Abstract
Background
Poor suppression of anticipated sensory information from muscle contractions is thought to underlie high fatigue. Such diminished task-related sensory attenuation is reflected in resting state connectivity. Here we test the hypothesis ‘altered electroencephalography (EEG)-derived functional connectivity in somatosensory network in the beta band, is a signature of fatigue in post-stroke fatigue’.
Methods
In non-depressed, minimally impaired stroke survivors (n = 29), with median disease duration of 5 years, resting state neuronal activity was measured using 64-channel EEG. Graph theory-based network analysis measure of functional connectivity via small-world index (SW) was calculated focusing on right and left motor (Brodmann areas 4, 6, 8, 9, 24 and 32) and sensory (Brodmann areas 1, 2, 3, 5, 7, 40 and 43) networks, in the beta (13–30 Hz) frequency range. Fatigue was measured using Fatigue Severity Scale - FSS (Stroke), with scores of > 4, defined as high fatigue.
Results
Results confirmed the working hypothesis, with high fatigue stroke survivors showing higher small-worldness in the somatosensory networks when compared to low fatigue.
Conclusion
High levels of small-worldness in somatosensory networks indicates altered processing of somesthetic input. Such altered processing would explain high effort perception within the sensory attenuation model of fatigue.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Stroke, a result of vascular insufficiency to neurons present with fatigue as a significant symptom. The severity of stroke does not explain reported levels of fatigue (Kutlubaev et al. 2012; van der Werf et al. 1998). Previously, we have proposed a sensory attenuation hypothesis of fatigue, based on poor suppression of anticipated sensory information –(Kuppuswamy 2017, 2022). Poor suppression of muscle sensory afferents results in assigning high effort to simple tasks which explains a significant proportion of post-stroke fatigue (Doncker et al. 2020a, b) In visual and auditory perception, poor distractor suppression explains post-stroke fatigue (Kuppuswamy et al. 2022; Doncker and Kuppuswamy 2022). While other brain regions/networks have been implicated in fatigue such as parietal, pre-frontal and sub-cortical networks (Jaeger et al. 2019; Finke et al. 2015; Cotter et al. 2021), here we specifically focus on somatosensory and motor networks which have been implicated in PSF; to test the predictions of the sensory attenuation hypothesis that a dysfunction within the somatosensory networks underlies fatigue.
From a neuronal network functioning point of view, behaviour mirrors structural and functional changes in networks, which persist in resting state (Graziadio et al. 2010). Neuronal networks at rest express features that keep trace of their ability to perform the required behaviour (Kim and Kang 2018; Wahlheim et al. 2022; Doucet et al. 2012; Y. Li et al. 2022a, b; Liu et al. 2022). These features of networks at rest display alterations that reflect chronic symptoms (Porcaro et al. 2019). Both in post-stroke fatigue (PSF) and other disease where fatigue is significant, neurophysiological (Kuppuswamy et al. 2015a, b, c; Ondobaka et al. 2021; De Doncker et al. 2021; Liepert et al. 2005; Morgante et al. 2011; Russo et al. 2017) and behavioural (Kuppuswamy et al. 2015a, b, c, 2016; De Doncker et al. 2020a; Doncker et al. 2020b) findings support an altered resting state, specifically sensory network activity.
Ensembles of neurons that fire at specific frequencies and communicate with each other by synchronising their firing, comprise a neuronal network. To understand a network’s activity, the strength of synchronicity between various nodes is mapped using functional connectivity methods (Bullmore and Sporns 2009). Functional connectivity is defined as the temporal correlation or dependency between distinct neuronal groups and areas (Fingelkurts et al. 2005; Rubinov and Sporns 2010). Such temporal correlation occurs in various frequency bands, with low frequencies associated with arousal, mid-range frequencies related to sensorimotor activity, and high frequencies representing higher order functions such as error detection and learning. With PSF proposed to be a problem of sensorimotor control, specifically arising from processing of incoming muscle related sensory information, we anticipated a fatigue related modulation of beta band frequency.
Here we investigate if a dysfunction of somatosensory networks underlies PSF, as demonstrated by changes in beta-band neuronal activity in sensory and motor networks at rest.
Methods
Participants
This study was approved by the London Bromley Research Ethics Committee (REC reference number: 16/LO/0714). Stroke survivors were recruited and tested at the Institute of Neurology, London, UK.
All stroke survivors were screened prior to the study based on the following criteria: first-time ischaemic or haemorrhagic stroke; stroke occurred at least 3 months prior to the study; no clinical diagnosis of any other neurological disorder; physically well recovered following their stroke defined as grip strength and manual dexterity of the affected hand being at least 60% of the unaffected hand assessed using a hand-held dynamometer and the nine-hole peg test (NHPT) respectively; not taking anti-depressants or any other medication that has a direct effect on the central nervous system; not clinically depressed with depression scores ≤ 11 assessed using the Hospital Anxiety and Depression Scale (HADS)(Snaith 2003).
