Abstract
For decades Darwinian processes were framed in the form of the Lewontin conditions: reproduction, variation and differential reproductive success were taken to be sufficient and necessary. Since Buss (The evolution of individuality, Princeton University Press, Princeton, 1987) and the work of Maynard Smith and Szathmary (The major transitions in evolution, Oxford University Press, Oxford, 1995) biologists were eager to explain the major transitions from individuals to groups forming new individuals subject to Darwinian mechanisms themselves. Explanations that seek to explain the emergence of a new level of selection, however, cannot employ properties that would already have to exist on that level for selection to take place. Recently, Hammerschmidt et al. (Nature 515:75–79, 2014) provided a ‘bottom-up’ experiment corroborating much of the theoretical work Paul Rainey has done since 2003 on how cheats can play an important role in the emergence of new Darwinian individuals on a multicellular level. The aims of this paper are twofold. First, I argue for a conceptual shift in perspective from seeing cheats as (1) a ‘problem’ that needs to be solved for multi-cellularity to evolve to (2) the very ‘key’ for the evolution of multicellularity. Secondly, I illustrate the consequences of this shift for both theoretical and experimental work, arguing for a more prominent role of ecology and the multi-level selection framework within the debate then they currently occupy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
When it comes to theorizing about evolution, much time and effort have been spent on developing a formalism capturing Darwinian mechanisms. Some of the elements that are employed in evolutionary explanations, however, are themselves products of Darwinian processes. Godfrey-Smith (2009) uses this as a justification to abstract the evolutionary mechanism even further, in order to deal with the marginal cases posing problems to theoretical biologists and philosophers. As Darwin explained, as long as there is reproduction, variation and differential reproductive success,Footnote 1 there will be natural selection. This, however, means that selection can occur on different levels of the biological hierarchy. Darwin (1871), who himself saw altruism as a problem for his theory, anticipated as much, suggesting that group selection may not only explain eusocial behaviour of insects, but also self-sacrificial behaviour within human tribes. With the publication of The Selfish Gene by Dawkins (1976), however, it became increasingly unpopular among biologists to cite group selection as the explanatory force behind a particular phenotype.
What followed was a turn not unlike the increasing dominance of methodological individualism in the social sciences during the same period.Footnote 2 Rather vague explanatory concepts such as the good of the speciesFootnote 3 were replaced with rigorous mathematical models such as those developed by evolutionary game theorists. In fact, evolutionary game theory was imported from economics by Maynard Smith and Price (1973), who saw game theory as an appropriate tool to formally explore George Price’s idea for conflict resolution. Maynard Smith’s and Price’s explicit goal was to provide a mechanistic explanation of conflict resolution in terms of individuals without the need to endorse group selection, something that seemed to them at odds with the idea of Darwinian selection acting within populations. Nevertheless, how evolution transitions from one level to the next has been a major research program on its own since Maynard Smith and Szathmary (1995) published their seminal work The Major Transitions in Evolution on transitions such as the evolution of sex, eukaryotic cells, and multicellularity. They were heavily influenced by Buss (1987) who wrote a pioneering work on the evolution of individuality specific to multicellular organisms. Despite being one of the strongest advocates for individualism in biology, making way for a mathematization of the discipline, Maynard Smith recognized the possibility of group selection being the driving force among the major transitions.
Although there were numerous biologists working on modelling the transition between multiple levels of selection, one of the most important ones being Michod (1996, 1999, 2005), Roze and Michod (2001), Michod and Nedelcu (2003), it arguably took the combined efforts of biologists and philosophers of science, (see Sober and Wilson 1998; Okasha 2006; Godfrey-Smith 2009), for multi-level selection to become an accepted explanatory strategy again, serving as an exemplary for the importance and impact of philosophy of biology.Footnote 4 Since then biologists have been much more eager to propose not only new models (see Traulsen and Nowak 2006), but also to conduct experiments in order to arrive at explanations for the evolution of multicellularity.Footnote 5 One of them is Paul Rainey, an experimental biologist, who explicitly uses multi-level selection to further scientific understanding of the transitions from singular cells to multicellular organisms, i.e. the major transition having occurred most often. Rainey’s research (Rainey and Rainey 2003; Rainey 2007; Rainey and Kerr 2010; Libby and Rainey 2013; Rainey and De Monte 2014) led to multiple experiments one of which (see Hammerschmidt et al. 2014) is the focus of this paper and as I am arguing has enormous potential to shed new light on this major transition and change the theoretical debates regarding the emergence of individuality.
In their paper “Life cycles, fitness decoupling and the evolution of multicellularity” Hammerschmidt et al. (2014) conclude the results of their experimental findings, trying to corroborate the research program of Paul Rainey. While working in the lab, Rainey became interested in the more philosophical problems of biology and started to think about possible mechanisms by which a transition towards multicellularity might occur. Traditionally cooperation has been the focus of such theorizing. However, Rainey argues that cooperation is not sufficient and has perhaps been overemphasized. When cooperation between the individuals in a group is ensured, there is no further selective pressure for biological integration and the emergence of a new level of selection. Even when cooperation persists for a number of generations among a group of cells, there will always remain the danger of cheaters arising, i.e. cells not contributing to the ‘good of the group’ but reaping the benefits thereof. Further emphasis, he argues, needs to be put on both a mechanism of group reproduction and a mechanism to minimize the adverse effects of cheats, explaining not only the emergence but also stability of cooperation among cells (2007, p. 616). Cheats here are defined as cells that reap the benefits of cooperation, without contributing to the public good themselves. This is the game-theoretic definition of cheats I intend to follow in the remainder of this paper. According to Rainey, the simultaneous occurrence of these three conditions is necessary for a major transition in individuality towards multicellularity to occur.
Rather than finding ways for optimal cheater suppressionFootnote 6 leading to multicellularity, Hammerschmidt et al. test the hypothesis of Rainey and Kerr (2010) that the appearance of cheats could play an important role for the evolution of multicellularity. Cheater suppression they argue could undermine the transition towards a new Darwinian individual at the level of groups. As I shall argue, Rainey provides a how-possibly explanation with considerable explanatory power. The Hammerschmidt et al. (2014) experiment is an attempt to provide further evidence that it could be the actual explanation behind the emergence of multicellularity.Footnote 7 This requires a conceptual shift in perspective from seeing cheats as (1) a ‘problem’ that needs to be solved for multi-cellularity to evolve to (2) the very ‘key’ for the evolution of multicellularity. As such cheats function as the saviours of the group organism: a new propagule enabling group reproduction and thereby taking the first steps towards a new unit of organization, a Darwinian individual at the group level of cells. This conceptual shift will have several important consequences. Let me now shortly give an overview of how this analysis will be structured before I proceed.
