Abstract
Objectives
Glycogen storage disease type 2(GSD2)/Pompe disease is characterized by respiratory and skeletal muscle weakness and atrophy, resulting in functional disability and reduced life span.
Methods
We present an open-label, investigator-initiated observational study of alglucosidase alfa enzyme replacement therapy (ERT) in 38 adult-onset GSD2 patients (20 female, 18 male) with a mean age at disease onset of 36.2 ± 10.5 years. Mean delay between symptom onset and start of ERT was 14.5 ± 7.2 years. Assessments included serial Walton Gardner Medwin scale, arm function tests, timed 10-meter walk tests, 4-stair climb tests, modified Gowers’ maneuvers, 6-minute walk test (6MWT), MRC sum score, forced vital capacities (FVC), creatine kinase (CK) levels, and SF-36 self-reporting questionnaires. All tests were performed at baseline and every 12 months for 36 months of ERT.
Results
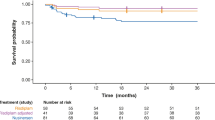
In the 6MWT we found 21 patients able to walk at baseline a mean distance of 312 ± 165.5 m, improving to 344 ± 165.8 m after 12 months (p = 0.006), remaining at 356.4 ± 155.9 m at 24 months (p = 0.033), and declining to 325.6 ± 174.8 m after 36 months of ERT (p = 0.49, n.s.). The mean FVC in 28 patients was 80.27 ± 14.08% of predicted normal at baseline, after 12 months 79.19 ± 13.09%, at 24 months 78.62 ± 16.55%, and 77.19 ± 18.05% after 36 months. Only mean CK levels were significantly decreased by 8.8% (p = 0.041). All other tests were statistically non-significant changed.
Conclusion
Our data denote a rather variable course of neuromuscular deficits in chronic adult-onset Pompe patients during 36 months of alglucosidase alfa ERT.


Similar content being viewed by others
References
Angelini C, Semplicini C, Ravaglia S, et al. The Italian GSDII Group. (2011) Observational clinical study in juvenile-adult glycogenosis type 2 patients undergoing enzyme replacement therapy for up to 4 years. J Neurol. PMID:22081099
Ausems MG, Wokke JH, Reuser AJ, van Diggelen OP (2001) Juvenile and adult-onset acid maltase deficiency in France: genotype-phenotype correlation. Neurology 57(10):1938
Bali DS, Tolun AA, Goldstein JL, Dai J, Kishnani PS (2011) Molecular analysis and protein processing in late-onset Pompe disease patients with low levels of acid alpha-glucosidase activity. Muscle Nerve 43(5):665–670
Banati M, Hosszu Z, Trauninger A, Szereday L, Illes Z (2011) Enzyme replacement therapy induces T-cell responses in late-onset POMPE disease. Muscle Nerve 44:720–726
Banugaria SG, Prater NA, Ng YK et al (2011) The impact of antibodies on clinical outcomes in diseases treated with therapeutic protein: Lessons learned from infantile Pompe disease. Genet Med 13:729–736
Bembi B, Pisa FE, Confalonieri M et al (2010) Long-term observational, non-randomized study of enzyme replacement therapy in late-onset glycogenosis type II. J Inherit Metab Dis 33:727–735
Case LE, Koeberl DD, Young SP et al (2008) Improvement with ongoing Enzyme Replacement Therapy in advanced late-onset Pompe disease: a case study. Mol Genet Metab 95:233–235
DeRuisseau LR, Fuller DD, Qiu K et al (2009) Neural deficits contribute to respiratory insufficiency in Pompe disease. Proc Natl Acad Sci U S A 106(23):9419–9424
de Vries JM, van der Beek NAME, Kroos MA et al (2010) High antibody titer in an adult with Pompe disease affects treatment with alglucosidase alfa. Mol Genet Metab 101:338–345
Fukuda T, Ewan L, Bauer M et al (2006) Dysfunction of endocytic and autophagic pathways in a lysosomal storage disease. Ann Neurol 59(4):700–708
Furusawa Y, Mori-Yoshimura M, Yamamoto T, et al (2011) Effects of enzyme replacment therapy on five patients with advanced late-onset glycogen storage disease type II : a 2- year follow-up study. J Inherit Metab Dis PMID: 21984055
Hermans MM, van Leenen D, Kroos MA et al (2004) Twenty-two novel mutations in the lysosomal alpha-glucosidase gene (GAA) underscore the genotype-phenotype correlation in glycogen storage disease type II. Hum Mutat 23(1):47–56
Joshi PR, Glaser D, Schmidt S et al (2008) Molecular diagnosis of German patients with late-onset glycogen storage disease type II. J Inherit Metab Dis 2(Suppl):261–265
Kroos MA, Pomponio RJ, Hagemans ML et al (2007) Broad spectrum of Pompe disease in patients with the same c.-32-13T->G haplotype. Neurology 68(2):110–115
Kroos M, Pomponio RJ, van Vliet L et al (2008) Update of the Pompe disease mutation database with 107 sequence variants and a format for severity rating. Hum Mutat 29(6):E13–26
