Abstract
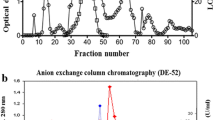
Lipoxygenase (LOX) from opium poppy (Papaver somniferum L.) chloroplasts was isolated and 126.1-fold purified to electrophoretic homogeneity by combination of ion-exchange chromatography on HA-Ultragel column and affinity chromatography on a linoleyl-aminopropyl agarose column. The relative molecular mass of the LOX determined by SDS-PAGE was 92 kDa. Kinetic properties of purified LOX were determined in spectrophotometric assay by using of linoleic acid (KM = 1.78 mM and Vmax = 11.4 μmol mg−1 min−1) and linolenic acid (KM = 1.27 mM and Vmax = 10.2 μmol mg−1 min−1). The optimum pH was 6.0 for both linoleic and linolenic acid dioxygenation catalyzed by LOX. HPLC analysis of the products revealed a dual positional specificity of linoleic acid dioxygenation at pH 6.0 with ratio of 9- and 13-hydroperoxide products being about 1:1. The activity of purified LOX was stimulated by Mg2+ and Ca2+.
Similar content being viewed by others

Abbreviations
- GrB:
-
grinding buffer
- HOD:
-
hydroxyoctadecadienoic acid
- HPOD:
-
hydroperoxyoctadecadienoic acid
- KPB:
-
potassium phosphate buffer
- LA:
-
linoleic acid
- LAPA:
-
linoleyl-aminopropyl agarose
- LeA:
-
linolenic acid
- LOD:
-
limit of detection
- LOQ:
-
limit of quantification
- LOX:
-
lipoxygenase
- PMSF:
-
phenylmethylsulfonyl fluoride
- RP HPLC:
-
reversed-phase highperformance liquid chromatography
- SP HPLC:
-
straight-phase high-performance liquid chromatography
- TX-114 :
-
Triton X-114
References
Allen, J.C., Eriksson, C., Galpin, J.R.: Affinity chromatography of lipoxygenases. — Eur. J. Biochem. 73: 171–177, 1977.
Andersen, M.D., Møller, B.L.: Double Triton X-114 phase partitioning for the purification of plant cytochromes P450 and removal of green pigments. — Protein Expression Purif. 13: 366–372, 1998.
Blum, H., Beier, H., Gross, H.J.: Improved silver staining of plant proteins, RNA and DNA in polyacrylamide gels. — Electrophoresis 8: 93–99, 1987.
Bowsher, C.G., Ferrie, B.J.M., Ghosh, S., Todd, G.J., Thompson, J.E., Rothstein, S.J.: Purification and partial characterization of a membrane associated lipoxygenase in tomato fruit. — Plant Physiol. 100: 1802–1807, 1992.
Daglia, M., Aceti, C., Giorgetti, S., Papetti, A., Gazzani, G.: Purification and characterization of soluble Cichorium intybus var. silvestre lipoxygenase. — J. Agr. Food Chem. 53: 6448–6454, 2005.
Feussner, I., Wasternack, C.: The lipoxygenase pathway. — Annu. Rev. Plant Biol. 53: 275–297, 2002.
Fornarolli, S., Petrussa, E., Braidot, E., Vianello, A., Macri, F.: Purification of a plasma membrane-bound lipoxygenase from soybean cotyledons. — Plant Sci. 145: 1–10, 1999.
Garavito, R.M., Ferguson-Miller, S.: Detergents as tools in membrane biochemistry. — J. biol. Chem. 276: 32403–32406, 2001.
Garbe, L.-A., De Almeida, R.B., Nagel, R., Wackerbauer, K., Tressl, R.: Dual positional and stereospecificity of lipoxygenase isoenzymes from germinating barley (Green Malt): biotransformation of free and esterified linoleic acid. — J. Agri. Food. Chem. 54: 946–955, 2006.
Hornung, E., Kunze, S., Liavonchanka, A., Zimmermann, G., Kühn, D., Fritsche, K., Renz, A., Kühn, H., Feussner, I.: Identification of an amino acid determinant of pH regiospecificity in a seed lipoxygenase of Momordica charantia. — Phytochemistry 69: 2774–2780, 2008.