If philosophers can contribute anything to the debates in biology, it is at least conceptual clarity. In Sect. 2, which is the largest part of this paper, I, therefore, sketch the historical debate of multi-level selection and clarify the conceptual issues at stake. In Sect. 3, I finally analyse the experiment by Hammerschmidt et al. (2014). Here, I evaluate whether their results can corroborate Rainey’s thesis on the evolution of multicellularity espoused in multiple papers since 2003. In Sect. 4, I sum up the results and assess their significance for future research on the major transitions.
Multicellularity, fitness decoupling, and the levels of selection
One of the major problems in evolutionary biology remains the study of earliest cooperative groups and their transition towards multicellular organisms. As one of the major transitions, the evolution of multicellularity provides one of the most fruitful research questions in biology to date. The basic problem is this: if the evolution of multicellularity involves the emergence of a higher level of selection then how is it that selection at the level of individual cells does not undermine any cooperation among cells? Okasha (2006) gives credit to Michod (1999) for stressing that:
“[M]ulticelled organisms did not come from nowhere, and a complete evolutionary theory must surely try to explain how they evolved, rather than just taking their existence for granted. So levels of selection other than that of the individual organism must have existed in the past, whether or not they still operate today.” (p. 17)
As creationists might put it; “Explaining the evolution of certain traits is all well and fine, but if your theory cannot explain the origin of life [insert: major transitions] why should I accept it?” Certainly, the theory of evolution should still be widely accepted as a fact if its intended domain were limited to explaining particular evolutionary processes on levels of selection already persisting, i.e. evolutionary change over generations. But, evolutionary theorists are well-justified in aiming to achieve more. However, models already assuming a level in the ‘hierarchy of life’, the evolution of which we want to explain must conceptually prove insufficient for the purpose of explaining the evolution and persistence of new Darwinian individuals on a higher level. After all, one needs to explain the emergence of a mechanism of group reproduction, without such a mechanism already being present. As Libby and Rainey (2013) point out: “[v]ariation, heritability and reproduction are derived properties and their emergence at the group level requires an evolutionary explanation” (p. 2).
Building on the work of Buss (1987), Michod (1996, 1999) and Maynard Smith and Szathmary (1995) on the emergence of new units of selection, Paul Rainey was dissatisfied with debates on cooperation and cheater suppression dominating research on the major transitions. This led him, an experimental biologist, to engage in theorizing on the evolution of individuality, seeking contact with philosophers such as Kim Sterelny and Peter-Godfrey Smith. The result is a series of intra-disciplinary papers, ranging from philosophy of biology and theoretical biology to experimental biology, proposing a re-evaluation of the role cheats play in evolution (2003, 2007, 2010, 2013, 2014).
The major theme in Rainey’s work is a criticism of Michod (1996, 1999) and contemporary research on the evolution of multicellularity, for putting too much emphasis on cooperation and the suppression of cheats. Rainey argues that mere cooperation among cells, though a necessary condition, is simply not sufficient for a transition in Darwinian individuality and hence has been overemphasized. After all, there is more to the evolution of multicellularity than ‘solving’ an n-players prisoner’s dilemma or more accurately a public goods game. A group of cooperating individuals is still distinct from a group of cells constituting a new higher-level individual. As previously indicated, Rainey puts emphasis on at least two further conditions, beyond mere cooperation, which need to be instantiated for the emergence of individuality on the level of the multicellular organism: a mechanism of group reproduction and a mechanism to minimize the adverse effects of cheats, i.e. cancer control (2007, p. 616). The more or less simultaneous occurrence of these three conditions, Rainey suggests, is necessary for a major transition in individuality towards multicellularity to occur. I argue that without a mechanism of group reproduction it does not make sense to speak of a group organism at a higher level. Nevertheless, the importance of reproduction for natural selection has been treated at length (see Godfrey-Smith 2009; Libby and Rainey 2013). However, in order to grasp the importance of these two mechanisms and the role of multi-level selection in Rainey’s fertile work it shall prove useful to take a look at the group selection debate from which multi-level selection theory (MLS) was born, a debate that in fact continues to this day.
Once the idea that cancer is natural selection acting on the level of cells in a multicellular organism became widespread through conceptual arguments, largely driven by evolutionary game theory models, dispute has arisen over the degree to which selection at a lower level undermines [the conceptually possible] group selection acting at the higher level (see Frank et al. 1995, 2003; Axelrod et al. 2006). This is captured in the ongoing debate on the best explanatory account for the emergence and persistence of altruism and cooperation. Despite the now strong opposition to group selection acting on the level of multiple organisms, the evolution of multicellularity aims to explain selection starting to act on the level of multiple cells, which is essentially group selection lowered by one level. Multicellular organisms are groups of cells with fitness being best attributed to the whole organism rather than the individual cells. However, selection does not stop to act on the level of individual cells. Sometimes cells stop contributing to the good of the organism; they ‘go rogue’ and become a tumour, potentially undermining the group organism. Humans are often taken as the paradigmatic example for a Darwinian individual, but as Godfrey-Smith (2009) emphasized, Darwinian properties come in degrees. Individuality is on a continuum, and the move on this continuum, from selection acting on the level of cells to selection acting on the level of groups, is according to Rainey and Kerr (2010) best described as fitness decoupling, i.e. fitness being prescribed not to the individual cells but independently to the multicellular organism, an idea first introduced by Michod (1996, 1999), see also Roze and Michod (2001). Given that Darwinian individuality always comes in degrees, after all, cancer is hardly avoidable; it might be misleading to speak of selection acting on this or that level. A more appropriate way of interpreting fitness decoupling is to recognize a shift in causal strength between selection acting on the lower and higher level. It is here that multi-level selection becomes relevant when selection is acting on multiple levels.
How could multicellularity evolve and be sustained without group selection acting on the level of groups of cells when selection is acting on the level of individual cells? To cope with the problem of cheats, i.e. cancer in multicellular organisms, the opposition to group selection was in need of clarification. Otherwise, selection at the level of cells would have been an imminent threat potentially eliminating the property of individuality from multicellular organisms. These groups of cells, i.e. multicellular organisms, would lose the property of being distinguishable and taking part, independently of the fitness of their constitutive members, under natural selection. Clearly, many biologists argued that there is a difference between a multicellular organism and a herd of such individuals being subjected to natural selection. But with cancer, and Darwinian mechanisms acting on the levels of singular cells of multicellular organisms, the orthodox opposition to group selection was nevertheless, in need of re-evaluating their arguments.