Twenty-nine stroke survivors took part in the study (Table 1) and provided written informed consent in accordance with the Declaration of Helsinki. A formal sample-size calculation was not performed due to lack of pilot data. However, in previous studies, differences could be observed in the measure of resting state functional connectivity in as few as 10 subjects per group (Nordin et al. 2016).
Fatigue
Trait fatigue was quantified using Fatigue Severity Scale, FSS-7. An average score of one indicates no fatigue while an average score of seven indicates maximum fatigue (Krupp et al. 1989). High fatigue was defined as FSS-7 > 4 (Valko et al. 2008).
Control group: Healthy humans were not recruited for this study as a control group, specifically as the state of the brain is likely to have changed after an injury or establishment of a disease (Kuppuswamy 2023), therefore an ideal control group will be a within-disease control group. The stroke low (no) fatigue group was used as a control for this study.
EEG Recording
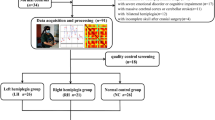
Whole-scalp electroencephalography (EEG) data was recorded using 64-channel systems, ActiCap, Herrsching, Germany, and a BrainAmp, at rest, with eyes open and focusing on a fixation cross. Duration of recording was seven minutes. The 64 electrodes were positioned on the cap in accordance with the 10–20 international EEG electrode array. During online recordings, channels FCz and AFz were used as reference and ground respectively. Impedances were kept below 10 kΩ throughout the recording. The EEG signal was sampled at 1 kHz and visualized online using the BrainVision Recorder Software (BrainVision Recorder, Version 1.21.0102 Brain Products GmbH, Gilching, Germany).
EEG Analysis
EEG analyses were performed with a combination of EEGLAB (Delorme and Makeig 2004) and custom Matlab scripts. EEG data was down-sampled to 250 Hz and then band-pass filtered from 0.1 to 47 Hz using a finite impulse response filter. Noisy channels were identified and removed using automated procedures. EEG data was subsequently segmented into two second epochs, and epochs containing noisy data were identified as follows: the mean activity of all EEG channels was computed, and the threshold was set at ± 2 times the standard deviation of the mean activity. Epochs containing activity exceeding the threshold value were marked and subsequently removed. This left a total of 160 (± 15) two second epochs. To identify and remove ocular movements and blink artifacts from the EEG data, an independent component analysis (ICA) implemented within EEGLAB was used. ICA is a blind source decomposition algorithm that enables the separation of statistically independent sources from multichannel data (Jung et al. 2000). The components were subsequently visually inspected and those containing ocular movements or blink artifacts were removed. The previously removed channels were then interpolated back into the dataset and finally, the EEG data was re-referenced against the grand average of all scalp electrodes.
Graph Theory Estimates
Functional Connectivity Analysis. EEG connectivity analysis was carried out using the exact low-resolution electromagnetic tomography (eLORETA) software (The KEY Institute of Brain-Mind Research University Hospital of Psychiatry, Zurich; http://www.uzh.ch/keyinst/NewLORETA/LORETA01.htm). The eLORETA algorithm is a well-established linear inverse solution for EEG signals (Pascual-Marqui 2002).
Following whole brain sources reconstruction, connectivity was computed using the eLORETA software on four brain regions, divided into motor and sensory networks of the left and right hemisphere based on Broadmann areas (BAs). Each BA is a region of interest (ROI). The BAs that formed the motor network for both the left and right hemisphere included BA4, 6, 8, 9, 24 and 32, while the BAs that formed the sensory network for both the left and right hemisphere included BA1, 2, 3, 5, 7, 40 and 43.
Current density time series of all BAs within each of the four networks was computed in eLORETA and used to estimate the functional connectivity using the Lagged Linear Coherence (LagR) algorithm, not affected by volume conductance and low spatial resolution in each of the four networks (Pascual-Marqui 2007). Lagged Linear Coherence was computed for beta (13–30 Hz) band frequency.
Graph Analysis. A network is a mathematical representation of a real-world complex system and is defined by a collection of nodes (vertices) and links (edges) between pairs of nodes. Nodes in large-scale brain networks represent brain regions, while links represent anatomical or functional connections. Nodes should ideally represent brain regions with coherent patterns of anatomical or functional connections. The connectivity parameters extracted between all pairs of ROIs for each frequency band is in the form of a square matrix W, with dimensions equal to the number of ROIs. Each row and column within matrix W represent nodes, while the values within the matrix represent the strength of connection between each pair of nodes.
