Abstract
Cadmium is a heavy metal that is widespread in the environment and has been described as a metalloestrogen and a cardiovascular risk factor. Experimental studies conducted in male animals have shown that cadmium exposure induces vascular dysfunction, which could lead to vasculopathies caused by this metal. However, it is necessary to investigate the vascular effects of cadmium in female rats to understand its potential sex-dependent impact on the cardiovascular system. While its effects on male rats have been studied, cadmium may act differently in females due to its potential as a metalloestrogen. In vitro studies conducted in a controlled environment allow for a direct assessment of cadmium's impact on vascular function, and the use of female rats ensures that sex-dependent effects are evaluated. Therefore, the aim of this study was to investigate the in vitro effects of Cadmium Chloride (CdCl2, 5 µM) exposure on vascular reactivity in the isolated aorta of female Wistar rats. Exposure to CdCl2 damaged the architecture of the vascular endothelium. CdCl2 incubation increased the production and release of O2•−, reduced the participation of potassium (K+) channels, and increased the participation of the angiotensin II pathway in response to phenylephrine. Moreover, estrogen receptors alpha (Erα) modulated vascular reactivity to phenylephrine in the presence of cadmium, supporting the hypothesis that cadmium could act as a metalloestrogen. Our results demonstrated that in vitro cadmium exposure induces damage to endothelial architecture and an increase in oxidative stress in the isolated aorta of female rats, which could precipitate vasculopathies.
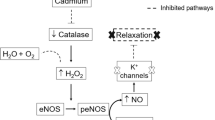
Graphical Abstract

Graphical Abstract. Own source from Canva and Servier Medical Art servers








Similar content being viewed by others
References
Abu-Hayyeh S, Sian M, Jones KG et al (2001) Cadmium accumulation in aortas of smokers. Arterioscler Thromb Vasc Biol 21:863–867. https://doi.org/10.1161/01.ATV.21.5.863
Almenara CCP, Broseghini-Filho GB, Vescovi MVA et al (2013) Chronic Cadmium Treatment Promotes Oxidative Stress and Endothelial Damage in Isolated Rat Aorta. PLoS ONE 8:1–8. https://doi.org/10.1371/journal.pone.0068418
Almenara CCP, Oliveira TF, Padilha AS (2020) The Role of Antioxidants in the Prevention of Cadmium-Induced Endothelial Dysfunction. Curr Pharm Des 26:3667–3675. https://doi.org/10.2174/1381612826666200415172338
Almenara CCP, de Oliveira TF, da Silva DCF et al (2022) Cessation Restores Blood Pressure Levels and Endothelial Function Affected by Cadmium Exposure on Rats. Biol Trace Elem Res. https://doi.org/10.1007/s12011-022-03312-5
Alvarez Y, Briones AM, Hernanz R, Pérez-Girón JV, Alonso MJ, Salaices M (2008Mar) Role of NADPH oxidase and iNOS in vasoconstrictor responses of vessels from hypertensive and normotensive rats. Br J Pharmacol. 153(5):926–35. https://doi.org/10.1038/sj.bjp.0707575
Angeli JK, Cruz Pereira C, De Oliveira T et al (2013) Cadmium exposure induces vascular injury due to endothelial oxidative stress: The role of local angiotensin II and COX-2. Free Radic Biol Med 65:838–848. https://doi.org/10.1016/j.freeradbiomed.2013.08.167
Aquino NB, Sevigny MB, Sabangan J, Louie MC (2012) The role of cadmium and nickel in estrogen receptor signaling and breast cancer: Metalloestrogens or not? J Environ Sci Heal - Part C Environ Carcinog Ecotoxicol Rev 30:189–224. https://doi.org/10.1080/10590501.2012.705159
Aryan L, Younessi D, Zargari M et al (2020) The role of estrogen receptors in cardiovascular disease. Int J Mol Sci 21:1–26. https://doi.org/10.3390/ijms21124314
ATSDR (2012) Public Health Statement for Cadmium. Public Heal Statement 1–10. Available in: https://www.atsdr.cdc.gov/ToxProfiles/tp5-c1-b.pdf