Unlike group selection, selection acting on the level of the organism, was never in serious doubt. Reproduction on the level of groups of organisms was viewed as either marginal or completely reducible to individual reproduction. For a transition in Darwinian individuality from groups of co-operators to a genuinely new individual at a higher level, however, a mechanism of group reproduction becomes necessary. One of the most important additions made to biology was to bring clarity into the group-selection debate, by clarifying what group selection actually means. Thanks to Damuth and Heisler (1988), much conceptual confusion between multi-level selection and group selection was able to be resolved. They introduced the concept of multi-level selection [1], let us abbreviate with MLS1, and multi-level selection [2], MLS2 respectively, and a set of mathematical tools in order to partition the fitness effects of group and individual properties. Here, it is best to quote them directly:
“In multilevel selection [1]: | In multilevel selection [2]: |
(1) “Group selection” refers to the effects of group membership on individual fitness | (1) “Group selection” refers to change in the frequencies of different kinds of groups |
(2) Fitnesses are properties of individuals | (2) Fitnesses are properties of groups |
(3) Characters are values attributed to individuals (including both individual and contextual characters—see below) | (3) Characters are values attributed to groups (including both aggregate and global characters—see below) |
(4) Populations consist of individuals, organized into groups | (4) Populations consist of groups, composed of individuals |
(5) Explicit inferences can be made only about the changing proportions of different kinds of individuals in the whole population (the metapopulation) | (5) Explicit inferences can be made only about the changing proportions of different kinds of groups in the population” (p. 410) |
Though they omit discussion of heritability in groups, they grant that a complete account of multi-level selection would need to include such specifications. Rainey’s proposal as we shall see can be interpreted as a hypothesis capable of filling this gap. When we look at group selection taking place from an MLS1 point of view, mere cooperation between individuals would be sufficient—after all, group fitness is here simply defined as the average fitness of the group’s constituent cells.Footnote 8 Groups reaping the benefits of cooperation will do better than groups who fail to do so. However, from the MLS2 point of view, selection acts on genuinely new distinct units. Groups themselves are here Darwinian individuals themselves, taking part in Darwinian processes without having their fitness reducible to the fitness of their constituent members. Nevertheless, akin to matryoshka dolls lower level entities constitute the higher level entity with selection acting on both levels. This ambiguity lies at the heart of much of the dispute in the group selection debate and needs to be thoroughly understood in order to understand the major transitions (see Okasha 2006).
While Sober and Wilson (1998) defend group selection as a form of MLS1, Maynard Smith (1998) attacked their trait group model for not presenting a genuine case of group selection. In terms of Damuth and Heislers MLS account, Maynard Smith’s criticism concerns trait group model failing to satisfy the criteria of MLS2. Both sides of the debate were partly right. As already noted, philosophers should be able to ease conceptual confusions and make further theoretical work much more fruitful. Okasha’s (2006) Evolution and The Levels of Selection elegantly showed that much of the debate was ill-informed and could have been resolved if only there had been more clarity of what group selection is actually supposed to mean. As group selection has become an almost derogatory term for models, some proponents of group selection such as Wilson and Sober were led to use the term multi-level selection instead. In order to avoid confusion, Okasha (2006) proposes to refer to the group fitness at stake in group selection of the MLS1 type as fitness1 and group fitness of the MLS2 type as fitness2, a suggestion I follow here. Rainey’s critique of cooperation being insufficient, is merely him pointing out that researchers focusing on the evolution of individuality have focused too much on group selection at the MLS1 level, rather than the necessary transition to MLS2, where group fitness is not merely the mean fitness of the constituent cells. Both processes are at work when a transition in Darwinian individuality occurs. As Damuth and Heisler have illustrated, mere cooperation between group members is insufficient to transition from MLS1 to MLS2. Though game theory has been very successful in analysing MLS1 it might be insufficient for the analysis of fitness decoupling. This is not to say that an attempt to model Rainey’s hypothesis should not be undertaken, but that the orthodox tools for doing so may not be sufficient. Van Gestel and Tarnita (2017) criticize previous research into Darwinian individuality for mostly taking a “top-down” approach, i.e. looking at paradigm cases of Darwinian individuality and identifying a number of properties that are supposedly necessary for such a qualification, as being insufficient for understanding the major transitions. Instead they argue for a “bottom-up” approach in both theory and practice, which is nicely captured by the Hammerschmidt et al. (2014) experiment analysed in this paper.Footnote 9
When it comes to selection acting on multicellular organisms, Godfrey-Smith (2009) suggests a mechanistic account for Darwinian processes coming in degrees (Fig. 1). The graph above depicts a very useful illustration for theorizing about “bottom-up” approaches. His account posits three conditions that most groups of organisms do not satisfy for Darwinian mechanisms to fully act upon them: First, a bottleneck during which a propagule marks the beginning of a new life cycle. Second, a germ line being specialized for the reproduction of the collective. And third, the overall integration of the individuals forming a new individual rather than just a group. A single cell propagule marks the beginning of a new life cycle through a bottleneck and helps to easily demarcate individuals and their parent–offspring relationships (De Monte and Rainey 2014). But this criterion is not only useful for observing the variation from parent to offspring; it is important precisely because the size of the bottleneck determines the causal strength natural selection can have. When biologists talk of multiple herds of deer and their swiftness, swiftness is usually thought of as an adaptation at the individual level and not the group (see Okasha 2006). However, when herds of deer go through very small bottlenecks, a case for group selection and collective fitness2 can be made much more easily, not only minimizing variance within the group but also accentuating variance between groups, further strengthening group selection. Hence, the causal strength of Darwinian processes comes once again in degrees. The same applies to the germ/soma distinction. The more cells are soma; the more cells work for the survival and replication of the germ-line or rather the group. Hence, the higher the share of soma cells, the stronger the case to be made for these groups being individual units themselves. Overall integration entails the division of labour beyond the soma/germ-line distinction and “the maintenance of a boundary between a collective and what is outside it” (Godfrey-Smith 2009, p. 21). While his and Kerr’s (2010) proposal of a single cheating cell as a new propagule marking the beginning of a new life cycle already weakly satisfies the criteria of Godfrey-Smith, overall integration is still lacking. Their proposal of a mechanism for group reproduction and a mechanism to minimize the adverse effects of cheats seems even less restrictive than Godfrey-Smith’s account. That is because Rainey wants to capture the very beginning of MLS2 selection before the Lewontin conditions can even be applied to the higher level. Godfrey-Smith himself (2009) stresses the role of marginal cases that though not satisfying all criteria may nevertheless have natural selection acting upon them. And this is exactly what is needed to explain the major transitions in individuality.
