Abstract
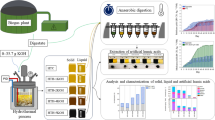
Vinasse is a high pollutant liquid residue from bioethanol production. Due to its toxicity, most vinasse is used not disposed of in water bodies but employed for the fertigation of sugarcane crops, potentially leading to soil salinization or heavy metal deposition. The anaerobic digestion of vinasse for energy production is the main alternative to fertigation, but the process cannot eliminate colored compounds such as melanoidins, caramels, or phenolic compounds. The treatment of raw vinasse with white-rot fungi could remove colored and persistent toxic compounds, but is generally considered cost-ineffective. We report the treatment of vinasse by an autochthonous Trametes sp. strain immobilized in polyurethane foam and the concomitant production of high titers of laccase, a high value-added product that could improve the viability of the process. The reuse of the immobilized biomass and the discoloration of raw vinasse, the concentration of phenolic compounds, BOD and COD, and the phytotoxicity of the treated vinasse were measured to assess the viability of the process and the potential use of treated vinasse in fertigation or as a complementary treatment to anaerobic digestion. Under optimal conditions (vinasse 0.25X, 30 °C, 21 days incubation, 2% glucose added in the implantation stage), immobilized Trametes sp. causes a decrease of 75% in vinasse color and total phenolic compounds, reaching 1082 U L−1 of laccase. The fungi could be used to treat 0.50X vinasse (BOD 44,400 mg O2 L−1), causing a 26% decolorization and a 30% removal of phenolic compounds after 21 days of treatment with maximum laccase titers of 112 U L−1, while reducing COD and BOD from 103,290 to 42,500 mg O2 L−1 (59%) and from 44,440 to 21,230 mg O2 L−1 (52%), respectively. The re-utilization of immobilized biomass to treat 0.50X vinasse proved to be successful, leading to the production of 361 U L−1 of laccase with 77% decolorization, 61% degradation of phenolic compounds, and the reduction of COD and BOD by 75% and 80%, respectively. Trametes sp. also reduced vinasse phytotoxicity to Lactuca sativa seedlings. The obtained results show that the aerobic treatment of vinasse by immobilized Trametes sp. is an interesting technology that could be employed as a sole treatment for the bioremediation of vinasse, with the concomitant the production of laccase. Alternatively, the methodology could be used in combination with anaerobic digestion to achieve greater decolorization and reduction of phenolic compounds, melanoidins, and organic load.
Graphical abstract



Similar content being viewed by others
Data availability
Additional data and fungal strain are available upon request.
Code availability
Not applicable.
References
Ahmed PM (2016) Biorremediación de vinazas de destilerías de alcohol, por microorganismos autóctonos aislados de ambientes contaminados. [Doctoral dissertation: Universidad Nacional de Tucuman]. Sistema Nacional de Repositorios Digitales. Accessed from https://repositoriosdigitales.mincyt.gob.ar/vufind/
Ahmed O, Sulieman AME, Elhardallou SB (2013) Physicochemical, chemical and microbiological characteristics of vinasse, A by-product from ethanol industry. Am J Biochem 3:80–83. https://doi.org/10.5923/j.ajb.20130303.03
Ahmed PM, Pajot HF, De Figueroa LIC, Gusils CH (2018) Sustainable bioremediation of sugarcane vinasse using autochthonous macrofungi. J Environ Chem Eng 6:5177–5185. https://doi.org/10.1016/j.jece.2018.08.007