Babatunde Ishola A, Michael Okechukwu I, Gregory Ashimedua U et al (2017) Serum Level of Lead, Zinc, Cadmium, Copper and Chromium Among Occupationally Exposed Automotive Woerkers In Benin City. Int J Environ Pollut Res 5:70–79
Blazka ME, Shaikh ZA (1991) Differences in cadmium and mercury uptakes by hepatocytes: Role of calcium channels. Toxicol Appl Pharmacol 110:355–363. https://doi.org/10.1016/S0041-008X(05)80018-3
Brama M, Gnessi L, Basciani S et al (2007) Cadmium induces mitogenic signaling in breast cancer cell by an ERα-dependent mechanism. Mol Cell Endocrinol 264:102–108. https://doi.org/10.1016/j.mce.2006.10.013
Byrne C, Divekar SD, Storchan GB et al (2009) Cadmium - A metallohormone? Toxicol Appl Pharmacol 238:266–271
Byrne C, Divekar SD, Storchan GB et al (2013) Metals and breast cancer. J Mammary Gland Biol Neoplasia 18:63–73. https://doi.org/10.1007/s10911-013-9273-9
Casalino E, Calzaretti G, Sblano C, Landriscina C (2002) Molecular inhibitory mechanisms of antioxidant enzymes in rat liver and kidney by cadmium. Toxicology 179:37–50. https://doi.org/10.1016/S0300-483X(02)00245-7
Cordeiro ER, Filetti FM, Simões MR, Vassallo DV (2019) Mercury induces nuclear estrogen receptors to act as vasoconstrictors promoting endothelial denudation via the PI3K/Akt signaling pathway. Toxicol Appl Pharmacol. https://doi.org/10.1016/j.taap.2019.114710
de Oliveira TF, Rossi EM, da Costa CS et al (2022) Sex-dependent vascular effects of cadmium sub-chronic exposure on rats. Biometals. https://doi.org/10.1007/s10534-022-00470-w
Dikalov SI, Nazarewicz RR, Bikineyeva A, Hilenski L, Lassègue B, Griendling KK, Harrison DG, Dikalova AE (2014) Nox2-induced production of mitochondrial superoxide in angiotensin II-mediated endothelial oxidative stress and hypertension. Antioxid Redox Signal. 20(2):281–94. https://doi.org/10.1089/ars.2012.4918
Do EK, Lee MS, Paek D (2008) Cadmium in blood and hypertension. Sci Total Environ 407:147–153. https://doi.org/10.1016/j.scitotenv.2008.08.037
Donpunha W, Kukongviriyapan U, Sompamit K et al (2011) Protective effect of ascorbic acid on cadmium-induced hypertension and vascular dysfunction in mice. Biometals 24:105–115. https://doi.org/10.1007/s10534-010-9379-0
Faroon O, Ashizawa A, Wright S et al (2012) Toxicological Profile for Cadmium. Chem Phys Inf. https://doi.org/10.1016/s1090-3798(09)70033-9
Fechner P, Damdimopoulou P, Gauglitz G (2011) Biosensors paving the way to understanding the interaction between cadmium and the estrogen receptor alpha. PLoS ONE. https://doi.org/10.1371/journal.pone.0023048
Friberg L, Kjellstrom TEC (1992) Environmental health criteria for cadmium - Environmental aspects. Environ Heal Criteria 1–156. Available in: https://apps.who.int/iris/bitstream/handle/10665/38998/9241571349-eng.pdf?sequence=1&isAllowed=y
Friques AGF, Arpini CM, Kalil IC et al (2015) Chronic administration of the probiotic kefir improves the endothelial function in spontaneously hypertensive rats. J Transl Med 13:1–16. https://doi.org/10.1186/s12967-015-0759-7
Garcia-Morales P, Saceda M, Kenney N et al (1994) Effect of cadmium on estrogen receptor levels and estrogen-induced responses in human breast cancer cells. J Biol Chem 269:16896–16901. https://doi.org/10.1016/s0021-9258(19)89474-7
Gaudet HM, Christensen E, Conn B et al (2018) Methylmercury promotes breast cancer cell proliferation. Toxicol Reports 5:579–584. https://doi.org/10.1016/j.toxrep.2018.05.002
Gökalp O, Özdem S, Dönmez S et al (2009) Impairment of endothelium-dependent vasorelaxation in cadmium-hypertensive rats. Toxicol Ind Health 25:447–453. https://doi.org/10.1177/0748233709106822
Hayashi K, Maeda S, Iemitsu M, Otsuki T, Sugawara J, Tanabe T, Miyauchi T, Kuno S, Ajisaka R, Matsuda M (2007) Sex differences in the relationship between estrogen receptor alpha gene polymorphisms and arterial stiffness in older humans. Am J Hypertens. 20(6):650–6. https://doi.org/10.1016/j.amjhyper.2007.01.007