This brings us back to the debate between Sober and Wilson (1998) and Maynard Smith (1998). Maynard Smith espoused Hamilton’s (1964) idea of kin selection and inclusive fitness as sufficient for explaining the ‘strange’ occurrences of altruism in groups of animals. If an organism acts altruistically and engages in costly behaviour towards its own fitness, but benefits others, the trait will spread as long as those others share the trait and the benefit to them outweighs the cost of the altruism. Without assuming asexual reproduction, we only need a sufficiently high degree of relatedness. Sober and Wilson, however, arguing for group selection argued that kin-selection is just one form of group selection generating a correlation between the trait in question and the probability of interaction between individuals possessing the same trait, as opposed to random encounters. Maynard Smith (1998) did not accept their version of ‘group selection’ as MLS2 and rightly so. Other possibilities would include the signalling of being a reciprocal altruist combined with higher interaction rates among altruists. Whether we accept it as ‘real’ group selection is more of a linguistic question rather than anything else. Unfortunately, considerable time and effort have been spent on this debate. Kin-selection and inclusive fitness, however, do not provide a genuine case of selection acting on groups that are individuals themselves.Footnote 10
De Monte and Rainey (2014) try to relax the Lewontin conditions in order to generate an even more minimalized account of evolution which can explain the evolution of individuality at the level of multicellular organisms. They take the argument of Sober and Wilson (1998) for the trait group model to show that such statistical bias can establish a genealogy already sufficient for Darwinian processes. Godfrey-Smith (2009), however, brought a halt to this line of argument for higher-level group selection by pointing out that such a correlation may happen in a mere two-dimensional spatial structure. The replacement of groups by neighbourhoods cannot satisfy the MLS2 criteria as acting on a higher level (pp. 117–118). Not only is it hard to distinguish groups in such a setting with borders being fluid but as Godfrey-Smith points out “there are as many groups as individuals” (2009, p. 118). Damuth and Heisler (1988) recognized as much, with MLS1 being “relevant to situations in which individual fitnesses are context-dependent, but there are no groups as such and individuals are more or less continuously distributed” (p. 411). What matters for cooperation among cells is then not as Sober and Wilson (1998) argued group selection, but rather any kind of correlated interaction (2009, p. 118). This correlation can be instantiated via appropriate ecological conditions that as we shall see play an important role in the formation of a group in the experimental work on Rainey’s hypothesis. Both kin selection and trait groups models are then subsumed under MLS1 and serve as appropriate tools for the evolution of cooperation, but as Rainey points out they are insufficient for explaining the transitions to MLS2.
Okasha (2006) formulates the question motivating the levels of selection debate as: “[W]hen is a character-fitness covariance indicative of direct selection at the level in question, and when is it a by-product of direct selection at another hierarchical level?” (p. 79). This is in effect a question of causal responsibility and not just a potential view one may or may not take for a specific research question in biology. For the pluralists, there is no such distinction possible, while the monist realist using the multi-level selection approach has a powerful tool at hand to settle questions of causal efficacy, while still having the benefit of different viewpoints. When it comes to explaining the major transitions in evolution, this realist picture may precisely be what is needed. Kin selection and inclusive fitness are not satisfactory for this purpose as they follow an MLS1 approach. There is just one major conceptual question left, and that is how we move from MLS1 and mere cooperation to MLS2 and having a genuine individual. Here the work of Michod (1996, 1999), Roze and Michod (2001) on a mechanism he termed fitness decoupling will be illuminating.
As already mentioned, fitness decoupling is the transition from MLS1 to MLS2. The fitness of the ‘group’ becomes independent of the average fitness of the constituent cells. It becomes a Darwinian individual, with multiple groups forming Darwinian populations themselves (Godfrey-Smith 2009). For Michod and Nedelcu (2003) conflict mediation between the higher level, i.e. the group, and the lower level, i.e. the cells, is the key to evolutionary transitions in individuality. The activities of the cells making up the organism affect the fitness2 of the whole rather than their own fitness.Footnote 11 But we need to remind ourselves that this is always a matter of degree, with cancer being a prevailing possibility. Michod proposes multiple ways in which such conflict mediation may occur. For the purpose of the Hammerschmidt et al. experiment, I will focus on the germ/soma distinction and the role of cheaters. Michod and Nedelcu (2003) argues for two crucial features a germ/soma distinction can accomplish: (1) reduced adverse effects of reproduction on the survival capacities of an organism and (2) “further increases in the survival component of group fitness” (p. 4). This division of labour opens the possibility of two distinct optimization problems to be solved, reproduction and survival. Once the ball gets rolling, the multicellular organism can become ever more specialized and integrated. This is what Rainey and Kerr (2010) argue for: cheats should be seen as a proto germ-line capable of saving the higher level entity rather than being their doom, a first step towards individuality.
Before the fitness at the multicellular organism-level is decoupled, adaptations at the group level are to be causally explained as a by-product of singular cell-level selection, i.e. MLS1 (Michod 1999). With a multi-level selection approach to the major transitions, a potentially large set of theoretical options that have been hitherto unexplored, such as a positive role for cheats, can come under serious scientific investigation. It is here that the environment will play an important role in enabling cheats to be interpreted differently. Paul Rainey refers to this special role of the environment making up for the lack of Darwinian properties as ecological scaffolding.Footnote 12 This new task of explaining the major transitions with seemingly non-Darwinian means then seems a far more adventurous enterprise than previous explanatory tasks of how already existing populations change over time with well-established methods readily available. Naturally, experiments are required in order to narrow this set of potential explanations down to the causal mechanisms behind fitness decoupling. As Godfrey-Smith (2009) stresses, Darwinian mechanisms can also act on marginal cases, and this is what the following experiment of Hammerschmidt et al. demonstrates: the fitness decoupling from MLS1 to MLS2, hence the evolution of individuality.
