Abstract
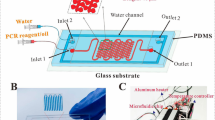
In the field of continuous-flow PCR, the amplification throughput in a single reaction solution is low and the single-plex PCR is often used. In this work, we reported a flow-based multiplex PCR microfluidic system capable of performing high-throughput and fast DNA amplification for detection of foodborne bacterial pathogens. As a demonstration, the mixture of DNA targets associated with three different foodborne pathogens was included in a single PCR solution. Then, the solution flowed through microchannels incorporated onto three temperature zones in an oscillatory manner. The effect factors of this oscillatory-flow multiplex PCR thermocycling have been demonstrated, including effects of polymerase concentration, cycling times, number of cycles, and DNA template concentration. The experimental results have shown that the oscillatory-flow multiplex PCR, with a volume of only 5 μl, could be completed in about 13 min after 35 cycles (25 cycles) at 100 μl/min (70 μl/min), which is about one-sixth of the time required on the conventional machine (70 min). By using the presently designed DNA sample model, the minimum target concentration that could be detected at 30 μl/min was 9.8 × 10−2 ng/μl (278-bp, S. enterica), 11.2 × 10−2 ng/μl (168-bp, E. coli O157: H7), and 2.88 × 10−2 ng/μl (106-bp, L. monocytogenes), which corresponds to approximately 3.72 × 104 copies/μl, 3.58 × 104 copies/μl, and 1.79 × 104 copies/μl, respectively. This level of speed and sensitivity is comparable to that achievable in most other continuous-flow PCR systems. In addition, the four individual channels were used to achieve multi-target PCR analysis of three different DNA samples from different food sources in parallel, thereby achieving another level of multiplexing.







Similar content being viewed by others
References
P.A. Auroux, P.J.R. Day, F. Niggli, A. Manz, NSTI Nanotech 2003, San Francisco, CA, February 23–27; pp. 55–58 (2003a).
P.A. Auroux, F.K. Niggli, A. Manz, P.J.R. Day, Nanotech. 1, 55–58 (2003b)
P.-A. Auroux, P.J.R. Day, A. Manz, NSTI Nanotech 2004, Boston, MA, March 7–11; pp. 67–69 (2004a)
P.A. Auroux, Y. Koc, A. deMello, A. Manz, P.J. Day, Lab Chip 4, 534–546 (2004b)
J. Baker, M. Strachan, K. Swartz, Y. Yurkovetsky, A. Rulison, C. Brooks, A. Kopf-Sill, μTAS 2003, Squaw Valley, CA, October 5–9; pp. 1335–1338 (2003)
M. Bu, T. Melvin, G. Ensell, J.S. Wilkinson, A.G.R. Evans, J. Micromech. Microeng. 13, S125–S130 (2003)
L. Chen, A. Manz, P.J. Day, Lab Chip 7, 1413–1423 (2007a)
L. Chen, J. West, P.A. Auroux, A. Manz, P.J.R. Day, Anal. Chem. 79, 9185–9190 (2007b)
J.Y. Cheng, C.J. Hsieh, Y.C. Chuang, J.R. Hsieh, Analyst 130, 931–940 (2005)
L.J. Chien, J.H. Wang, T.M. Hsieh, P.H. Chen, P.J. Chen, D.S. Lee, C.H. Luo, G.B. Lee, Biomed. Microdevices 11, 359–367 (2009)
J. Chiou, P. Matsudaira, A. Sonin, D. Ehrlich, Anal. Chem. 73, 2018–2021 (2001)
J.T. Chiou, P.T. Matsudaira, D.J. Ehrlich, Biotechniques 33, 557–564 (2002)
O. Frey, S. Bonneick, A. Hierlemann, J. Lichtenberg, Biomed. Microdevices 9, 711–718 (2007)
S. Hardt, D. Dadic, F. Doffing, K.S. Drese, G. Munchow, O. Sorensen, NSTI Nanotech 2004, Boston, MA, March 7–11; pp. 55–58 (2004)
R. Hartung, A. Brösing, G. Sczcepankiewicz, U. Liebert, N. Häfner, M. Dürst, J. Felbel, D. Lassner, J.M. Köhler, Biomed. Microdevices 11, 685–692 (2009)
O. Henegariu, N.A. Heerema, S.R. Dlouhy, G.H. Vance, P.H. Vogt, Biotechniques 23, 504–511 (1997)
G.Q. Hu, Q. Xiang, R. Fu, B. Xu, R. Venditti, D.Q. Li, Anal. Chim. Acta 557, 146–151 (2006)
Z. Hua, J.L. Rouse, A.E. Eckhardt, V. Srinivasan, V.K. Pamula, W.A. Schell, J.L. Benton, T.G. Mitchell, M.G. Pollack, Anal. Chem. 82, 2310–2316 (2010)
D.Y. Kong, T.W. Kang, C.T. Seo, C.S. Cho, J.H. Lee, Biochip J. 4, 179–183 (2010)
M.U. Kopp, A.J. de Mello, A. Manz, Science 280, 1046–1048 (1998)
G. Münchow, D. Dadic, F. Doffing, S. Hardt, K.S. Drese, Expert Rev. Mol. Diagn. 5, 613–620 (2005)