Hengstler JG, Bolm-Audorff U, Faldum A et al (2003) Occupational exposure to heavy metals: DNA damage induction and DNA repair inhibition prove co-exposures to cadmium, cobalt and lead as more dangerous than hitherto expected. Carcinogenesis 24:63–73. https://doi.org/10.1093/carcin/24.1.63
Henkler F, Brinkmann J, Luch A (2010) The role of oxidative stress in carcinogenesis induced by metals and xenobiotics. Cancers (basel) 2:376–396. https://doi.org/10.3390/cancers2020376
Heumüller S, Wind S, Barbosa-Sicard E et al (2008) Apocynin is not an inhibitor of vascular NADPH oxidases but an antioxidant. Hypertension 51:211–217. https://doi.org/10.1161/HYPERTENSIONAHA.107.100214
Jacobson KB, Turner JE (1980) The interaction of cadmium and certain other metal ions with proteins and nucleic acids. Toxicology 16:1–37. https://doi.org/10.1016/0300-483X(80)90107-9
Jamakala O, Rani UA (2015) Amelioration effect of zinc and iron supplementation on selected oxidative stress enzymes in liver and kidney of cadmium-treated male albino rat. Toxicol Int 22:1–9. https://doi.org/10.4103/0971-6580.172289
Johnson MD, Kenney N, Stoica A et al (2003) Cadmium mimics the in vivo effects of estrogen in the uterus and mammary gland. Nat Med 9:1081–1084. https://doi.org/10.1038/nm902
Kim KH, Young BD, Bender JR (2014) Endothelial estrogen receptor isoforms and cardiovascular disease. Mol Cell Endocrinol 389:65–70. https://doi.org/10.1016/j.mce.2014.02.001
Kukongviriyapan U, Pannangpetch P, Kukongviriyapan V et al (2014) Curcumin protects against cadmium-induced vascular dysfunction, hypertension and tissue cadmium accumulation in mice. Nutrients 6:1194–1208. https://doi.org/10.3390/nu6031194
Lobato NS, Filgueira FP, Akamine EH et al (2011) Obesity induced by neonatal treatment with monosodium glutamate impairs microvascular reactivity in adult rats: Role of NO and prostanoids. Nutr Metab Cardiovasc Dis 21:808–816. https://doi.org/10.1016/j.numecd.2010.02.006
Majumder S, Muley A, Kolluru GK et al (2008) Cadmium reduces nitric oxide production by impairing phosphorylation of endothelial nitric oxide synthase. Biochem Cell Biol 86:1–10. https://doi.org/10.1139/O07-146
Martínez-Campa C, Alonso-González C, Mediavilla MD et al (2006) Melatonin inhibits both ERα activation and breast cancer cell proliferation induced by a metalloestrogen, cadmium. J Pineal Res 40:291–296. https://doi.org/10.1111/j.1600-079X.2006.00315.x
Nagarajan S, Rajendran S, Saran U, Priya MK, Swaminathan A, Siamwala JH, Sinha S, Veeriah V, Sonar P, Jadhav V, Jaffar Ali BM, Chatterjee S (2013) Nitric oxide protects endothelium from cadmium mediated leakiness. Cell Biol Int. 37(5):495–506. https://doi.org/10.1002/cbin.10070
Nelson MT, Quayle JM (1995) channels in arterial smooth muscle I. Am J Physiol 268:C799–C822
Nguyen Dinh Cat A, Montezano AC, Burger D, Touyz RM (2013) Angiotensin II, NADPH oxidase, and redox signaling in the vasculature. Antioxidants Redox Signal 19:1110–1120. https://doi.org/10.1089/ars.2012.4641
Nigra AE, Ruiz-Hernandez A, Redon J et al (2016) Environmental Metals and Cardiovascular Disease in Adults: A Systematic Review beyond Lead and Cadmium Compliance with Ethics Guidelines Conflict of Interest HHS Public Access. Curr Env Heal Rep 3:416–433. https://doi.org/10.1007/s40572-016-0117-9.Environmental
Novella S, Laguna-Fernández A, Lázaro-Franco M et al (2013) Estradiol, acting through estrogen receptor alpha, restores dimethylarginine dimethylaminohydrolase activity and nitric oxide production in oxLDL-treated human arterial endothelial cells. Mol Cell Endocrinol 365:11–16. https://doi.org/10.1016/j.mce.2012.08.020
Nunes DO, Almenara CCP, Broseghini-Filho GB et al (2014) Flaxseed oil increases aortic reactivity to phenylephrine through reactive oxygen species and the cyclooxygenase-2 pathway in rats. Lipids Health Dis 13:1–2. https://doi.org/10.1186/1476-511X-13-107
