baumgartner T, Knoch D (2013) Electrophysiological indices of response inhibition in a Go/NoGo task predict self-control in a social context. PLoS ONE 8:e79462. https://doi.org/10.1371/journal.pone.0079462
Nash K, Kleinert T, Leota J, Scott A, Schimel J (2022) Resting-state networks of believers and non-believers: an EEG microstate study. Biol Psychol 169:108283. https://doi.org/10.1016/j.biopsycho.2022.108283
Open Science Collaboration (2015) Estimating the reproducibility of psychological science. Science 349:aac4716. https://doi.org/10.1126/science.aac4716
Ortigue S, Thompson JC, Parasuraman R, Grafton ST (2009) Spatio-temporal dynamics of human intention understanding in temporo-parietal cortex: a combined EEG/fMRI repetition suppression paradigm. PLoS ONE 4:e6962. https://doi.org/10.1371/journal.pone.0006962
Ortigue S, Sinigaglia C, Rizzolatti G, Grafton ST (2010) Understanding actions of others: the electrodynamics of the left and right hemispheres. A high-density EEG neuroimaging study. PLoS ONE 5:e12160. https://doi.org/10.1371/journal.pone.0012160
Panitz C, Hermann C, Mueller EM (2015) Conditioned and extinguished fear modulate functional corticocardiac coupling in humans. Psychophysiology 52:1351–1360. https://doi.org/10.1111/psyp.12498
Panitz C, Sperl MFJ, Hennig J, Klucken T, Hermann C, Mueller EM (2018) Fearfulness, neuroticism/anxiety, and COMT Val158Met in long-term fear conditioning and extinction. Neurobiol Learn Mem 155:7–20. https://doi.org/10.1016/j.nlm.2018.06.001
Pedroni A, Gianotti LRR, Koenig T, Lehmann D, Faber P, Knoch D (2017) Temporal characteristics of EEG microstates mediate trial-by-trial risk taking. Brain Topogr 30:149–159. https://doi.org/10.1007/s10548-016-0539-6
Pegna AJ, Gehring E, Meyer G, Del Zotto M (2015) Direction of biological motion affects early brain activation: a link with social cognition. PLoS ONE 10:e0131551. https://doi.org/10.1371/journal.pone.0131551
Pipinis E, Melynyte S, Koenig T, Jarutyte L, Linkenkaer-Hansen K, Ruksenas O, Griskova-Bulanova I (2017) Association between resting-state microstates and ratings on the Amsterdam Resting-State Questionnaire. Brain Topogr 30:245–248. https://doi.org/10.1007/s10548-016-0522-2
Pizzagalli D, Lehmann D, Koenig T, Regard M, Pascual-Marqui RD (2000) Face-elicited ERPs and affective attitude: brain electric microstate and tomography analyses. Clin Neurophysiol 111:521–531. https://doi.org/10.1016/S1388-2457(99)00252-7
Pizzagalli DA, Greischar LL, Davidson RJ (2003) Spatio-temporal dynamics of brain mechanisms in aversive classical conditioning: high-density event-related potential and brain electrical tomography analyses. Neuropsychologia 41:184–194. https://doi.org/10.1016/s0028-3932(02)00148-3
Prete G, Croce P, Zappasodi F, Tommasi L, Capotosto P (2022) Exploring brain activity for positive and negative emotions by means of EEG microstates. Sci Rep 12:3404. https://doi.org/10.1038/s41598-022-07403-0
Rand DG, Peysakhovich A, Kraft-Todd GT, Newman GE, Wurzbacher O, Nowak MA, Greene JD (2014) Social heuristics shape intuitive cooperation. Nat Commun 5:3677. https://doi.org/10.1038/ncomms4677
Schiller B, Brustkern J, Habermann M, Pacurar M, von Dawans B, Heinrichs M (2023a) Social high performers under stress behave more prosocially and detect happy emotions better in a male sample. Psychoneuroendocrinology. https://doi.org/10.1016/j.psyneuen.2023.106338
Schiller B, Brustkern J, Walker M, Hamm A, Heinrichs M (2023b) Oxytocin has sex-specific effects on trust and underlying neurophysiological processes. Psychoneuroendocrinology 151:106076. https://doi.org/10.1016/j.psyneuen.2023.106076
Schiller B, Domes G, Heinrichs M (2020a) Oxytocin changes behavior and spatio-temporal brain dynamics underlying inter-group conflict in humans. Eur Neuropsychopharm 31:119–130. https://doi.org/10.1016/j.euroneuro.2019.12.109
