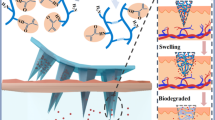
Abstract
To enhance the compliance of drug delivery for patients, the novel near-infrared (NIR) light-triggered and separable microneedles (MNs) have been developed in this work. Firstly, prussian blue nanoparticles (PB NPs) as the photo-thermal conversion factor and metformin as the hypoglycemic drug were embedded into the separable arrowheads, which consisted by poly (vinyl alcohol) and sucrose (PVA/Suc). The arrowheads of MNs were located on soluble solids supporting substrates that produced by poly(vinyl pyrrolidone) (PVP). Lauric acid (LA) as the phase transition coating covered on the surface of the MNs due to its lower phase transition temperature (~44 °C). Then, the separable arrowheads could be left into the skin because of the absorbing the interstitial fluid (IF) by the solid supporting substrates. With the irradiation of NIR light, LA could be melted due to the role of PB NPs in photo-thermal conversion, thus releasing the metformin from arrowheads. Compared with the traditional subcutaneous injections, the hypoglycemic effect was evaluated by the drug-release behaviors induced by NIR in vivo. The results showed that metformin could be allowed to on-demand release under the NIR irradiation. And the as-obtained MNs exhibited a good hypoglycemic effect, hypotoxicity and low inflammation reaction compared with those of traditional subcutaneous injections. The results indicate that the fabricated MNs have the potential treatment for diabetes due to their safety, convenience and painlessness.






Similar content being viewed by others
References
N.K. Archer, J.-H. Jo, S.K. Lee, D. Kim, B. Smith, R.V. Ortines, Y. Wang, M.C. Marchitto, A. Ravipati, S.S. Cai, C.A. Dillen, H. Liu, R.J. Miller, A.G. Ashbaugh, A.S. Uppal, M.K. Oyoshi, N. Malhotra, S. Hoff, L.A. Garza, H.H. Kong, J.A. Segre, R.S. Geha, L.S. Miller, J. Allergy Clin. Immunol. 143, 1426–1443 (2019)
Y. Cao, Y. Chen, T. Yu, Y. Guo, F. Liu, Y. Yao, P. Li, D. Wang, Z. Wang, Y. Chen, H. Ran, Theranostics 8, 1327–1339 (2018)
X. Chen, G.J.P. Fernando, M.L. Crichton, C. Flaim, S.R. Yukiko, E.J. Fairmadi, H.J. Corbett, C.A. Primiero, A.B. Ansaldo, L.H. Frazer, L.E. Brown, M.A.F.J. Kendall, J. Control. Release 152, 349–355 (2011)
M.-C. Chen, M.-H. Ling, K.-W. Wang, Z.-W. Lin, B.-H. Lai, D.-H. Chen, Biomacromolecules 16, 1598–1607 (2015)
M.-C. Chen, H.-A. Chan, M.-H. Ling, L.-C. Su, J. Mater. Chem. B 3, 496–503 (2017)
C. Chen, Y. Liu, H. Wanag, G. Chen, X. Wu, J. Ren, H. Zhagn, Y. Zhao, ACS Nano 12, 10493–10500 (2018a)
C.-H. Chen, V.B.-H. Shyu, C.-T. Chen, Materials 11, 1625–1635 (2018b)