Evidence from experimental evolution
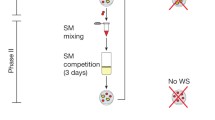
Rather than finding ways for optimal cheater suppression leading to multicellularity, Hammerschmidt et al. (2014) test the hypothesis of Rainey and Kerr (2010) that cheater suppression may, in fact, hinder the evolution of multicellularity with cheats being necessary for the generation of a propagule. This enables a form of group reproduction which simultaneously solves the problem of ensuring the right amount of cooperation,Footnote 13 providing both a mechanism of group reproduction and a mechanism to minimize the adverse effects of cheats. As illustrated by debates on group selection, cheats have the potential to undermine the selection at higher levels, reaping the benefit of belonging to a collective without contributing themselves. This led to multiple researchers, in particular, Michod (1996, 1999), to argue for the necessity of cheater suppression for multicellular organisms to emerge. Contrary to what common sense might suggest, cheater suppression is not the only mechanism able to minimize the adverse effects of cheats for the group. As highlighted by Rainey and Kerr (2010), cheats may be necessary for the generation of a propagule and the enabling of group reproduction. In order to provide evidence for this hypothesis Hammerschmidt et al. conducted the following experiment (Fig. 2)Footnote 14:
They propagated the bacterium Pseudomonas fluorescens in spatially structured, undisturbed microcosms, i.e. beakers filled with a nutritious broth able to support their propagation as depicted above. The original genotype, with which the experiment starts has a ‘smooth’ phenotype expression with individual cells freely floating within the broth. Due to short life cycles of the individual cells (they reproduce every hour) and a high mutation rate, some of them quickly mutated from the ancestral ‘smooth’ genotype (SM) to ‘wrinkly’ spreaders (WS), each labelled by the distinct appearance of their respective colony morphologies. These WS mutants overproduced acetylated cellulose i.e. a costly cell–cell glue preventing their daughter cells [also producing this cell glue] from detaching themselves after cell division.Footnote 15 Normally such groups of cells have a lower fitness, not only because the production of such a polymer is expensive in terms of fitness but also because these associations take up space close to each other and thereby become competitors for resources.Footnote 16
In the setting depicted above (Fig. 3), however, the phenotype [here: blue] spreads because these cell-glue associations are able to reap the benefits of access to oxygen by staying at the interface between the air and the liquid growth medium, something the ancestral type was not able to achieve. As Rainey et al. (2017) points out, these ‘mats’ are not buoyant and hence require a physical structure to attach themselves and reap the benefits of this ecological niche, further illustrating the importance of the environment in the evolution of multicellularity, i.e. here the beaker. The mat gets bigger and bigger taking over the whole surface, a niche previously unexplored, but selection on the individual cell level will make mutant-cheats eventually prosper leading to cheating cells [here: green] reaping the benefits of cooperation without contributing to the glue necessary for the mat to survive. This spells doom for the WS mats, as the public good, i.e. access to oxygen cannot be sustained. While these mutants can stay at the top of the mat and reproduce, the mat gets heavier while simultanously lowering the integrity of the mat itself. When it collapses all WS cells in the mat are driven to extinction due to their lower fitness. This is a classical tragedy of the commons.Footnote 17
This conflict between cooperating WS cells and cheats, they argue, “could fuel evolution” (2014, p. 75) by giving selection something to act on. As the WS mats lack means of collective reproduction, they are similar to soma an evolutionary dead end. While cheats do spell doom for the mat, they might also be its saviour by becoming a germ-line of sorts leading to a primitive life cycle switching between WS mats and mutant propagules. This may then enable mats to participate marginally as a unit of selection. As Godfrey-Smith (2009) explained; a life cycle and a single-cell propagule will be able to drive Darwinian mechanisms, which is what this experiment tries to capture. Let me now turn to a detailed analysis of the experiment responsible for the creation of this paper (see Fig. 4).Footnote 18
Initially, Hammerschmidt et al. only tested one regime consisting of 120 of these spatially undisturbed microcosms. Every microcosm starts with a WS genotype [here: yellow]. In phase I (see the illustration above) each of the 120 microcosms is left alone for 6 days after which the cells are harvested and screened for SM colonies. If no WS mat has formed or no SM are found the line ends, i.e. it goes extinct. If no WS mat is present, the multicellular body has not remained intact, while failure to produce SM cells [here: red] stands for a failure to produce propagules. In phase 2, which only lasted for 3 days the microcosms continued solely with the newly formed SM cells. If they failed to transition back to WS cells within this time period the line was also taken to be extinct. By analogy imagine the failure of a propagule to develop further. After 6 generations all lines were extinct. However, Hammerschmidt et al. argue that even this persistence is sufficiently surprising and could bring these lineages under developmental control. They suggest that if successful lineages were allowed to take over the microcosms left by those lineages going extinct, it could “allow the possibility for life-cycle-enhancing mutations, which are beneficial over the longer time scale of the life cycle, [able] to outrun life-cycle-disrupting mutations” (p. 76) (Fig. 5).Footnote 19,Footnote 20
In response to this theoretical possibility, Hammerschmidt et al. devised two regimes (depicted above), consisting of one in which cheats were embraced and one in which they were purged, each of which consisting again of 120 spatially structured microcosms, however, now divided into 15 populations of 8 lines. Both regimes went through a single-cell bottleneck, one SM cell and one WS cell respectively. For the cheat-embracing regime nothing changes from the initial experiment above except for the fact that the microcosms left by extinct lineages are taken over by successful ones within the same population. Contrary to what one might expect, the cheat-embracing regime lead to life-cycle-enhancing mutations outweighing the effects of life-cycle-disrupting mutations while the cheat-purging regime did not show such an evolutionary response as they don’t have a (proto) germ-line for Darwinian mechanism to act upon. What may seem abstruse at first is perhaps a major step towards explaining the transition towards multicellularity.
Rather than cheater suppression, cheat embracing simultaneously solves the problem of group reproduction and minimization of the adverse effects of cheats. While cheats are detrimental to the fitness of the mat organism, without them there would be no group reproduction. As cheats are needed, the mat organism has to bring cheats under developmental control in order to move from MLS1 to MLS2. The Hammerschmidt et al. experiment (2014) is then a classic example for survival-reproduction tradeoffs at the level of mats. While cheats lead to an earlier death of the mat organism, seeing them as germ-cells makes it obvious that more cheats allow for a higher fecundity i.e. higher number of propagules. However, sometimes higher fecundity may be more valuable than higher survival, which is why the focus in the literature on mere cooperation among constituent cells has led to Rainey’s new hypothesis: given the right ecological conditions, cheats can justifiably be treated as both the doom and saviour of the multicellular organism. Their results suggest that fitness decoupling is taking place in the cheat-embracing regime, just as Rainey’s theoretical work on the major transitions predicted. While the collective fitness of the mat in the cheat-purging regime is simply the average fitness of its constituent cells i.e. MLS1, the fitness of the collective in the cheat-embracing regime is taking its first marginal steps towards MLS2 (Okasha 2006) or, as Godfrey-Smith (2009) might put it, the mats take their first steps towards becoming Darwinian populations themselves.
Interesting work on the relation between group formation via adhesion, and the evolution of group reproduction has been conducted using game theoretic models. Garcia et al. (2015) argue that cellular adhesion is a key ingredient to the evolution of multicellularity, as exemplified by the propensity of glue production by WS cells. Furthermore, Tarnita et al. (2010) have highlighted the importance of staying together, with daughter cells staying attached, thereby enabling further integration and the germline coming under development control. In addition, Pichugin et al. (2017) show, such a multicellular strategy of fragmentation via single-cell propagules may often be superior to individual dispersion, making Rainey’s hypothesis of cheats generating new life cycles all the more credible. In the cheat-embracing regime the fitness of the mat lineage increases, while the fitness of individual cells is actually decreased. The longer life-cycle of the mat organism stands to conquer the selection among individual cells, making them ‘work for’ the benefit of the group. Though the group reproduction via switching cheats is only a marginal case for reproduction, it gives evolution something to act on potentially bringing the propagule under developmental control. After all, a small number of mutations in the original SM genotype lead to the emergence of group properties and a proto life cycle.