Once the networks of interest were constructed, the core measures of graph theory that summarize the aspects of segregation and integration of a network were computed using the Brain Connectivity Toolbox (Rubinov and Sporns 2010). Segregation refers to the degree to which network elements form individual and separate clusters and is measured by the clustering coefficient (C). Integration refers to the capacity of the network to become interconnected and exchange information and is measured by the parameter characteristic path length (L). The clustering coefficient and characteristic path length represent the efficiency of the network with respect to local and global connectedness respectively. Weighted clustering (Cw) coefficient and weighted characteristic path length (Lw) were computed as a measure of segregation and integration of the network as follows:
Where Cbrain and Lbrain are the clustering coefficient and characteristic path length derived from the connectivity matrix of each participant. Crandom and Lrandom are the mean values of the clustering coefficient and characteristic path length of 100 surrogate random networks that have the same basic characteristics as the original network that were derived by randomly rewiring the nodes using the original network edge weights. The random networks preserve the same basic characteristics as the original network such as the number of nodes and edge weights. The random networks were constructed by Brain Connectivity Toolbox29. A measure of network small-worldness (Sw) was therefore defined as the ratio between Cw and Lw; the ratio between local connectedness and the global integration of the network.
When Sw has a value of approximately 1, a network is said to have “small-world properties” meaning a good combination of high levels of local clustering among nodes and proper paths that globally link all network nodes (all nodes of a large system are linked through relatively few intermediate steps). Sw values greater than 1 suggest high levels of local clustering among nodes and many short paths that globally link all nodes of the network, while Sw values less than 1 suggest poor local connectivity and stunted connections.
Statistical Analysis
All statistical analysis was performed using R (RStudio Version 1.2.5033). Spearman rank correlations identified associations between trait fatigue (FSS-7) and demographic variables (age, grip strength, NHPT, HADS – Depression, HADS – Anxiety and Time Post-Stroke). Wilcoxon rank sum tests identified association between trait fatigue (FSS-7 scores) and categorical measures (sex, hemisphere affected and type of stroke). The effect of vascular territory on FSS was not analysed as there were too few in each group. Shapiro-Wilk’s test of normality assessed distribution of dependent variable and Levene’s test assessed homogeneity of variances.
In normally distributed variables, a three-way mixed ANOVA was performed to evaluate the effects of fatigue (between subject factor: StrokeLow, StrokeHigh) and sex (between subject factor: Male, Female) on small worldness within the beta frequency band (dependent variable), for two networks (sensory and motor) and two hemispheres (left and right). Greenhouse-Geisser epsilon adjustment corrected any deviations from sphericity. Post-hoc pairwise comparisons (t-tests) with Bonferroni adjustment was performed to identify main effects.
Results
Participant Demographics
Twenty-nine stroke survivors completed the study (11 females and 18 males). The median FSS-7 score was 5.29 (IQR = 2.57) in females and 2.50 (IQR = 2.46) in males. The Wilcoxon test showed that the difference in FSS-7 score was marginally non-significant (p = 0.05, effect size = 0.37). Spearman rank correlations between trait fatigue (FSS-7) and all continuous demographic measures revealed a significant positive association between trait fatigue and HADS-Depression (Spearman ρ = 0.41, p = 0.03), while no other variable correlated with trait fatigue (Age: Spearman ρ = 0.02, p = 0.91; Grip strength: Spearman ρ = -0.23, p = 0.24; NHPT: Spearman ρ = -0.25, p = 0.19; HADS-Anxiety: Spearman ρ = 0.35, p = 0.06).
Clinical Characteristics
There were no confirmed MRI lesions in any of the stroke survivors in the study. The association between trait fatigue (FSS-7) and the clinical characteristics of the stroke was assessed across all stroke survivors. The median FSS-7 score in those with right hemisphere strokes was 4.43 (IQR = 3.00) and 2.71 (IQR = 3.75) in those with left hemisphere strokes (Wilcoxon test: p = 0.50, effect size r = 0.13). The median FSS-7 score in those with ischemic strokes was 2.93 (IQR = 3.46) and 3.86 (IQR = 2.29) in those with hemorrhagic strokes (Wilcoxon test: p = 0.51, effect size r = 0.13). Regarding the vascular territory affected, the data from five stroke survivors was missing as the clinical notes could not be retrieved. The median FSS-7 score in those where the MCA was affected was 3.36 (IQR = 3.25), the median FSS-7 score in those where the PCA was affected was 3.07 (IQR = 2.07), while the median FSS-7 score in those where the Brainstem/Cerebellum was affected was 4.21 (IQR = 2.68) (Kruskal-Wallis test: p = 0.57, effect size η2=-0.04). A spearman rank correlation between FSS-7 and the Time Post-Stroke at which the participants took part in the study showed no significant association (spearman ρ = 0.08, p = 0.67). Any meaningful interpretation of the effect of the type of stroke and vascular territory affected on FSS-7 in the current cohort of stroke survivors is difficult given the skewed numbers.