A.M. de Groot, G. Du, J. Monkare, A.C.M. Platteel, F. Broere, J.A. Bouwstra, A.J.A.M. Sijts, J. Control. Release 266, 27–35 (2017)
M. Frenz, C. Misehler, V. Romano, M. Forrer, O.M. Miiller, Appl. Phys. B Lasers Opt. 52, 251–258 (1991)
A. GhacamiNejad, J. Li, B. Lu, L. Zhou, L. Lam, A. Giacca, X. Wu, Adv. Mater. 31, 1901051 (2019)
H.S. Gill, M.R. Prausnitz, J. Control. Release 117, 227–237 (2007)
Y. Hao, M. Dong, T. Zhang, J. Peng, Y. Jia, Y. Cao, Z. Qian, ACS Appl. Mater. Interfaces 9, 15317–15327 (2017a)
Y. Hao, W. Li, X. Zhou, F. Yang, Z. Qian, J. Biomed. Nanotechnol. 13, 1581–1597 (2017b)
B.A. Jana, A.D. Wadhwani, Indian J. Pharmacol. 51, 4–10 (2019)
V.R. Jayaneththi, K. Aw, M. Sharma, J. Wen, D. Svirskis, A.J. Mcdaid, Sens. Actuators B-Chem. 297, 126708 (2019)
G. Jiang, B. Xu, J. Zhu, Y. Zhang, T. Liu, G. Song, Biomed. Phys. Eng. Express 5, 045038 (2019)
L. Jing, S. Shao, Y. Wang, Y. Yang, X. Yue, Z. Dai, Theranostics 6, 40–53 (2016)
S. Lau, J. Fei, H. Liu, W. Chen, R. Liu, J. Control. Release 265, 113–119 (2016)
B. Laverdet, D. Girard, A. Bayout, N. Bordeau, C. Demiot, A. Desmouliere, Burns 43, 562–572 (2017)
I.-C. Lee, W.-M. Lin, J.-C. Shu, S.-W. Tsai, C.-H. Chen, M.-T. Tsai, J. Biomed. Mater. Res. A 105, 84–93 (2017)
M.S. Lhernould, C. Gobillon, P. Lambert, Ondrugdelivery 40, 29–32 (2013)
Y. Li, G. Bai, S. Zeng, J. Hao, ACS Appl. Mater. Interfaces 11, 4737–4744 (2019a)
W. Li, R.N. Terry, J. Tang, M.R. Feng, S.P. Schwendeman, M.R. Pranusnitz, Nat. Biomed. Eng. 3, 220–229 (2019b)
E.Q. Littauer, L.K. Mills, N. Brock, E.S. Esser, A. Romanyuk, J.A. Pulit-Penaloza, E.V. Vassilieca, J.T. Beaver, O. Antao, F. Krammer, R.W. Compans, M.R. Prausnitz, I. Skoutzou, J. Control. Release 276, 1–16 (2018)
D. Liu, B. Yu, G. Jiang, W. Yu, Y. Zhang, B. Xu, Mater. Sci. Eng. C 90, 180–188 (2018a)
D. Liu, Y. Zhang, G. Jiang, W. Yu, B. Xu, J. Zhu, ACS Biomater. Sci. Eng. 4, 1687–1695 (2018b)
C. Liu, S. Zhang, J. Li, J. Wei, K. Miillen, M. Yin, Angew. Chem.-Int. Edit. 58, 1638–1642 (2019)
K. Migalska, D.I.J. Morrow, M.J. Garland, R. Thakur, A. David Woolfson, R.F. Donnelly, Pharm. Res. 28, 1919–1930 (2011)
T. Miyano, Y. Tobinaga, T. Kanno, Y. Matsuzaki, H. Takeda, M. Wakui, K. Hanada, Biomed. Microdevices 7, 185–188 (2005)
B. Moller, P.M. Villiger, Springer Semin. Immunopathol. 27, 391–408 (2006)
A.H. Montoto, A. Llopis-Lorente, M. Gorbe, J.M. Terres, R. Cao-Milan, B.D. de Grenu, M. Alfonso, Chem. Eur. J. 25, 8471–8478 (2019)
L. Niu, L.Y. Chu, S.A. Burton, K.J. Hansen, J. Panyam, J. Control. Release 294, 268–278 (2019)
Z. Qin, Y. Li, N. Gu, Adv. Healthc. Mater. 7, 1800347 (2018)
