Abstract
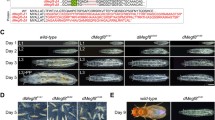
While the effects of systemic zinc ion deficiency and toxicity on animal health are well documented, the impacts of localized, tissue-specific disturbances in zinc homeostasis are less well understood. Previously we have identified zinc dyshomeostasis scenarios caused by the targeted manipulation of zinc transport genes in the Drosophila eye. Over expression of the uptake transporter dZIP42C.1 (dZIP1) combined with knockdown of the efflux transporter dZNT63C (dZNT1) causes a zinc toxicity phenotype, as does over expression of dZIP71B or dZNT86D. However, all three genotypes result in different morphologies, responses to dietary zinc, and genetic interactions with the remaining zinc transport genes, indicating that each causes a different redistribution of zinc within affected cells. dZNT86D eGFP over expression generates a completely different phenotype, interpreted as a Golgi zinc deficiency. Here we assess the effect of each of these transgenes when targeted to a range of Drosophila tissues. We find that dZIP71B is a particularly potent zinc uptake gene, causing early developmental lethality when targeted to multiple different tissue types. dZNT86D over expression (Golgi-only zinc toxicity) is less deleterious, but causes highly penetrant adult cuticle, sensory bristle and wing expansion defects. The dZIP42C.1 over expression, dZNT63C knockdown combination causes only moderate adult cuticle defects and sensitivity to dietary zinc when expressed in the midgut. The Golgi-only zinc deficiency caused by dZNT86D eGFP expression results in mild cuticle defects, highly penetrant wing expansion defects and developmental lethality when targeted to the central nervous system and, uniquely, the fat bodies.


Similar content being viewed by others
Abbreviations
- (e)GFP:
-
(enhanced) green fluorescent protein
- TPEN:
-
N,N,N′,N′-tetrakis (2-pyridylmethyl)-ethylenedidiamine
References
Adlard PA, Parncutt JM, Finkelstein DI, Bush AI (2010) Cognitive Loss in Zinc Transporter-3 Knock-Out Mice: a phenocopy for the synaptic and memory deficits of Alzheimer’s disease? J Neurosci 30:1631–1636. doi:10.1523/Jneurosci.5255-09.2010
Andrews GK, Wang H, Dey SK, Palmiter RD (2004) Mouse zinc transporter 1 gene provides an essential function during early embryonic development. Genesis 40:74–81
Binks T, Lye JC, Camakaris J, Burke R (2010) Tissue-specific interplay between copper uptake and efflux in Drosophila. J Biol Inorg Chem 15:621–628
Calleja M, Moreno E, Pelaz S, Morata G (1996) Visualization of gene expression in living adult Drosophila. Science 274:252–255
Dechen K, Richards CD, Lye JC, Hwang JE, Burke R (2015) Compartmentalized zinc deficiency and toxicities caused by ZnT and Zip gene over expression result in specific phenotypes in Drosophila. Int J Biochem Cell Biol 60C:23–33. doi:10.1016/j.biocel.2014.12.017
Dufner-Beattie J, Kuo YM, Gitschier J, Andrews GK (2004) The adaptive response to dietary zinc in mice involves the differential cellular localization and zinc regulation of the zinc transporters ZIP4 and ZIP5. J Biol Chem 279:49082–49090
Dufner-Beattie J, Weaver BP, Geiser J, Bilgen M, Larson M, Xu W, Andrews GK (2007) The mouse acrodermatitis enteropathica gene Slc39a4 (Zip4) is essential for early development and heterozygosity causes hypersensitivity to zinc deficiency. Hum Mol Genet 16:1391–1399
Fukada T et al (2008) The zinc transporter SLC39A13/ZIP13 is required for connective tissue development; its involvement in BMP/TGF-beta signaling pathways. PLoS One 3:e3642
Geiser J, Venken KJ, De Lisle RC, Andrews GK (2012) A mouse model of acrodermatitis enteropathica: loss of intestine zinc transporter ZIP4 (Slc39a4) disrupts the stem cell niche and intestine integrity. PLoS Genet 8:e1002766. doi:10.1371/journal.pgen.1002766