Nwokocha CR, Baker A, Douglas D et al (2013) Apocynin ameliorates cadmium-induced hypertension through elevation of endothelium nitric oxide synthase. Cardiovasc Toxicol 13:357–363. https://doi.org/10.1007/s12012-013-9216-0
Oliveira TF, Batista PR, Leal MA et al (2019) Chronic Cadmium Exposure Accelerates the Development of Atherosclerosis and Induces Vascular Dysfunction in the Aorta of ApoE −/− Mice. Biol Trace Elem Res 187:163–171. https://doi.org/10.1007/s12011-018-1359-1
Orshal JM, Khalil RA (2004) Gender, sex hormones, and vascular tone. Am J Physiol Regul Integr Comp Physiol. 286(2):233–49. https://doi.org/10.1152/ajpregu.00338.2003
Pérez-Cremades D, Mompeón A, Gómez XV et al (2018) MiRNA as a new regulatory mechanism of estrogen vascular action. Int J Mol Sci. https://doi.org/10.3390/ijms19020473
Petrônio MS, Zeraik ML, Da Fonseca LM, Ximenes VF (2013) Apocynin: Chemical and biophysical properties of a NADPH oxidase inhibitor. Molecules 18:2821–2839. https://doi.org/10.3390/molecules18032821
Prozialeck WC, Edwards JR, Woods JM (2006) The vascular endothelium as a target of cadmium toxicity. Life Sci 79:1493–1506. https://doi.org/10.1016/j.lfs.2006.05.007
Sadeghi N, Oveisi MR, Jannat B et al (2014) The relationship between bone health and plasma zinc, copper lead and cadmium concentration in osteoporotic women. J Environ Heal Sci Eng 12:1–5. https://doi.org/10.1186/s40201-014-0125-3
Sangartit W, Kukongviriyapan U, Donpunha W et al (2014) Tetrahydrocurcumin protects against cadmium-induced hypertension, raised arterial stiffness and vascular remodeling in mice. PLoS ONE 9:1–21. https://doi.org/10.1371/journal.pone.0114908
Santamaria-Juarez C, Atonal-Flores F, Diaz A et al (2020) Aortic dysfunction by chronic cadmium exposure is linked to multiple metabolic risk factors that converge in anion superoxide production. Arch Physiol Biochem 128:748–756. https://doi.org/10.1080/13813455.2020.1726403
Satarug S (2018) Dietary cadmium intake and its effects on kidneys. Toxics 6:1–23. https://doi.org/10.3390/toxics6010015
Satarug S, Baker JR, Urbenjapol S et al (2003) A global perspective on cadmium pollution and toxicity in non-occupationally exposed population. Toxicol Lett 137:65–83. https://doi.org/10.1016/S0378-4274(02)00381-8
Schereider IRG, Vassallo DV, Simões MR (2021) Chronic mercury exposure induces oxidative stress in female rats by endothelial nitric oxide synthase uncoupling and cyclooxygenase-2 activation, without affecting oestrogen receptor function. Basic Clin Pharmacol Toxicol 129:470–485. https://doi.org/10.1111/bcpt.13655
Sherman TS, Chambliss KL, Gibson LL et al (2002) Estrogen acutely activates prostacyclin synthesis in ovine fetal pulmonary artery endothelium. Am J Respir Cell Mol Biol 26:610–616. https://doi.org/10.1165/ajrcmb.26.5.4528
Sobrino A, Oviedo PJ, Novella S et al (2010) Estradiol selectively stimulates endothelial prostacyclin production through estrogen receptor-α. J Mol Endocrinol 44:237–246. https://doi.org/10.1677/JME-09-0112
Sobrino A, Vallejo S, Novella S et al (2017) Mas receptor is involved in the estrogen-receptor induced nitric oxide-dependent vasorelaxation. Biochem Pharmacol 129:67–72. https://doi.org/10.1016/j.bcp.2017.01.012
Sompamit K, Kukongviriyapan U, Donpunha W et al (2010) Reversal of cadmium-induced vascular dysfunction and oxidative stress by meso-2,3-dimercaptosuccinic acid in mice. Toxicol Lett 198:77–82. https://doi.org/10.1016/j.toxlet.2010.04.007
Sripada K, Lager AM (2022) Interventions to reduce cadmium exposure in low- and middle-income countries during pregnancy and childhood: A systematic review. J Glob Health. https://doi.org/10.7189/jogh.12.04089
Stockand J, Sultan A, Molony D, Dubose T, Sansom S (1993) Interactions of cadmium and nickel with k channels of vascular smooth muscle. Toxicol Appl Pharm. 121(1):30–35. https://doi.org/10.1006/taap.1993.1125