M.J. Reed, K. Meszaros, L.J. Entes, M.D. Claypool, J.G. Pinkett, T.M. Gadois, G.M. Reaven, Metabolism 49, 1390–1394 (2000)
S. Skovso, J. Damgaard, J.J. Fels, G.S. Olsen, X.A. Wolf, J.J. Holst, Int. J. Obes. 39, 1531–1538 (2018)
S.P. Sulivan, N. Murthy, M.R. Pranusnitz, Adv. Mater. 20, 933–938 (2008)
T. Tanakal, M. Narazaki, T. Kishimoto, Cold Spring Harb. Perspect. Biol. 6, a016295 (2014)
B.P. Timko, T. Dvir, D.S. Kohane, Adv. Mater. 22, 4925–4943 (2010)
S. Wild, R. Sicree, G. Roglic, H. King, A. Green, Diabetes Care 27, 1047–1053 (2004)
Y. Xia, B. Liu, P. Ye, B. Xu, Appl. Therm. Eng. 142, 524–529 (2018)
B. Xu, Q. Cao, Y. Zhang, W. Yu, J. Zhu, D. Liu, G. Jiang, ACS Biomater. Sci. Eng. 4, 2473–2483 (2018a)
B. Xu, G. Jiang, W. Yu, D. Liu, Y. Zhang, J. Zhou, S. Sun, Y. Liu, J. Mater. Chem. B 5, 8200–8208 (2018b)
J. Xu, B. Xia, X. Niu, J. Cai, Z. Han, Q. Wang, X. Lu, Q. Fan, W. Huang, Dyes Pigments 170, 107664 (2019)
J. Yang, Z. Chen, R. Ye, J. Li, Y. Lin, J. Gao, L. Ren, B. Liu, L. Jiang, Drug Deliv. 25, 1728–1739 (2018)
W. Yu, G. Jiang, D. Liu, L. Li, Z. Tong, J. Yao, X. Kong, Mater. Sci. Eng. C 73, 425–428 (2015)
W. Yu, G. Jiang, D. Liu, L. Li, H. Chen, Q. Huang, Z. Tong, J. Yao, X. Kong, Mater. Sci. Eng. C 71, 725–734 (2017a)
W. Yu, G. Jiang, Y. Zhang, D. Liu, B. Xu, J. Zhou, J. Mater. Chem. B 5, 9507–9513 (2017b)
K. Zeng, M. Yang, Y.-N. Liu, A. Rasooly, Anal. Methods 10, 3951–3957 (2018)
Y. Zhang, D. Wang, M. Gao, B. Xu, J. Zhu, W. Yu, D. Liu, G. Jiang, ACS Biomater. Sci. Eng. 4, 2879–2888 (2018)
Y. Zhang, D. Chai, M. Gao, W. Yu, D. Liu, B. Xu, G. Jiang, Int. J. Polym. Mater. Polym. Biomater. 68, 850–858 (2019)
C. Zhou, X. Xie, H. Yang, S. Zhang, Y. Li, C. Kuang, S. Fu, L. Cui, M. Liang, C. Gao, Y. Yang, C. Gao, C. Yang, Mol. Pharm. 16, 2956–2965 (2019)
C. Zhu, D. Huo, Q. Chen, J. Xue, S. Shen, Y. Xia, Adv. Mater. 29, 1703702 (2017)
Funding
This work was financially supported by the National Natural Science Foundation of China (51873194, 51373155), Natural Science Foundation of Zhejiang Province (LY18E030006) and “521 Talents Training Plan” in Zhejiang Sci-Tech University (ZSTU). We also gratefully acknowledge Hua-An Biotechnology Co., Ltd. (Hangzhou, China) for histological experiments.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 2548 kb)
Rights and permissions
About this article
Cite this article
Liu, T., Jiang, G., Song, G. et al. Fabrication of separable microneedles with phase change coating for NIR-triggered transdermal delivery of metformin on diabetic rats. Biomed Microdevices 22, 12 (2020). https://doi.org/10.1007/s10544-019-0468-8
Published:
DOI: https://doi.org/10.1007/s10544-019-0468-8