Geiser J, De Lisle RC, Andrews GK (2013) The zinc transporter Zip5 (Slc39a5) regulates intestinal zinc excretion and protects the pancreas against zinc toxicity. PLoS One 8:e82149. doi:10.1371/journal.pone.0082149
Gho M, Bellaiche Y, Schweisguth F (1999) Revisiting the Drosophila microchaete lineage: a novel intrinsically asymmetric cell division generates a glial cell. Development 126:3573–3584
Hwang JE, de Bruyne M, Warr CG, Burke R (2014) Copper overload and deficiency both adversely affect the central nervous system of Drosophila Metallomics 6:2223–2229. doi:10.1039/c4mt00140k
Inoue Y et al (2014) ZIP2 protein, a zinc transporter, is associated with keratinocyte differentiation. J Biol Chem 289:21451–21462. doi:10.1074/jbc.M114.560821
Lazareva AA, Roman G, Mattox W, Hardin PE, Dauwalder B (2007) A role for the adult fat body in Drosophila male courtship behavior PLoS Genet 3:e16. doi:10.1371/journal.pgen.0030016
Lin DM, Goodman CS (1994) Ectopic and increased expression of Fasciclin II alters motoneuron growth cone guidance. Neuron 13:507–523
Lye JC et al (2012) Systematic functional characterization of putative zinc transport genes and identification of zinc toxicosis phenotypes in Drosophila melanogaster. J Exp Biol 215:3254–3265. doi:10.1242/jeb.069260
Lye J, Richards C, Dechen K, Warr C, Burke R (2013a) In vivo zinc toxicity phenotypes provide a sensitized background that suggests zinc transport activities for most of the Drosophila Zip and ZnT genes. J Biol Inorg Chem JBIC 18:323–332. doi:10.1007/s00775-013-0976-6
Lye JC, Richards CD, Dechen K, Warr CG, Burke R (2013b) In vivo zinc toxicity phenotypes provide a sensitized background that suggests zinc transport activities for most of the Drosophila Zip and ZnT genes. J Biol Inorg Chem. doi:10.1007/s00775-013-0976-6
Park JH, Schroeder AJ, Helfrich-Forster C, Jackson FR, Ewer J (2003) Targeted ablation of CCAP neuropeptide-containing neurons of Drosophila causes specific defects in execution and circadian timing of ecdysis behavior. Development 130:2645–2656
Phillips MD, Thomas GH (2006) Brush border spectrin is required for early endosome recycling in Drosophila. J Cell Sci 119:1361–1370. doi:10.1242/jcs.02839
Prasad AS (2012) Discovery of human zinc deficiency: 50 years later. J Trace Elem Med Biol 26:66–69. doi:10.1016/j.jtemb.2012.04.004
Qin Q, Wang X, Zhou B (2013) Functional studies of Drosophila zinc transporters reveal the mechanism for dietary zinc absorption and regulation. BMC Biol 11:101. doi:10.1186/1741-7007-11-101
Wang X, Wu Y, Zhou B (2009) Dietary zinc absorption is mediated by ZnT1 in Drosophila melanogaster. Faseb J 23:2650–2661
Wijesekara N et al (2010) Beta cell-specific Znt8 deletion in mice causes marked defects in insulin processing, crystallisation and secretion. Diabetologia 53:1656–1668
Yepiskoposyan H et al (2006) Transcriptome response to heavy metal stress in Drosophila reveals a new zinc transporter that confers resistance to zinc. Nucleic Acids Res 34:4866–4877
Acknowledgments
The Australian Drosophila Biomedical Research Support Facility assisted in the importation and quarantine of fly strains used in this research. All transgenic Drosophila experiments carried out in this research were performed with the approval of the Monash University Institutional Biosafety Committee.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Richards, C.D., Burke, R. Local and systemic effects of targeted zinc redistribution in Drosophila neuronal and gastrointestinal tissues. Biometals 28, 967–974 (2015). https://doi.org/10.1007/s10534-015-9881-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-015-9881-5