Abstract
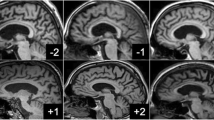
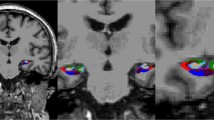
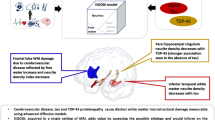
Cerebral micro- and macro-vasculopathy have been described in Fabry disease (FD). Neuronal globotriaosylceramide accumulation in selective cortical and brain stem areas including the hippocampus has been reported by autopsy studies in FD, but clinical surrogates as well as the clinical relevance of these findings have not been investigated so far. We measured the hippocampus volumes in a group of clinically affected patients with FD and correlated the findings with the cognitive performance of the patients. Hippocampal volumes were determined manually on T1-weighted MR-images of 25 FD patients (age 36.5 ± 11.0 years) and 20 age-matched controls. Additionally, individual white matter (WM) and gray matter (GM) volumes were measured using brain segmentation analyses. After controlling for age, white matter lesion (WML) volume, and WM/GM-volumes hippocampal volumes were significantly decreased in FD. These findings were substantially more pronounced in a subgroup of men with FD. WM and WM/GM volumes, and memory function did not significantly differ between patients and controls. In patients with FD hippocampal volumes were neither significantly correlated to WML volume nor to WM or WM/GM volumes. Hippocampus atrophy was not driven by the WML or other brain tissue atrophy and seems to correlate with the neuronal involvement in FD. In this young to middle-aged Fabry cohort the hippocampus degeneration was functionally compensated without memory impairment. Longitudinal studies are needed to determine whether this degenerative component in FD will progress and, in concert with the individual WML-load, predict subsequent cognitive decline.


Similar content being viewed by others
References
Albrecht J, Dellani PR, Müller MJ et al (2007) Voxel based analyses of diffusion tensor imaging in Fabry disease. J Neurol Neurosurg Psychiatry 78:964–969
Ashburner J, Friston KJ (2000) Voxel-based morphometry–the methods. NeuroImage 11:805–821
Crutchfield KE, Patronas NJ, Dambrosia JM et al (1998) Quantitative analysis of cerebral vasculopathy in patients with Fabry disease. Neurology 50:1746–1749
Desnick RJ, Brady R, Barranger J et al (2003) Fabry disease, an under-recognized multisystemic disorder: expert recommendations for diagnosis, management, and enzyme replacement therapy. Ann Intern Med 138:338–346
deVeber GA, Schwarting GA, Kolodny EH et al (1992) Fabry disease: immunocytochemical characterization of neuronal involvement. Ann Neurol 31:409–415
Fellgiebel A, Müller MJ, Mazanek M et al (2005) White matter lesion severity in male and female patients with Fabry disease. Neurology 65:600–602
Fellgiebel A, Muller MJ, Ginsberg L (2006) CNS manifestations of Fabry's disease. Lancet Neurol 5:791–795
Fellgiebel A, Keller I, Marin D et al (2009) Diagnostic utility of different MRI and MR angiography measures in Fabry disease. Neurology 72:63–68
Godin O, Maillard P, Crivello F et al (2009) Association of white-matter lesions with brain atrophy markers: the three-city Dijon MRI study. Cerebrovasc Dis 28:177–184
Hamilton M (1986) The Hamilton Rating Scale for Depression. In: Sartorius N, Ban TA (eds) Assessment of depression. Springer Verlag, Berlin, pp 143–152
Kaye EM, Kolodny EH, Logigian EL et al (1988) Nervous system involvement in Fabry's disease: clinicopathological and biochemical correlation. Ann Neurol 23:505–509
Mitsias P, Levine SR (1996) Cerebrovascular complications of Fabry's disease. Ann Neurol 40:8–17
Moore DF, Ye F, Schiffmann R et al (2003) Increased signal intensity in the pulvinar on T1-weighted images: a pathognomonic MR imaging sign of Fabry disease. Am J Neuroradiol 24:1096–1101
Niemann K, Hammers A, Coenen VA et al (2000) Evidence of a smaller left hippocampus and left temporal horn in both patients with first episode schizophrenia and normal control subjects. Psychiatry Res 99:93–110
Nitkunan A, Lanfranconi S, Charlton RA et al (2010) Brain atrophy and cerebral small vessel disease: a prospective follow-up study. Stroke 42:133–138
Okeda R, Nisihara M (2008) An autopsy case of Fabry disease with neuropathological investigation of the pathogenesis of associated dementia. Neuropathology 28:532–540
Schermuly I, Müller MJ, Müller KM et al (2010) Neuropsychiatric symptoms and brain structural alterations in Fabry disease. Eur J Neurol 18:347–353
Takanashi J, Barkovich AJ, Dillon WP, Sherr EH, Hart KA, Packman S (2003) T1 hyperintensity in the pulvinar: key imaging feature for diagnosis of Fabry disease. AJNR Am J Neuroradiol 24:916–921
Whybra C, Kampmann C, Krummenauer F et al (2004) The Mainz Severity Score Index: a new instrument for quantifying the Anderson-Fabry disease phenotype, and the response of patients to enzyme replacement therapy. Clin Genet 65:299–307
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Gregory M. Pastores
Competing interest: None declared
Rights and permissions
About this article
Cite this article
Fellgiebel, A., Wolf, D.O., Kolodny, E. et al. Hippocampal atrophy as a surrogate of neuronal involvement in Fabry disease. J Inherit Metab Dis 35, 363–367 (2012). https://doi.org/10.1007/s10545-011-9390-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-011-9390-9