Laforet P, Nicolino M, Eymard PB et al (2000) Juvenile and adult-onset acid maltase deficiency in France: genotype-phenotype correlation. Neurology 55(8):1122–1128
Müller-Felber W, Horvath R, Gempel K et al (2007) Late onset Pompe disease: clinical and neurophysiological spectrum of 38 patients including long-term follow-up in 18 patients. Neuromuscul Disord 17:698–706
Raben N, Roberts A, Plotz PH (2007) Role of autophagy in the pathogenesis of Pompe disease. Acta Myol 26(1):45–48
Raben N, Ralston E, Chien YH et al (2010a) Differences in the predominance of lysosomal and autophagic pathologies between infants and adults with Pompe disease: implications for therapy. Mol Genet Metab 101(4):324–331
Raben N, Schreiner C, Baum R et al (2010b) Suppression of autophagy permits successful enzyme replacement therapy in a lysosomal storage disorder–murine Pompe disease. Autophagy 6(8):1078–1089
Roberts M, Kishnani PS, van der Ploeg AT et al (2011) The Impact of Scoliosis in individuals with Pompe disease: lessons learned from the Pompe registry. Mol Genet Metab 104(4):574–582
Schoser B, Muller-Hocker J, Horvath R et al (2007) Adult-onset glycogen storage disease type 2: clinico-pathological phenotype revisited. Neuropathol Appl Neurobiol 33(5):544–559
Schoser B, Hill V, Raben N (2008) Therapeutic approaches in glycogen storage disease type II/Pompe disease. Neurotherapeutics 5:569–578
Schüller A, Wenninger S, Strigl-Pill N, Schoser B (2012) Towards deconstructing the phenotype of late-onset Pompe disease. Am J Med Genet C doi: 10.1002/ajmg.c.31322.
Strothotte S, Strigl-Pill N, Grunert B et al (2010) Enzyme replacement therapy with alglucosidase alfa in 44 patients with late-onset glycogen storage disease type 2: 12- month results of an observational clinical trial. J Neurol 257:91–97
Swash M, Schwartz MS, Apps MC (1985) Adult onset acid maltase deficiency. Distribution and progression of clinical and pathological abnormality in a family. J Neurol Sci 68:61–74
Van Capelle CI, Winkel LP, Hagemans ML et al (2008) Eight years experience with enzyme replacement therapy in two children and one adult with Pompe disease. Neuromuscul Disord 18:447–452
Van Capelle CI, van der Beek NA, Hagemans ML et al (2010) Effect of enzyme therapy in juvenile patients with Pompe disease: a three-year open-label study. Neuromuscul Disord 20:775–782
Van der Beek NA, Hagemans ML, Reuser AJ, Hop WC, Van der Ploeg AT, Van Doorn PA, Wokke JH (2009) Rate of disease progression during long-term follow-up of patients with late-onset Pompe disease. Neuromuscul Disord 19:113–117
Van der Beek NA, van Capelle CI, van der Velden KI et al (2011) Rate of progression and predictive factors for pulmonary outcome in children and adults with Pompe disease. Mol Genet Metab 104:129–136
Van der Ploeg AT, Reuser AJ (2008) Pompe’s disease. Lancet 372:1342–1353
Van der Ploeg AT, Clemens PR, Corzo D et al (2010) A randomized study of alglucosidase alfa in late- onset Pompe’s disease. N Engl J Med 362:1396–1406
Vielhaber S, Brejova A, Debska-Vielhaber G et al (2011) 24-months results in two adults with Pompe disease on enzyme replacement therapy. Clin Neurol Neurosurg 113:350–357
Winkel LP, Hagemans ML, van Doorn PA et al (2005) The natural course of non-classic Pompe’s disease; a review of 225 published cases. J Neurol 252:875–884
Yang CC, Chien YH, Lee NC et al (2011) Rapid progressive course of later-onset Pompe disease in Chinese patients. Mol Genet Metab 104(3):284–288
Acknowledgment
We thank the patients and their families for their patience and long-term cooperation. We also thank the technical team at the involved treatment centers. Research conducted in MC Johannes-Gutenberg-University, Mainz, Germany was supported by the European Union, 7th Framework Programme ‘EUCLYD- A European Consortium for Lysosomal Storage Diseases’ [health F2/2008 grant agreement 201678 to EM].
Competing interest
M. Deschauer, C. Kornblum, E. Mengel, W. Müller-Felber, M. Spranger, B. Schoser, S. Vielhaber have received lecturer honoraria from Genzyme Corporation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Ed Wraith
Rights and permissions
About this article
Cite this article
Regnery, C., Kornblum, C., Hanisch, F. et al. 36 months observational clinical study of 38 adult Pompe disease patients under alglucosidase alfa enzyme replacement therapy. J Inherit Metab Dis 35, 837–845 (2012). https://doi.org/10.1007/s10545-012-9451-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-012-9451-8