Small Worldness
Across the two networks (sensory/motor) and hemispheres (left/right) there were two extreme outliers (both in the motor network of the right hemisphere). After exclusion of outliers, the data was normally distributed, (p > 0.05) and variances were homogenous (p > 0.05). The three-way-ANOVA revealed a main effect of network type (F(1,23) = 15.59, p < 0.01, η2 = 0.13) and a significant interaction between network type and fatigue level (F(1,23) = 16.79, p < 0.01, η2 = 0.14) on small worldness in beta frequency band. There was also a significant three way interaction between network type, fatigue level and sex (F(1,23) = 5.09, p = 0.03, η2 = 0.05), however there was no significant two way interaction between sex and network type (F(1,23) = 0.65, p = 0.43, η2 = 0.006) nor between sex and fatigue (F(1,23) = 1.05, p = 0.32, η2 = 0.009). Post-hoc multiple pairwise comparisons revealed a significant difference in small worldness within the beta frequency band (Fig. 1) between the StrokeLow and StrokeHigh groups in the sensory network of the right hemisphere (p = 0.01), driven by the difference in female participants (P = 0.0127). There was also a significant difference between StrokeLow and StrokeHigh in left and right motor networks (p = 0.02 and p = 0.03 respectively), with females driving the difference in the right hemisphere (P = 0.0138).
Functional Connectivity of the Sensory and Motor Networks in the beta band frequency. The value of Small Worldness across the two fatigue groups both in males and females (StrokeMale in blue and StrokeFemale in yellow) is displayed using boxplots for the sensory network in the left (A) and right (B) hemispheres and for motor networks in the left (C) and right (D) hemispheres. Significant differences between fatigue groups are indicated using asterisks (* = p < 0.05). In the right motor and sensory networks, the difference is driven by females
Discussion
In twenty-nine patients with stroke, we show that those with high levels of trait fatigue exhibited significantly higher levels of small worldness in the sensory networks and significantly lower levels of small worldness in the motor networks in the beta band frequency (13-30 Hz). There was no association between clinical features of stroke, and trait fatigue which confirms previous findings (Kutlubaev et al. 2012). As investigated by small worldness, functional brain connectivity simultaneously reconciles the opposing demands of functional integration and segregation. The small-world index reflects the balance of functionally specialized (segregated) modules with a robust number of intermodular (integrating) links. Here we observed that fatigue is paralleled by the alteration of small-world organization in sensory and motor networks.
The observed changes in sensory networks are lateralised. A possible trivial explanation could be that the small numbers have resulted in one side reaching significance and not the other. If the observed lateralisation is not due to limited numbers, ‘hyper-connectivity’ in right sensory networks is in keeping with recent findings of a shift towards right hemispheric dominance in sensorimotor networks in those with high post-stroke fatigue (Ondobaka et al. 2021). Healthy brains exhibit left-hemispheric dominance (Netz et al. 1995; Giovannelli et al. 2009) and a shift towards right dominance has been observed in several psychiatric diseases including depression (Lefaucheur et al. 2008). A third possible non-trivial explanation could be the small difference in the number of left hemispheric strokes in the low fatigue group, however, this is unlikely as previous studies show shift in hemispheric dominance regardless of the side of stroke (Ondobaka et al. 2021).
Beta band activity is commonly known as the sensorimotor ‘idling’ rhythm seen in all cortical and sub-cortical motor areas at rest. Movement desynchronises beta band oscillations which lead to the idea that beta frequency is the rhythm of rest for sensorimotor areas. Recent proposals suggest that beta band activity may not simply reflect a lack of movement but is rather an indicator for maintenance of sensorimotor status quo (Engel and Fries 2010). During periods of spontaneous enhancement in resting beta band activity, movements are slower, than when resting beta activity is lower (Gilbertson et al. 2005). In light of such findings, the current results indicate that those with high fatigue are likely to have slowed movements. In post-stroke fatigue, while there is no difference in reaction times, there is slowing of movements (Kuppuswamy et al. 2015a, b, c), perhaps because there is a resistance to change sensorimotor status quo as reflected by enhanced beta rhythm small-worldness shown in this study.
It is well-established that fatigue is only marginally associated with motor and cognitive deficits (Ingles et al. 1999; Winward et al. 2009). However, despite good functional ability, markers of poor behavioural flexibility is associated with high fatigue (De Doncker et al. 2021; Morgante et al. 2011). Enhanced sensory, and diminished motor network functional connectivity that seeks to maintain a sensory state, thereby making new sensory states less desirable, lends further support to the idea of poor behavioural flexibility underpinning high fatigue.