M.A. Northrup, M.T. Ching, R.M. White, R.T. Watson, Transducers’93, Yokohama, Japan, June 7–10; pp. 924–926 (1993)
P.J. Obeid, T.K. Christopoulos, H.J. Crabtree, C.J. Backhouse, Anal. Chem. 75, 288–295 (2003)
T. Ohashi, H. Kuyama, N. Hanafusa, Y. Togawa, Biomed. Microdevices 9, 695–702 (2007)
P.Y. Paik, D.J. Allen, A.E. Eckhardt, V.K. Pamula, M.G. Pollack, μTAS 2007, Paris, France, October 7–11; pp. 1559–1561 (2007)
J.R. Peham, W. Grienauer, H. Steiner, R. Heer, M.J. Vellekoop, C. Nöhammer, H. Wiesinger-Mayr, Biomed. Microdevices 13, 463–473 (2011)
I. Pjescic, C. Tranter, P.L. Hindmarsh, N.D. Crews, Biomed. Microdevices 12, 333–343 (2010)
A. Polini, E. Mele, A.G. Sciancalepore, S. Girardo, A. Biasco, A. Camposeo, R. Cingolani, D.A. Weitz, D. Pisignano, Biomicrofluidics 4, 036502 (2010)
I. Schneegaβ, R. Bräutigam, J.M. Köhler, Lab Chip 1, 42–49 (2001)
A.G. Sciancalepore, A. Polini, E. Mele, S. Girardo, R. Cingolani, D. Pisignano, Biosens. Bioelectron. 26, 2711–2715 (2011)
R. Sista, Z. Hua, P. Thwar, A. Sudarsan, V. Srinivasan, A. Eckhardt, M. Pollack, V. Pamula, Lab Chip 8, 2091–2104 (2008)
D. Sugumar, A. Ismail, M. Ravichandran, I. Aziah, L.X. Kong, Biomicrofluidics 4, 024103 (2010)
Y. Sun, N.T. Nguyen, Y.C. Kwok, Anal. Chem. 80, 6127–6130 (2008)
K. Ugsornrat, T. Maturus, A. Jomphoak, T. Pogfai, N.V. Afzulpurkar, A. Wisitsoraat, A. Tuantranont, ICBME 2008, Singapore, December 3–6; pp. 859–862 (2008)
W. Wang, Z.X. Li, R. Luo, S.H. Lü, A.D. Xu, Y.J. Yang, J. Micromech. Microeng. 15, 1369–1377 (2005)
C.S. Zhang, D. Xing, Nucleic Acids Res. 35, 4223–4237 (2007)
C.S. Zhang, D. Xing, Biomed. Microdevices 12, 1–12 (2010)
C.S. Zhang, J.L. Xu, W.L. Ma, W.L. Zheng, Biotechnol. Adv. 24, 243–284 (2006)
Acknowledgements
This research is supported by the National Natural Science Foundation of China (61072030, 30700155), the Key Program of NSFC-Guangdong Joint Funds of China (U0931005), and the Program for Changjiang Scholars and Innovative Research Team in University (IRT0829).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. S1
Effects of Taq DNA polymerase concentration on conventional multiplex PCR. Lane M: the DNA marker; Lanes 1-7: the Taq DNA polymerase concentrations used in the conventional multiplex PCR were 0, 0.025, 0.05, 0.1, 0.2, 0.3, and 0.5 unit/μl, respectively. (DOC 119 kb)
Fig. S2
Effect of various concentrations of BSA on oscillatory-flow multiplex PCR. Lanes 1-5: the oscillatory-flow multiplex PCR reactions were performed on the microfluidic device with five different concentrations of BSA (0, 0.3, 0.5, 0.7, and 0.9 μg/μl , respectively); Lane M: the DNA marker. (DOC 91 kb)
Fig. S3
Influence of the cycle number on the oscillatory-flow multiplex PCR in the case of the flow rate of 50 μl/min. Lane 1: the positive-control multiplex PCR product; Lanes 2-5: the oscillatory-flow multiplex PCR was carried out with the cycle number of 35, 30, 25, and 20, respectively; Lane 6: the negative-control multiplex PCR; Lane M: the DNA marker. (DOC 113 kb)
Fig. S4
Oscillatory-flow multiplex PCR from different concentrations of relatively pure DNA samples extracted by using the bacterial DNA extraction kit with a preliminary lysozyme digestion. Lane M: the DNA marker; Lane 1: the positive-control PCR product; Lane 2: the concentration of each bacterial genomic DNA template was 1.0 ng/μl; Lanes 3-8: the concentrations of the used DNA template samples were the 101, 102, 103, 104, 105, and 106-fold dilutions of the DNA template concentration stated in Lane 2; Lane 9: the negative-control multiplex PCR solution without DNA template flowed at 30 μl/min. (DOC 173 kb)
Rights and permissions
About this article
Cite this article
Zhang, C., Wang, H. & Xing, D. Multichannel oscillatory-flow multiplex PCR microfluidics for high-throughput and fast detection of foodborne bacterial pathogens. Biomed Microdevices 13, 885–897 (2011). https://doi.org/10.1007/s10544-011-9558-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10544-011-9558-y