Schiller B, Gianotti LRR, Baumgartner T, Knoch D (2019a) Theta resting EEG in the right TPJ is associated with individual differences in implicit intergroup bias. Soc Cogn Affect Neurosci 14:281–289. https://doi.org/10.1093/scan/nsz007
Schiller B, Gianotti LRR, Baumgartner T, Nash K, Koenig T, Knoch D (2016) Clocking the social mind by identifying mental processes in the IAT with electrical neuroimaging. Proc Natl Acad Sci U S A 113:2786–2791. https://doi.org/10.1073/pnas.1515828113
Schiller B, Koenig T, Heinrichs M (2019b) Oxytocin modulates the temporal dynamics of resting EEG networks. Sci Rep 9:13418. https://doi.org/10.1038/s41598-019-49636-6
Schiller B, Kleinert T, Teige-Mocigemba S, Klauer KC, Heinrichs M (2020b) Temporal dynamics of resting EEG networks are associated with prosociality. Sci Rep 10:13066. https://doi.org/10.1038/s41598-020-69999-5
Schlegel F, Lehmann D, Faber PL, Milz P, Gianotti LRR (2012) EEG microstates during resting represent personality differences. Brain Topogr 25:20–26. https://doi.org/10.1007/s10548-011-0189-7
Seitzman BA, Abell M, Bartley SC, Erickson MA, Bolbecker AR, Hetrick WP (2017) Cognitive manipulation of brain electric microstates. Neuroimage 146:533–543. https://doi.org/10.1016/j.neuroimage.2016.10.002
Sikka A, Jamalabadi H, Krylova M, Alizadeh S, van der Meer JN, Danyeli L, Deliano M, Vicheva P, Hahn T, Koenig T, Bathula DR, Walter M (2020) Investigating the temporal dynamics of electroencephalogram (EEG) microstates using recurrent neural networks. Hum Brain Mapp 41:2334–2346. https://doi.org/10.1002/hbm.24949
Skrandies W (1990) Global field power and topographic similarity. Brain Topogr 3:137–141. https://doi.org/10.1007/BF01128870
Spengler FB, Schultz J, Scheele D, Essel M, Maier W, Heinrichs M, Hurlemann R (2017) Kinetics and dose dependency of intranasal oxytocin effects on amygdala reactivity. Biol Psychiatry 82:885–894. https://doi.org/10.1016/j.biopsych.2017.04.015
Sperl MFJ, Wroblewski A, Mueller M, Straube B, Mueller EM (2021) Learning dynamics of electrophysiological brain signals during human fear conditioning. Neuroimage 226:117569. https://doi.org/10.1016/j.neuroimage.2020.117569
Sur S, Sinha VK (2009) Event-related potential: An overview. Ind Psychiatry J 18:70–73. https://doi.org/10.4103/0972-6748.57865
Takehara H, Ishihara S, Iwaki T (2020) Comparison between facilitating and suppressing facial emotional expressions using frontal EEG asymmetry. Front Behav Neurosci 14:554147. https://doi.org/10.3389/fnbeh.2020.554147
Tanaka M, Yamada E, Maekawa T, Ogata K, Takamiya N, Nakazono H, Tobimatsu S (2021) Gender differences in subliminal affective face priming: a high-density ERP study. Brain Behav 11:e02060. https://doi.org/10.1002/brb3.2060
Tarailis P, Šimkutė D, Koenig T, Griškova-Bulanova I (2021) Relationship between spatiotemporal dynamics of the brain at rest and self-reported spontaneous thoughts: an EEG microstate approach. J Pers Med 11:1216. https://doi.org/10.3390/jpm11111216
Tarailis P, Koenig T, Michel CM, Griškova-Bulanova I (2023) The functional aspects of resting EEG microstates: a systematic review. Brain Topogr. https://doi.org/10.1007/s10548-023-00958-9
Thierry G, Pegna AJ, Dodds C, Roberts M, Basan S, Downing P (2006) An event-related potential component sensitive to images of the human body. Neuroimage 32:871–879. https://doi.org/10.1016/j.neuroimage.2006.03.060
Thigpen NN, Bartsch F, Keil A (2017) The malleability of emotional perception: short-term plasticity in retinotopic neurons accompanies the formation of perceptual biases to threat. J Exp Psychol 146:464–471. https://doi.org/10.1037/xge0000283