In this study we provide evidence for alteration in somatosensory processing, which may indicate a possible mechanism that drives fatigue is poor somatosensory processing. Both in post-stroke fatigue and other neurological conditions such as MS, there are several reports of altered resting state connectivity (Ondobaka et al. 2021; Woodward et al. 2019; Bisecco et al. 2018; Jaeger et al. 2019; Stefancin et al. 2019; Cotter et al. 2021). In stroke, suggestions of parietal hypoconnectivity and frontal hyper connectivity (Cotter et al. 2021) with reversed inter-hemispheric balance of connectivity (Ondobaka et al. 2021), are implicated in manifestation of fatigue. In MS, changes in default mode network (Jaeger et al. 2019) and involvement of striatal circuits involved in movement, sensation and motivation (Bisecco et al. 2018) have all been implicated in development of fatigue. While several brain regions have been implicated in both diseases as the core regions involved in fatigue, very few of the studies performed hypothesis driven analysis on resting state activity. In the present study, we hypothesised that attention to somatosensory input in not suppressed as normal [indicated by high perceived effort during muscle contraction (Doncker et al. 2020a, b)] which will be reflected in the resting state both in sensory and motor networks. Hyper-connectivity in sensory networks, commonly seen as a marker of tendency to maintain sensory states quo, and resultant hypoconnectivity in motor networks that indicate lower M1 excitability (Kuppuswamy et al. 2015a, b, c) supports the premise that poor somatosensory attenuation underpins high PSF. In summary, increased small world-ness in somatosensory networks suggests a propensity to maintain the status quo i.e. rest; an increased propensity to inaction (rest) translates into an increase in the effort needed to initiate an action; increased effort results in fatigue.
The high fatigue group had a disproportionately high number of females when compared to the low fatigue group. Previous studies have reported greater incidence of fatigue in female stroke survivors (Cumming et al. 2016) and sex significantly influences measures of resting state connectivity (Stumme et al. 2020). While difference in incidence of fatigue was previously attributed to factors such as reporting biases influenced by sociocultural factors, the difference in resting state connectivity between males and females with high fatigue seen in this study opens up the possibility of a biological basis for differences in incidence of PSF. The influence of sex on resting state connectivity regardless of fatigue or stroke is unlikely to be the driver of the differences seen here, as sensorimotor networks are less likely to be influenced by sex (L. Li et al. 2022), unlike other brain networks. While further speculation on differences in biological mechanisms driving fatigue in males and females is beyond the scope of this paper, future mechanistic studies in fatigue must consider the possibility of sex being a confounding factor in interpretation of findings, and also directly study differences between males and females in PSF.
Limitations: While the hypothesis-driven approach of this study is a strength, it could also be a limitation. No neural network in the brain operates in isolation and the influences of other networks on sensorimotor areas of the brain are also likely influenced by the differences seen in this study, which needs further investigation. The relatively small numbers of participants in this study is further highlighted by the differences seen between sexes in this study. Lesion location does not influence PSF incidence or severity, however, to definitively exclude influence of lesion location on neural network activity relevant to fatigue, greater numbers in each lesion type is warranted.
Conclusion
Chronic fatigue is a feature of several established long-term disorders. However, there is no principled framework to understand the mechanisms of fatigue. In this paper, we demonstrate sensorimotor network activity is altered in line with the predictions of the sensory attenuation model of fatigue in chronic stroke survivors. This is a promising framework which could explain altered connectivity seen in other neurological disorders with long-term fatigue and future work must focus on exploring sensory attenuation in chronic fatigue.
Data Availability
The processed EEG data is available on request from a.kuppuswamy@ucl.ac.uk.