Due to the artificial nature of their laboratory setting, some may doubt the validity of their results. To combat such claims and make the experiment more realistic Rainey et al. (2017) suggest the example of a pond with randomly located reeds as both a thought experiment and a potential field experiment. As mat-forming cells are non-buoyant they require something to attach themselves, otherwise not being able to reap the benefits of access to oxygen. The reeds provide one such environmental option at which mats can form cooperative group organisms with higher fitness than individual ancestral types (hence ecological scaffolding). Mats here just like in the experimental setting inevitably fall to their doom, however, if a new germ-line by detaching cheats can be created, then a new life cycle at the same reed or another one can begin, even allowing for the spread of these mat organisms. One important further environmental condition is the sufficient distance between reeds to prevent overlapping growth of mats and allow for variation, and hence selection between mat-organisms. The two timescales between cell and mat reproduction generate two levels of selection, even though the Darwinian properties at the mat level are in the beginning only exogenously provided. That, however, is exactly what is needed in order to take the first steps towards the evolution of a new Darwinian individual, with these properties becoming endogenized through the “evolution of a developmentally determined life cycle” (p. 105), generating eco-evolutionary feedback loops. The importance of ecology has here not only a biotic dimension (interaction among organisms, i.e. WS and SM types), but also an abiotic dimension (interaction with the environment) which makes the modelling of these processes much more difficult. As Rainey et al. (2017) put it: “ecology is everything: the structure of the environment permits realisation of Darwinian properties at the collective level even in the absence of these properties being endogenously determined” (pp. 104–105). The Hammerschmidt et al. experiment does exactly that: suggest possibilities for Darwinian properties to become endogenized. One such way was elegantly illustrated in the cheat-embracing regime. Cheats evolved a simple genetic switch to secure the switching from soma-mats to germ-line propagules marking the beginning of a new, though unstable, life cycle and the emergence of a proto multicellular organism. The idea here is simple, most multicellular organisms reproduce via single-sell propagules, something the origin of which is to be explained. Rainey’s hypothesis has the potential of doing so.
Citing a study of eusociality (Nowak et al. 2010), Tarnita (2017) draws an analogy between Rainey’s proposal and eusocial colonies of ants. Both the mat organism and the colony have a differential life span for both the ‘group organism’ and the individual members. Except for the queen in these ant colonies, however, all the individuals in the colony are sterile making them ‘slaves’ to the group or to as the name suggests: the queen. The new-born queens Tarnita points out can just as much be interpreted as a cheat, ‘detaching’ themselves once they have matured in order to mate forming a new group organism. Naturally, some will respond that the new-born queens are, in fact, not selfish but rather one integral part of the whole organism responsible for reproduction. Just as detaching cheats have to switch back to glue production in order to form a new mat, the queens need to form a new colony. It may not be reasonable at this point, to treat these propagules as cheats, as they serve an important role in the life cycle of the group organism. There is an inherent ambiguity between the shorter life cycle of individuals and the longer life cycle of the group. However, this realization only came to be, by questioning the very notion of cheats being always bad simply in virtue of their evaluative name. Traditional game-theoretic analyses, excluding such roles for cheats, must necessarily cloud the generation of new hypotheses granting them such an important role in the evolution of multicellularity. Tarnita (2017) argues that it is precisely because the ecology and development of eusocial ant colonies are much better understood that we understand their transition to a new Darwinian individual. She further suggests that we stop using anthropomorphising terms such as “cooperation, cheating and selfishness […] biasing our study of eco-evolutionary processes” (p. 22). Though I deem it unlikely that these traditional terms can be cast-off overnight, (if ever,) biologists need to be aware of their negative impact on the generation of new hypotheses. Let me now summarize the discussion and analyse the impact of Rainey’s work for both conceptual issues and future experimental work in biology.
Further directions and conclusion
The primary aim of this paper was to illustrate that when it comes to the evolution of multicellular organisms and the emergence of new Darwinian individuals on a higher level, too much focus has been spent on cooperation alone. To understand the evolution of multicellularity a conceptual shift in perspective is required. Cheats are no longer just (1) a ‘problem’ that needs to be solved for multi-cellularity to evolve but rather (2) as argued in this paper the very ‘key’ for the evolution of multicellularity.
As Rainey noted, a mechanism of group reproduction and a mechanism to minimize the adverse effects of cheats are necessary conditions that might together with cooperation be sufficient for the emergence of new Darwinian individuals. As I showed, this neatly matches onto the 3D Space for Darwinian individuality Godfrey-Smith proposed: a bottleneck during which a propagule marks the beginning of a new life cycle, a germ line being specialized for reproduction of the collective and overall integration of the individuals forming a new individual rather than just a group. All of these come in degrees, and the Hammerschmidt et al. paper beautifully shows how the first steps can be taken towards Darwinian individuality.
By seeing cheats as the first single-cell propagules marking the beginning of a new life cycle through a bottleneck, the problem of ensuring group reproduction and minimization of adverse cheats can be solved simultaneously. This introduces the germ/soma distinction and makes further refinements possible. Before such a propagule can come under developmental control, a random mutation is necessary, allowing for a switching mechanism from a SM to a WS phenotype. The cheat-purging regime did not manage to decouple mat fitness from the fitness of the cells, while contrary to intuition the cheat-embracing regime captures the first step towards Darwinian individuality, by showcasing that mat fitness increases independently from the decreasing cell fitness. The cells start to “work for” the organism. Their carefully conducted experiment corroborates much of Rainey’s theoretical work over the last decade. Their MLS approach provides strong evidence that cheats and conflict, contrary to intuition are what drives the evolution of multicellularity, a sign that intentional notions such as cheats and altruism can cloud the generation of fruitful hypotheses. It is unlikely that these notions will be replaced, perhaps due to an inherent desire to explain apparently purposive behaviour in biology or the import of game theory into biology, from economics where intentions and goals play a central role (see Rosenberg 2009).
However, more emphasis should be put on the fact that cooperation is a technical term, intended to be value-free, merely referring to phenotypes that increase the fitness of others, though associated with a cost to oneself. The importance of the environment can here not be understated, for it calls into question the very labels of cheats and co-operators, and highlights the importance of eco-evolutionary feedback.Footnote 21 Because of these, more than just curious, results Tarnita (2017) even calls for a revision of the orthodox cooperation framework leading to misleading interpretations when the environment is omitted from the analysis at hand. Rainey’s work may then provide a strong case for the extended evolutionary synthesis highlighting the importance of the environment and the evolution of development. By largely omitting developmental and environmental factors playing a role in the evolution of multicellularity, cooperation and the suppression of cheats has been deemed to be more important than it actually is. However, as Libby and Rainey (2013) point out the ecological conditions of multicellular organisms having evolved millions of years ago are “essentially unknowable” (p. 1). Therefore, one should not be too quick in dismissing previous and extensive work in evolutionary game theory on the emergence and persistence of cooperation. The best we might be able to arrive at are robust how-possibly explanations of how multicellularity could have arisen. Furthermore, through further advances in experimental and synthetic biology, we may even be able to have multicellular organisms emerge in the lab. The analysis of environmental factors will help to narrow down the set of how-possibly explanations and increase our confidence that proposed mechanisms such as the cheats as propagules hypothesis by Rainey may have been at work millions of years ago.