Stoica A, Katzenellenbogen BS, Martin MB (2000) Activation of estrogen receptor-α by the heavy metal cadmium. Mol Endocrinol 14:545–553. https://doi.org/10.1210/me.14.4.545
Su EJ, Lin ZH, Zeine R et al (2009) Estrogen receptor-beta mediates cyclooxygenase-2 expression and vascular prostanoid levels in human placental villous endothelial cells. Am J Obstet Gynecol 200:427.e1-427.e8. https://doi.org/10.1016/j.ajog.2009.01.025
Tang X, Garcia M, Heinemann S et al (2004) Reactive oxygen species impair Slo1 BK channel function by altering cysteine-mediated calcium sensing. Nat Struct Mol Biol 11:171–178. https://doi.org/10.1038/nsmb725
Tellez-Plaza M, Navas-Acien A, Crainiceanu CM, Guallar E (2008) Cadmium exposure and hypertension in the 1999–2004 National Health and Nutrition Examination Survey (NHANES). Environ Health Perspect 116:51–56. https://doi.org/10.1289/ehp.10764
Thévenod F (2009) Cadmium and cellular signaling cascades: To be or not to be? Toxicol Appl Pharmacol 238:221–239. https://doi.org/10.1016/j.taap.2009.01.013
Tzotzes V, Tzilalis V, Giannakakis S et al (2007) Effects of acute and chronic cadmium administration on the vascular reactivity of rat aorta. Biometals 20:83–91. https://doi.org/10.1007/s10534-006-9017-z
Vassallo DV, Almenara CCP, Broseghini-Filho GB et al (2018) Preliminary Studies of Acute Cadmium Administration Effects on the Calcium-Activated Potassium (SKCa and BKCa) Channels and Na+/K+-ATPase Activity in Isolated Aortic Rings of Rats. Biol Trace Elem Res 183:325–334. https://doi.org/10.1007/s12011-017-1150-8
Vassallo DV, Wiggers GA, Padilha AS, Simões MR (2020) Endothelium: A Target for Harmful Actions of Metals. Curr Hypertens Rev 16:201–209. https://doi.org/10.2174/1573402115666190115153759
Yoopan N, Watcharasit P, Wongsawatkul O et al (2008) Attenuation of eNOS expression in cadmium-induced hypertensive rats. Toxicol Lett 176:157–161. https://doi.org/10.1016/j.toxlet.2007.11.002
Yuan P, Wu WH, Gao L et al (2013) Oestradiol ameliorates monocrotaline pulmonary hypertension via NO, prostacyclin and endothelin-1 pathways. Eur Respir J 41:1116–1125. https://doi.org/10.1183/09031936.00044112
Zafari AM, Ushio-fukai M, Akers M et al (1998) Angiotensin II – Induced Vascular Hypertrophy. Hypertension 32:488–495
Funding
This study was supported by grants from Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES—Financing code 001); Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq—4/2021) and Fundação de Amparo à Pesquisa do Espírito Santo (FAPES/CNPq—nº 25/2022; 80600115). The funders had no role in the study design, data collection, data analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
All authors whose names appear on the submission: (1) made substantial contributions to the conception or design of the work; or the acquisition, analysis, or interpretation of data; or the creation of new software used in the work; (2) drafted the work or revised it critically for important intellectual content; (3) approved the version to be published; and (4) agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
The research protocols performed in this study were in accordance with the guidelines recommended by the Brazilian College of Animal Experimentation (COBEA). The project was previously approved by the Ethics Committee on Experimentation and Use of Animals of the Federal University of Espírito Santo (UFES—CEUA 10/2020).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sepulchro Mulher, L.C.C., Simões, R.P., Rossi, K.A. et al. In vitro cadmium exposure induces structural damage and endothelial dysfunction in female rat aorta. Biometals 36, 1405–1420 (2023). https://doi.org/10.1007/s10534-023-00526-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-023-00526-5