References
Bisecco A, Nardo FD, Docimo R, Caiazzo G, d’Ambrosio A, Bonavita S, Capuano R et al (2018) Fatigue in multiple sclerosis: the contribution of resting-state functional connectivity reorganization. Mult Scler 24(13):1696–1705. https://doi.org/10.1177/1352458517730932
Bullmore Ed, and Olaf Sporns (2009) Complex brain networks: Graph Theoretical Analysis of Structural and Functional Systems. Nat Rev Neurosci 10(3):186–198. https://doi.org/10.1038/nrn2575
Cotter G, Khlif MS, Bird L, Howard ME, Brodtmann A (2021) and Natalia Egorova-Brumley. “Post-Stroke Fatigue Is Associated with Resting State Posterior Hypoactivity and Prefrontal Hyperactivity.” International Journal of Stroke: Official Journal of the International Stroke Society, September, 17474930211048324. https://doi.org/10.1177/17474930211048323
Cumming TB, Packer M, Kramer SF, and Coralie English (2016) The prevalence of fatigue after stroke: a systematic review and Meta-analysis. Int J Stroke: Official J Int Stroke Soc 11(9):968–977. https://doi.org/10.1177/1747493016669861
De Doncker W, Charles L, Ondobaka S (2020a) and Annapoorna Kuppuswamy. “Exploring the Relationship between Effort Perception and Post-Stroke Fatigue.” Neurology, October. https://doi.org/10.1212/WNL.0000000000010985
De Doncker, William KE, Brown, and Annapoorna Kuppuswamy (2021) Influence of post-stroke fatigue on reaction Times and Corticospinal excitability during Movement Preparation. Clin Neurophysiol 132(1):191–199. https://doi.org/10.1016/j.clinph.2020.11.012
Delorme A, and Scott Makeig (2004) EEGLAB: an Open source toolbox for analysis of single-trial EEG Dynamics Including Independent Component Analysis. J Neurosci Methods 134(1):9–21. https://doi.org/10.1016/j.jneumeth.2003.10.009
Doncker WD, and Annapoorna Kuppuswamy (2022) The Cocktail Party Effect in post-stroke fatigue: an EEG study.” bioRxiv. https://doi.org/10.1101/2022.03.17.484808
Doncker W, De L, Charles S, Ondobaka, and Annapoorna Kuppuswamy (2020b) Exploring the relationship between effort perception and poststroke fatigue. Neurology 95(24):e3321–e3330. https://doi.org/10.1212/WNL.0000000000010985
Doucet BM, Lam A, and Lisa Griffin (2012) Neuromuscular electrical stimulation for skeletal muscle function. Yale J Biol Med 85(2):201–215
Engel AK, and Pascal Fries (2010) Beta-Band Oscillations–Signalling the Status Quo? Curr Opin Neurobiol 20(2):156–165. https://doi.org/10.1016/j.conb.2010.02.015
Fingelkurts AA, Alexander A, Fingelkurts, Kähkönen S (2005) Functional connectivity in the Brain–Is it an Elusive Concept? Neurosci Biobehav Rev 28(8):827–836. https://doi.org/10.1016/j.neubiorev.2004.10.009
Finke C, Schlichting J, Papazoglou S, Scheel M, Freing A, Soemmer C, Pech LM et al (2015) Altered basal ganglia functional connectivity in multiple sclerosis patients with fatigue. Mult Scler 21(7):925–934. https://doi.org/10.1177/1352458514555784
Gilbertson T, Lalo E, Doyle L, Lazzaro VD, Cioni B, and Peter Brown (2005) Existing Motor State is favored at the expense of New Movement during 13–35 hz oscillatory synchrony in the human Corticospinal System. J Neurosci 25(34):7771–7779. https://doi.org/10.1523/JNEUROSCI.1762-05.2005
Giovannelli F, Borgheresi A, Balestrieri F, Zaccara G, Viggiano MP, Cincotta M, and Ulf Ziemann (2009) Modulation of interhemispheric inhibition by Volitional Motor Activity: an Ipsilateral Silent Period Study. J Physiol 587(Pt 22):5393–5410. https://doi.org/10.1113/jphysiol.2009.175885
Graziadio S, Basu A, Tomasevic L, Zappasodi F, Tecchio F, Eyre JA (2010) Developmental tuning and Decay in Senescence of Oscillations linking the Corticospinal System. J Neuroscience: Official J Soc Neurosci 30(10):3663–3674. https://doi.org/10.1523/JNEUROSCI.5621-09.2010
Ingles JL, Eskes GA, Phillips SJ (1999) Fatigue after stroke. Arch Phys Med Rehabil 80(2):173–178
Jaeger S, Paul F, Scheel M, Brandt A, Heine J, Pach D, Witt CM, Judith Bellmann-Strobl, and Carsten Finke (2019) Multiple sclerosis-related fatigue: altered resting-state functional connectivity of the ventral striatum and Dorsolateral Prefrontal Cortex. Mult Scler 25(4):554–564. https://doi.org/10.1177/1352458518758911