Tomescu MI, Papasteri CC, Sofonea A, Boldasu R, Kebets V, Pistol CAD, Poalelungi C, Benescu V, Podina IR, Nedelcea CI, Berceanu AI, Carcea I (2022) Spontaneous thought and microstate activity modulation by social imitation. Neuroimage 249:118878. https://doi.org/10.1016/j.neuroimage.2022.118878
Vaughan HG (1982) The neural origins of human event-related potentials. Ann N Y Acad Sci 388:125–138. https://doi.org/10.1111/J.1749-6632.1982.TB50788.X
Vohs KD, Schmeichel BJ, Lohmann S et al (2021) A multisite preregistered paradigmatic test of the ego-depletion effect. Psychol Sci 32:1566–1581. https://doi.org/10.1177/0956797621989733
Walker PM, Silvert L, Hewstone M, Nobre AC (2008) Social contact and other-race face processing in the human brain. Soc Cogn Affect Neurosci 3:16–25. https://doi.org/10.1093/scan/nsm035
Walter Y, Koenig T (2022) Neural network involvement for religious experiences in worship measured by EEG microstate analysis. Soc Neurosci 17:258–275. https://doi.org/10.1080/17470919.2022.2083228
Yao D, Qin Y, Hu S, Dong L, Bringas Vega ML, Valdés Sosa PA (2019) Which reference should we use for EEG and ERP practice? Brain Topogr 32:530–549. https://doi.org/10.1007/s10548-019-00707-x
Zanesco AP, Denkova E, Jha AP (2021a) Associations between self-reported spontaneous thought and temporal sequences of EEG microstates. Brain Cogn 150:105696. https://doi.org/10.1016/j.bandc.2021.105696
Zanesco AP, King BG, Skwara AC, Saron CD (2020) Within and between-person correlates of the temporal dynamics of resting EEG microstates. Neuroimage 211:116631. https://doi.org/10.1016/j.neuroimage.2020.116631
Zanesco AP, Skwara AC, King BG, Powers C, Wineberg K, Saron CD (2021b) Meditation training modulates brain electric microstates and felt states of awareness. Hum Brain Mapp 42:3228–3252. https://doi.org/10.1002/hbm.25430
Zelenina M, Kosilo M, da Cruz J, Antunes M, Figueiredo P, Mehta MA, Prata D (2022) Temporal dynamics of intranasal oxytocin in human brain electrophysiology. Cereb Cortex 32:3110–3126. https://doi.org/10.1093/cercor/bhab404
Zerna J, Strobel A, Scheffel C (2021) EEG microstate analysis of emotion regulation reveals no sequential processing of valence and emotional arousal. Sci Rep 11:21277. https://doi.org/10.1038/s41598-021-00731-7
Zhang M, Li Z, Wang L, Yang S, Zou F, Wang Y, Wu X, Luo Y (2021) The resting-state electroencephalogram microstate correlations with empathy and their moderating effect on the relationship between empathy and disgust. Front Hum Neurosci 15:626507. https://doi.org/10.3389/fnhum.2021.626507
Acknowledgements
This review was initiated at the conference “50 years of microstates,” which took place in Bern (Switzerland) in 2022.
Funding
Open Access funding enabled and organized by Projekt DEAL. B.S. acknowledges support from the European Research Council (ERC; ERC starting grant “From face-to-face to face-to-screen: Social animals interacting in a digital world,” 101076414). M.F.J.S. was supported by the “Justus Liebig University Postdoc Fund,” provided by the Postdoc Career and Mentoring Office of the University of Giessen.
Author information
Authors and Affiliations
Contributions
All authors contributed to conceiving and drafting the manuscript and to the literature search. B.S. compiled the final draft. K.N. revised the manuscript. L.R.R.G. created the figures. All authors edited and commented on the final draft.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Handling Editor: Christoph Michel.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Schiller, B., Sperl, M.F.J., Kleinert, T. et al. EEG Microstates in Social and Affective Neuroscience. Brain Topogr 37, 479–495 (2024). https://doi.org/10.1007/s10548-023-00987-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10548-023-00987-4