Hughes, R.K., West, S.I., Hornostaj, A.R., Lawson, D.M., Fairhurst, S.A., Sanchez, R.O., Hough, P., Robinson, B.H., Casey, R.: Probing a novel potato lipoxygenase with dual positional specificity reveals primary determinants of substrate binding and requirements for a surface hydrophobic loop and has implications for the role of lipoxygenases in tubers. — Biochem. J. 15: 345–355, 2001.
Kim, E.S., Kim, H., Park, R.D., Lee, Y., Han, O.: Dual positional specificity of wound-responsive lipoxygenase from maize seedlings. — J. Plant. Physiol. 159: 1263–1265, 2002.
Kuo, J.M., Hwang, A., Yeh, D.B., Pan, M.H., Tsai, M.L., Pan, B.S.: Lipoxygenase from banana leaf. Purification and characterization of an enzyme that catalyzes linoleic acid oxygenation at the 9-position. — J. Agr. Food Chem. 54: 3151–3156, 2006.
Laemmli, U.K.: Cleavage of structural proteins during the assembly of the head of bacteriophage T4. — Nature 227: 680–685, 1970.
Lang, I., Feussner, I.: Oxylipin formation in Nostoc punctiforme (PCC73102). — Phytochemistry 68: 1120–1127, 2007.
López-Nicolas, J.M., Pérez-Gilabert, M., García-Carmona, F.: Eggplant lipoxygenase (Solanum melongena): product characterization and effect of physicochemical properties of linoleic acid on the enzymatic activity. — J. Agr. Food Chem. 49: 433–438, 2001.
Lorenzi, V., Maury, J., Casanova, J., Berti, L.: Purification, product characterization and kinetic properties of lipoxygenase from olive fruit (Olea europaea L.). — Plant Physiol. Biochem. 44: 450–454, 2006.
Palmieri-Thiers, C., Canaan, S., Brunini, V., Lorenzi, V., Tomi, F., Desseyn, J.L., Garscha, U., Oliw, E.H., Berti, L., Maury, J.: A lipoxygenase with dual positional specificity is expressed in olives (Olea europaea L.) during ripening. — Biochim. biophys. Acta 1791: 339–346, 2009.
Pérez-Gilabert, M, López-Nicolás, J.M., García-Carmona, F.: Purification of a novel lipoxygenase from eggplant (Solanum melongena) chloroplasts. — Physiol. Plant. 111: 276–282, 2001.
Porra, R.J.: The chequered history of the development and use of simultaneous equations for the accurate determination of chlorophylls a and b. — Photosynth. Res. 73: 149–156, 2002.
Portis, A.R., Heldt, H.W.: Light-dependent changes of the Mg2+ concentration in the stroma in relation to the Mg2+ dependency of CO2 fixation in intact chloroplasts. — Biochim. biophys. Acta 449: 434–436, 1976.
Purkait, M.K., Das Gupta, S., De, S.: Determination of thermodynamic parameters for the cloud point extraction of different dyes using TX-100 and TX-114. — Desalination 244: 130–138, 2009.
Rangel, M., Machado, O.L.T., Da Cunha, M., Jacinto, T.: Accumulation of chloroplast-targeted lipoxygenase in passion fruit leaves in response to methyl jasmonate. — Phytochemistry 60: 619–625, 2002.
Wang, R., Shen, W., Liu, L., Jiang, L., Liu, Y., Su, N., Wan, J.: A novel lipoxygenase gene from developing rice seeds confers dual position specificity and responds to wounding and insect attack. — Plant mol. Biol. 66: 401–414, 2008.
Acknowledgements
This work was supported by grant of Slovak ministry of education VEGA 1/4294/07 and VEGA 1/0089/10.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vanko, M., Rauová, D., Bezáková, L. et al. Biochemical properties of lipoxygenase from opium poppy chloroplasts. Biol Plant 56, 105–110 (2012). https://doi.org/10.1007/s10535-012-0023-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10535-012-0023-4