Jung T-P, Makeig S, Humphries C, Lee T-W, McKEOWN MJ, Iragui V, Terrence J, Sejnowski (2000) Removing Electroencephalographic Artifacts by Blind source separation. Psychophysiology 37(2):163–178. https://doi.org/10.1111/1469-8986.3720163
Kim J, and Eunjoo Kang (2018) Strength of resting-state functional connectivity Associated with Performance-Adjustment ability. Behav Brain Res 347(July):377–384. https://doi.org/10.1016/j.bbr.2018.02.024
Krupp LB, Nicholas G, LaRocca JMuir-Nash, and Alfred D. Steinberg (1989) The fatigue severity scale: application to patients with multiple sclerosis and systemic Lupus Erythematosus. Arch Neurol 46(10):1121–1123. https://doi.org/10.1001/archneur.1989.00520460115022
Kuppuswamy A (2017) The fatigue conundrum. Brain 140(8):2240–2245. https://doi.org/10.1093/brain/awx153
Kuppuswamy A (2022) The neurobiology of pathological fatigue: New Models, new questions. The Neuroscientist: A Review Journal Bringing Neurobiology Neurology and Psychiatry 28(3):238–253. https://doi.org/10.1177/1073858420985447
Kuppuswamy A (2023) On the importance of not comparing apples with pears. Eur J Neurol 30(2):557. https://doi.org/10.1111/ene.15587
Kuppuswamy A, Clark EV, Sandhu KS, Rothwell JC, Ward NS (2015a) Post-stroke fatigue: a problem of altered Corticomotor Control? J Neurol Neurosurg Psychiatry 86(8):902–904. https://doi.org/10.1136/jnnp-2015a-310431
Kuppuswamy A, Ella V, Clark, Isobel F, Turner JC, Rothwell, Ward NS (2015b) Post-stroke fatigue: a deficit in Corticomotor Excitability? Brain 138(Pt 1):136–148. https://doi.org/10.1093/brain/awu306
Kuppuswamy A, Clark EV, Isobel F, Turner JC, Rothwell, Ward NS (2015c) Post-stroke fatigue: a deficit in Corticomotor Excitability? Brain 138(Pt 1):136–148. https://doi.org/10.1093/brain/awu306
Kuppuswamy A, Clark E, Rothwell J, and Nick S. Ward (2016) Limb heaviness: a perceptual Phenomenon Associated with poststroke fatigue? Neurorehabilit Neural Repair 30(4):360–362. https://doi.org/10.1177/1545968315597071
Kuppuswamy A, Harris AM, Doncker WD, Alexander A, and Nilli Lavie (2022) Diminished distractor filtering with increased perceptual load and sustained effort explains attention deficit in post-stroke fatigue. https://doi.org/10.1101/2022.03.17.484709. ” bioRxiv
Kutlubaev MA, Duncan FH, Mead GE (2012) Biological correlates of post-stroke fatigue: a systematic review. Acta Neurol Scand 125(4):219–227. https://doi.org/10.1111/j.1600-0404.2011.01618.x
Lefaucheur JP, Lucas B, Andraud F, Hogrel JY, Bellivier F, Del Cul A, Rousseva A, Leboyer M, Paillère-Martinot ML (2008) Inter-hemispheric asymmetry of Motor Corticospinal excitability in Major Depression studied by Transcranial magnetic stimulation. J Psychiatr Res 42(5):389–398. https://doi.org/10.1016/j.jpsychires.2007.03.001
Li L, Cui Z, Li, Wang (2022a) A more female-characterized resting-state brain: graph similarity analyses of sex influence on the human brain intrinsic functional network. Brain Topogr 35(3):341–351. https://doi.org/10.1007/s10548-022-00900-5
Li Y, Yang L, Li L, Xie Y, and Peng Fang (2022b) The resting-state cerebro-cerebellar function Connectivity and Associations with Verbal Working Memory performance. Behav Brain Res 417(January):113586. https://doi.org/10.1016/j.bbr.2021.113586
Liepert J, Mingers D, Heesen C, Bäumer T, and C Weiller (2005) Motor Cortex excitability and fatigue in multiple sclerosis: a transcranial magnetic stimulation study. Mult Scler 11(3):316–321
Liu Y, Ren X, Zeng M, Li J, Zhao X, Zhang X, and Juan Yang (2022) Resting-state dynamic functional connectivity predicts the psychosocial stress response. Behav Brain Res 417:113618. https://doi.org/10.1016/j.bbr.2021.113618
Morgante F, Dattola V, Crupi D, Russo M, Rizzo V, Ghilardi MF, Terranova C, Girlanda P, and Angelo Quartarone (2011) Is central fatigue in multiple sclerosis a disorder of Movement Preparation? J Neurol 258(2):263–272. https://doi.org/10.1007/s00415-010-5742-x
Netz J, Ziemann U, Hömberg V (1995) Hemispheric Asymmetry of Transcallosal Inhibition in Man. Exp Brain Res 104(3):527–533. https://doi.org/10.1007/BF00231987
Nordin L, Engström MC, Möller P, Julin A, Bartfai F, Hashim, and Tie-Qiang Li (2016) Post MTBI fatigue is Associated with abnormal brain functional connectivity. Sci Rep 6(February):21183. https://doi.org/10.1038/srep21183
