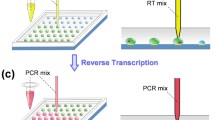
Abstract
Continuous-flow analysis, where samples circulate encapsulated in a carrier fluid is an attractive alternative to batch processing for high-throughput devices that use the polymerase chain reaction (PCR). Challenges of continuous-flow prototypes include the hydrodynamic and biological incompatibility of the carrier fluid, microchannel fouling, sample carryover and the integration of a nucleic acid extraction and reverse transcription step. We tested two homemade, continuous-flow thermocycler microdevices for amplification of reverse-transcribed messages from cell lysates without nucleic acid extraction. Amplification yield and specificity were assessed with state-of-the-art, real-time quantitative equipment. Carryover contamination between consecutive samples was absent. Amplification specificity and interference by genomic DNA were optimized by primer design. Robust detection of the low-copy transcript CLIC5 from 18 cells per microliter is demonstrated in cultured lymphoblasts. The results prove the concept that the development of micro-total analysis systems (μ-TAS) for continuous gene expression directly from cell suspensions is viable with current technology.





Similar content being viewed by others
References
W.A. Al-Soud, P. Radstrom, J. Clin. Microbiol. 39, 485–493 (2001)
L.R. Bisset, S. Bosbach, Z. Tomasik, H. Lutz, J. Schupbach, J. Boni, J. Virol. Methods 91, 149–155 (2001)
M. Brivio, W. Verboom, D.N. Reinhoudt, Lab. Chip. 6, 329–344 (2006)
M. Burgener, U. Candrian, M. Gilgen, J. Virol. Methods 108, 165–170 (2003)
P. Chomczynski, M. Rymaszewski, BioTechniques 40, 454, 456, 458 (2006)
K.D. Dorfman, M. Chabert, J.H. Codarbox, G. Rousseau, P. de Cremoux, J.L. Viovy, Anal. Chem. 77, 3700–3704 (2005)
W.M. Gallagher, O.E. Bergin, M. Rafferty, Z.D. Kelly, I.M. Nolan, E.J. Fox, A.C. Culhane, L. McArdle, M.F. Fraga, L. Hughes, C.A. Currid, F. O’Mahony, A. Byrne, A.A. Murphy, C. Moss, S. McDonnell, R.L. Stallings, J.A. Plumb, M. Esteller, R. Brown, P.A. Dervan, D.J. Easty, Carcinogenesis 26, 1856–1867 (2005)
A.B. Goulter, D.W. Harmer, K.L. Clark, BMC Genomics 7, 34 (2006)
F. Han, S.J. Lillard, Anal. Chem. 72, 4073–4079 (2000)
M. Hashimoto, P.C. Chen, M.W. Mitchell, D.E. Nikitopoulos, S.A. Soper, M.C. Murphy, Lab. Chip. 4, 638–645 (2004)
J. Hoorfar, P. Wolffs, P. Radstrom, Apmis 112, 808–814 (2004)
R.J. Klebe, G.M. Grant, A.M. Grant, M.A. Garcia, T.A. Giambernardi, G.P. Taylor, BioTechniques 21, 1094–1100 (1996)
L.J. Kricka, P. Wilding, Anal. Bioanal. Chem. 377, 820–825 (2003)
M. Kubista, J.M. Andrade, M. Bengtsson, A. Forootan, J. Jonak, K. Lind, R. Sindelka, R. Sjoback, B. Sjogreen, L. Strombom, A. Stahlberg, N. Zoric, Mol. Aspects Med. 27, 95–125 (2006)
L.A. Legendre, J.M. Bienvenue, M.G. Roper, J.P. Ferrance, J.P. Landers, Anal. Chem. 78, 1444–1451 (2006)
D.E. Macfarlane, C.E. Dahle, Nature 362, 186–188 (1993)
H. Matsunaga, T. Anazawa, E.S. Yeung, Electrophoresis 24, 458–465 (2003)
T.W. Myers, D.H. Gelfand, Biochemistry 30, 7661–7666 (1991)
P.J. Obeid, T.K. Christopoulos, H.J. Crabtree, C.J. Backhouse, Anal. Chem. 75, 288–295 (2003)
N. Park, S. Kim, J.H. Hahn, Anal. Chem. 75, 6029–6033 (2003)
B. Pastorino, M. Bessaud, M. Grandadam, S. Murri, H.J. Tolou, C.N. Peyrefitte, J. Virol. Methods 124, 65–71 (2005)
M.G. Roper, C.J. Easley, J.P. Landers, Anal. Chem. 77, 3887–3893 (2005)
I. Schneegass, J.M. Kohler, J. Biotechnol. 82, 101–121 (2001)
E. Segal, N. Friedman, N. Kaminski, A. Regev, D. Koller, Nat. Genet. 37(Suppl), S38–S45 (2005)
S.S. Shevkoplyas, T. Yoshida, L.L. Munn, M.W. Bitensky, Anal. Chem. 77, 933–937 (2005)
M.D. To, S.J. Done, M. Redston, I.L. Andrulis, Am. J. Pathol. 153, 47–51 (1998)
P.L. Urban, D.M. Goodall, N.C. Bruce, Biotechnol. Adv. 24, 42–57 (2006)
E.J. Walsh, C. King, D. Ciobanu, R. Grimes, A. Gonzalez (ed.), Biomed. Microdev. 8, 59–64 (2006)
M.L. Wong, J.F. Medrano, Biotechniques 39, 75–85 (2005)
Acknowledgements
We thank Paddy O’Regan for device manufacture, Angela Morris and Dr. Fiona Gilchrist for help with cell culture and Dr.Eric Dalton for help with computerized thermal control of the device. DC and AG were supported by the Marie Curie Research Program ToK FP6 (MTKD-CT-2004-509790).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gonzalez, A., Ciobanu, D., Sayers, M. et al. Gene transcript amplification from cell lysates in continuous-flow microfluidic devices. Biomed Microdevices 9, 729–736 (2007). https://doi.org/10.1007/s10544-007-9083-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10544-007-9083-1