Further work may focus on the impact of anthropomorphising language on research. More attention should also be brought onto the inherent differences between eukaryotes and prokaryotes, as the most interesting cases of evolutionary transitions towards multicellularity tend to involve eukaryotes, even though the Hammerschmidt experiment provided evidence that even the prokaryote Pseudomonas fluorescens can take the first steps towards Darwinian individuality. Additional marginal cases for Darwinian individuality in the case of prokaryotes worthy of studying are social bacteria such as Myxococcus xanthus that form fruiting bodies with reproductive division of labour (see Crespi 2001) and filaments of cyanobacteria with cellular division of labour (see Rossetti et al. 2010). Furthermore, one could shed light on the development process involving proto-germ lines coming under development control. Recent literature on size-specialization feedback-loops may provide further insight into division of labour (see Bourke 2011 and Birch 2017) or as Godfrey-Smith (2009) calls it: integration, something that is still lacking in our account in the Hammerschmidt et al. experiment, only marking the very beginning of a major transition in individuality. Also as Rainey and Kerr note at the end of their (2010) paper, Heininger (2002) has done interesting work on the ageing mechanism being a “deprivation syndrome driven by the tension between the soma and germ line” (p. 879). Once it is established that cheats serve as the origin of the germ/soma distinction, many research questions will be illuminated. At the very least, this opens up a new theoretical option that has been largely unexplored. Let me conclude this paper with a quote from the last page of Samir Okasha’s Evolution and the Levels of Selection:
“The study of evolutionary transitions is still in its infancy, with much empirical work remaining to be done, so it is difficult to say whether the foregoing analysis will prove satisfactory in all respects. But whatever future developments in the field look like, it is likely that multi-level selection will remain crucial for theorizing about evolutionary transitions” (2006, p. 240).
And in this Okasha’s philosophical work on multi-level selection proved to be quite successful in generating a progressive research program. The experimental work of Hammerschmidt, Rose, Kerr and Rainey on the question of how evolution transitions from singular cells to multicellular organisms, is a prime example for the strength of the multi-level selection approach. In a mere decade, it seems the study of evolutionary transitions underwent significant progress, offering a more positive view for the role of cheats and a recognition of the importance of ecology. Much work, however, remains to be done, with prospects for the work of biologists and philosophers of biology blending together appearing more than promising.
Change history
24 December 2019
The author would like to notify the readers about the following.
24 December 2019
The author would like to notify the readers about the following.
24 December 2019
The author would like to notify the readers about the following.
Notes
These three conditions are also known as Lewontin conditions published in The Units of Selection (1970).
Damuth and Heisler (1988) explicitly reference work in the social sciences as inspiration for their multi-level selection account (pp. 416–417).
See Bouchard and Huneman (2013) for a recent collection of essays on the emergence of individuality on a higher level.
See Travisano and Velicer (2004) for strategies of microbial cheater control.
In at least some of the multiple and independently emerged multicellular organisms.
Godfrey-Smith (2009, p. 95).
Godfrey-Smith (2009) prefers to think of this process as “de-Darwinization” at the lower level (p. 101).
See Rainey et al. (2017).
As the experiment will illustrate, cheats are necessary for the creation of a propagule and the beginning of a new life-cycle. If no cheats arise, the group is effectively an evolutionary dead-end with nothing for natural selection to act on. ‘Perfect cooperation’ can then be a hindrance to the evolution of multicellularity. Unlike linguistic convention suggests, cheats are to be embraced, rather than avoided at all costs.
Rainey and Kerr (2010, p. 875).
See Spiers et al. (2003).
Rainey and Kerr (2010, p. 876).
See Hardin (1968).
Hammerschmidt et al. (2014, p. 76).
Hammerschmidt et al. (2014, p. 76).
See Moreno-Fenoll et al. (2017) for an exploration of such eco-evolutionary population dynamics.
References
Axelrod R, Axelrod DE, Pienta KJ (2006) Evolution of cooperation among tumor cells. Proc Natl Acad Sci USA 103(36):13474–13479
Birch J (2017) The philosophy of social evolution. Oxford University Press, Oxford
Bouchard F, Huneman P (eds) (2013) From groups to individuals: evolution and emerging individuality, Vienna series in theoretical biology. MIT Press, Cambridge
Bourke AFG (2011) In: Harvey PH, May RM, Godfray CH, Dunne JA (eds) Principles of social evolution. Oxford series in ecology and evolution. Oxford University Press, Oxford, pp xii + 267
Buss LW (1987) The evolution of individuality. Princeton University Press, Princeton
Crespi BJ (2001) The evolution of social behaviour in microorganisms. Trends Ecol Evol 16(4):178–183
Damuth J, Heisler IL (1988) Alternative formulations of multilevel selection. Biol Philos 3:407–430
Darwin C (1871) The descent of man. The Heritage Press, New York, p 1972
Dawkins R (1976) The selfish gene. Oxford University Press, Oxford
Dawkins R (1984) Replicators and vehicles. In: Brandon RN, Burian R (eds) Genes, organisms, populations: controversies over the units of selection. Cambridge University Press, Cambridge, pp 161–180
De Monte S, Rainey PB (2014) Nascent multicellular life and the emergence of individuality. J Biosci 39:237–248
Frank SA (1995) Mutual policing and repression of competition in the evolution of cooperative groups. Nature 377:520–522
Frank SA, Iwasa Y, Nowak MA (2003) Patterns of cell division and the risk of cancer. Genetics 163:1527–1532
Garcia T, Doulcier G, De Monte S (2015) The evolution of adhesiveness as a social adaptation. eLife 4:e08595. https://doi.org/10.7554/elife.08595
Godfrey-Smith P (2009) Darwinian populations and natural selection. Oxford University Press, Oxford
Hamilton WD (1964) The genetical evolution of social behaviour. I. J Theor Biol 7(1):1–16