Ondobaka S, Doncker WD, Ward N (2021) and Annapoorna Kuppuswamy. “Neural Effective Connectivity Explains Subjective Fatigue in Stroke.” Brain: A Journal of Neurology, November, awab287. https://doi.org/10.1093/brain/awab287
Pascual-Marqui RD (2002) Standardized low-resolution brain electromagnetic tomography (SLORETA): Technical Details. Methods Find Exp Clin Pharmacol 24(Suppl D):5–12
Pascual-Marqui RD (2007) “Instantaneous and Lagged Measurements of Linear and Nonlinear Dependence between Groups of Multivariate Time Series: Frequency Decomposition.” ArXiv:0711.1455 [Stat], November. http://arxiv.org/abs/0711.1455
Porcaro C, Cottone C, Cancelli A, Rossini PM, Zito G, and Franca Tecchio (2019) Cortical Neurodynamics Changes mediate the efficacy of a personalized neuromodulation against multiple sclerosis fatigue. Sci Rep 9(1):18213. https://doi.org/10.1038/s41598-019-54595-z
Rubinov M, and Olaf Sporns (2010) Complex Network Measures of Brain Connectivity: uses and Interpretations. NeuroImage 52(3):1059–1069. https://doi.org/10.1016/j.neuroimage.2009.10.003
Russo M, Calamuneri A, Cacciola A, Bonanno L, Naro A, Dattola V, Sessa E et al (2017) Neural correlates of fatigue in multiple sclerosis: a Combined Neurophysiological and Neuroimaging Approach (R1). Arch Ital Biol 155(3):142–151
Snaith RP (2003) The hospital anxiety and Depression Scale. Health Qual Life Outcomes 1:29. https://doi.org/10.1186/1477-7525-1-29
Stefancin P, Govindarajan ST, Krupp L, Charvet L, Timothy QD (2019) Resting-State Functional Connectivity Networks Associated with fatigue in multiple sclerosis with early age onset. Multiple Scler Relat Disorders 31(June):101–105. https://doi.org/10.1016/j.msard.2019.03.020
Stumme J, Jockwitz C, Hoffstaedter F, Amunts K, and Svenja Caspers (2020) Functional Network reorganization in older adults: graph-theoretical analyses of Age, Cognition and Sex. NeuroImage 214:116756. https://doi.org/10.1016/j.neuroimage.2020.116756
Valko PO, Bassetti CL, Konrad E, Bloch U, Held, and Christian R. Baumann (2008) Validation of the fatigue severity scale in a swiss cohort. Sleep 31(11):1601–1607
Wahlheim CN, Alexander P, Christensen ZM, Reagh, and Brittany S. Cassidy (2022) Intrinsic functional connectivity in the default Mode Network predicts Mnemonic discrimination: a connectome-based modeling Approach. Hippocampus 32(1):21–37. https://doi.org/10.1002/hipo.23393
Werf SP, van der PJ, Jongen GJ, Lycklama à Nijeholt F, Barkhof OR, Hommes, Bleijenberg G (1998) Fatigue in multiple sclerosis: interrelations between fatigue complaints, cerebral MRI abnormalities and neurological disability. J Neurol Sci 160(2):164–170
Winward C, Sackley C, Metha Z, Peter MR (2009) A Population-Based study of the prevalence of fatigue after transient ischemic attack and minor stroke. Stroke 40(3):757–761. https://doi.org/10.1161/STROKEAHA.108.527101
Woodward KE, Carlson HL, Kuczynski A, Saunders J, Hodge J, Kirton A (2019) Sensory-Motor Network Functional Connectivity in Children with Unilateral Cerebral Palsy secondary to Perinatal Stroke. NeuroImage Clin 21:101670. https://doi.org/10.1016/j.nicl.2019.101670
Acknowledgements
We would like to thank Prof Franca Tecchio, Dr Pierepaolo Croche and Dr Massimo Bertoli for the many discussions and input into this manuscript. While the intention of this paper was to include a multiple sclerosis resting state dataset from Prof Tecchio’s group, this did not come to fruition due to several incompatibilities. Nevertheless, the highly enthusiastic discussions were key to bringing this paper to print and must be acknowledged.
Funding
Wellcome trust: 202346/Z/16/Z.
Author information
Authors and Affiliations
Contributions
CW performed the functional connectivity analyses, and wrote the manuscript. WD performed data collection in stroke cohort, produced the figures and performed the statistical analyses. AK obtained funding, wrote and revised the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Communicated by Glenn Wylie.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wu, CH., De Doncker, W. & Kuppuswamy, A. Electroencephalography-Derived Functional Connectivity in Sensorimotor Networks in Post Stroke Fatigue. Brain Topogr 36, 727–735 (2023). https://doi.org/10.1007/s10548-023-00975-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10548-023-00975-8