Hammerschmidt K, Rose CJ, Kerr B, Rainey PB (2014) Life cycles, fitness decoupling and the evolution of multicellularity. Nature 515:75–79
Hardin G (1968) The tragedy of the commons. Science 162:1243–1248
Heininger K (2002) Aging is a deprivation syndrome driven by a germ–soma conflict. Aging Res Rev 1:481–536
Leigh EG (1977) How does selection reconcile individual advantage with the good of the group? Proc Natl Acad Sci USA 74:4542–4546
Lewontin RC (1970) The units of selection. Annu Rev Ecol Syst 1:1–18
Libby E, Rainey PB (2013) A conceptual framework for the evolutionary origins of multicellularity. Phys Biol 10:035001
Lorenz KZ (1966) On aggression. Methuen, London
Maynard Smith J (1964) Group selection and kin selection. Nature 201:1145–1147
Maynard Smith J (1976) Group selection. Q Rev Biol 51:277–283
Maynard Smith J (1998) The origin of altruism. Nature 393:639–640
Maynard Smith J, Price G (1973) The logic of animal conflicts. Nature 246:15–18
Maynard Smith J, Szathmary E (1995) The major transitions in evolution. Oxford University Press, Oxford
Michod RE (1996) Cooperation and conflict in the evolution of individuality. 2. Conflict mediation. Proc R Soc Lond B 263:813–822
Michod RE (1999) Darwinian dynamics: evolutionary transitions in fitness and individuality. Princeton University Press, Princeton
Michod RE (2005) On the transfer of fitness from the cell to the multicellular organism. Biol Philos 20:967
Michod RE, Nedelcu AM (2003) On the reorganization of fitness during evolutionary transitions in individuality. Integr Comput Biol 43:64–73
Michor F, Frank SA, May RM, Iwasa Y, Nowak MA (2003) Somatic selection for and against cancer. J Theor Biol 225:377–382
Moreno-Fenoll C, Cavaliere M, Martínez-García E, Poyatos JF (2017) Eco-evolutionary feedbacks can rescue cooperation in microbial populations. Sci Rep 7:42561
Nowak MA, Tarnita CE, Wilson EO (2010) The evolution of eusociality. Nature 466:1057–1062
Nunney L (1985) Group selection, altruism, and structured-deme models. Am Nat 126:212–230
Nunney L (1999) Lineage selection and the evolution of multistage carcinogenesis. Proc R Soc Lond B 266:493–498
Okasha S (2006) Evolution and the levels of selection. Oxford University Press, Oxford
Pichugin Y, Peña J, Rainey PB, Traulsen A (2017) Fragmentation modes and the evolution of life cycles. PLoS Comput Biol 13(11):e1005860
Rainey PB (2007) Unity from conflict. Nature 446(7136):616
Rainey PB, De Monte S (2014) Resolving conflicts during the evolutionary transition from cells to multicellular life. Ann Rev Ecol Syst 45:599–620
Rainey PB, Kerr B (2010) Cheats as first propagules: a new hypothesis for the evolution of individuality during the transition from single cells to multicellularity. BioEssays 32:872–888
Rainey PB, Rainey K (2003) Evolution of cooperation and conflict in experimental bacterial populations. Nature 425:72–74
Rainey PB, Remigi P, Farr AD, Lind PA (2017) Darwin was right: Where now for experimental evolution? Curr Opin Genet Dev 47:102–109
Ratcliff WC, Denison RF, Borrello M, Travisano M (2012) Experimental evolution of multicellularity. Proc Natl Acad Sci USA 2012(109):1595–1600
Ratcliff WC, Herron MD, Howell K, Pentz JT, Rosenzweig F, Travisano M (2013) Experimental evolution of an alternating uni- and multicellular life cycle in Chlamydomonas reinhardtii. Nat Commun 2013(4):2742
Rosenberg A (2009) If economics is a science, what kind of science is it? In: Kincaid H, Ross D (eds) The oxford handbook of philosophy of economics. Oxford University Press, Oxford, pp 55–67
Rossetti R, Schirrmeister BE, Bernasconi MV, Bagheri HC (2010) The evolutionary path to terminal differentiation and division of labor in cyanobacteria. J Theor Biol 262(1):23–34
Roze D, Michod RE (2001) Mutation, multilevel selection, and the evolution of propagule size during the origin of multicellularity. Am Nat 158(6):638–654
Sober E, Wilson DS (1998) Unto others: the evolution and psychology of unselfish behavior. Harvard University Press, Cambridge
Spiers AJ, Bohannon J, Gehrig SM, Rainey PB (2003) Biofilm formation at the air–liquid interface by the Pseudomonas fluorescens SBW25 wrinkly spreader requires an acetylated form of cellulose. Mol Microbiol 50:15–27
Tarnita CE (2017) The ecology and evolution of social behavior in microbes. J Exp Biol 220:18–24
Tarnita CE, Nowak MA, Wilson EO (2010) The evolution of eusociality. Nature 466(7310):1057–1062
Traulsen A, Nowak MA (2006) Evolution of cooperation by multilevel selection. Proc Natl Acad Sci 103(29):10952–10955
Travisano M, Velicer G (2004) Strategies of microbial cheater control. Trends Microbiol 12:72–78
van Gestel J, Tarnita CE (2017) On the origin of biological construction, with a focus on multicellularity. Proc Natl Acad Sci 114(42):11018–11026
Van Veelen M (2009) Group selection, kin selection, altruism and cooperation: when inclusive fitness is right and when it can be wrong. J Theor Biol 259:589–600
van Veelen M, García J, Sabelis MW, Egas M (2012) Group selection and inclusive fitness are not equivalent; the price equation vs. models and statistics. J Theor Biol 299:64–80
Wynne-Edwards VC (1962) Animal dispersion in relation to social behavior. Oliver & Boyd, London
Acknowledgements
First of all, I would like to thank Arne Traulsen and Chaitanya S. Gokhale who enabled me to conduct this research project at the Max Planck Institute for Evolutionary Biology, the latter of which integrated me into his research group and supported me throughout the creation of this paper. Furthermore, I thank Samir Okasha, Paul B. Rainey, Katrin Hammerschmidt, Yuriy Pichugin, Hanna Schenk, Maria Bargués Ribera, Vandana R. Venkateswaran, Jacqueline Wallis, Alison K. McConwell, Shaun Stanley, and two anonymous referees for comments on earlier versions of this paper. Also, I would like to thank audiences at the Evolution Evolving conference in Cambridge, the First International PhilInBioMed Meeting at the University of Bordeaux, the Generalized Theory of Evolution Conference at the University of Düsseldorf, the 7th Philosophy of Biology Graduate Conference at the University of Calgary, and the TINT Centre for Philosophy of Social Science in Helsinki, among which special thanks goes to Alkistis Elliott-Graves. Sincere apologies to anyone I forgot to mention.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Veit, W. Evolution of multicellularity: cheating done right. Biol Philos 34, 34 (2019). https://doi.org/10.1007/s10539-019-9688-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10539-019-9688-9