Ahmed PM, de Figueroa LIC, Pajot HF (2020) Dual Purpose of ligninolytic—basidiomycetes: mycoremediation of bioethanol distillation vinasse coupled to sustainable bio-based compounds production. Fungal Biol Rev. https://doi.org/10.1016/j.fbr.2019.12.001
American Public Health Association, (APHA). Water Environment Federation (WEF) (1995) Standard methods for the examination of water and wastewater. American Public Health Association. Inc., Baltimore
Apohan E, Yesilada O (2017) Production of laccase in olive oil mill wastewater and vinasse by immobilized Trametes versicolor and Trametes trogii with different supports. Fresenius Environ Bull 26:4261–4267. https://doi.org/10.5923/j.ajb.20130303.03
Ba S, Vinoth Kumar V (2017) Recent developments in the use of tyrosinase and laccase in environmental applications. Crit Rev Biotechnol 37:819–832. https://doi.org/10.1080/07388551.2016.1261081
Bilal M, Rasheed T, Nabeel F, Iqbal HMN, Zhao Y (2019) Hazardous contaminants in the environment and their laccase-assisted degradation—a review. J Environ Manage 234:253–264. https://doi.org/10.1016/j.jenvman.2019.01.001
Bourbonnais R, Paice MG (1990) Oxidation of non-phenolic substrates. FEBS Lett 267:99–102. https://doi.org/10.1016/0014-5793(90)80298-W
Calvelo Pereira R, Monterroso C, Macías F (2010) Phytotoxicity of hexachlorocyclohexane: effect on germination and early growth of different plant species. Chemosphere 79:326–333. https://doi.org/10.1016/j.chemosphere.2010.01.035
Castro LEN, Santos JVF, Fagnani KC, Alves HJ, Colpini LMS (2019) Evaluation of the effect of different treatment methods on sugarcane vinasse remediation. J Environ Sci Health Part B 54:791–800. https://doi.org/10.1080/03601234.2019.1669981
Chairattanamanokorn P, Imai T, Kondo R, Sekine M, Higuchi T, Ukita M (2005) Decolorization of alcohol distillery wastewater by thermotolerant white rot fungi. Appl Biochem Microbiol 41:583–588. https://doi.org/10.1007/s10438-005-0106-5
Chiriboga G, De La Rosa A, Molina C, Velarde S, Carvajal C (2020) Energy return on investment (EROI) and life cycle analysis (LCA) of biofuels in Ecuador. Heliyon 6(6):e04213. https://doi.org/10.1016/j.heliyon.2020.e04213
Christofoletti CA, Escher JP, Correia JE, Marinho JFU, Fontanetti CS (2013) Sugarcane vinasse: environmental implications of its use. Waste Manage 33:2752–2761. https://doi.org/10.1016/j.wasman.2013.09.005
Clarke EA, Anliker R (1980) Organic dyes and pigments. In: Hutzinger O (ed) Handbook of environmental chemistry, vol 3. Springer-Verlag, Berlin, pp 181–215
Cortez LAB, Brossard Pérez LE (1997) Experiences on vinasse disposal: part III: combustion of vinasse-# 6 fuel oil emulsions. Braz J Chem Eng 14:9–18. https://doi.org/10.1590/S0104-66321997000100002
da Silva Neto JV, Gallo WLR, Nour EAA (2020) Production and use of biogas from vinasse: implications for the energy balance and GHG emissions of sugar cane ethanol in the brazilian context. Environ Prog Sustain Energy 39(1):13226. https://doi.org/10.1002/ep.13226
de Godoi LAG, Camiloti PR, Bernardes AN, Sanchez BLS, Torres APR, da Conceição Gomes A, Botta LS (2019) Seasonal variation of the organic and inorganic composition of sugarcane vinasse: main implications for its environmental uses. Environ Sci Pollut Res 26:29267–29282. https://doi.org/10.1007/s11356-019-06019-8
del Gobbo LM, Villegas LB, Colin VL (2019) The potential application of an autochthonous fungus from the northwest of Argentina for treatment of sugarcane vinasse. J Hazard Mater 365:820–826. https://doi.org/10.1016/j.jhazmat.2018.11.071
España-Gamboa E, Mijangos-Cortes J, Barahona-Perez L, Dominguez-Maldonado J, Hernández-Zarate G, Alzate-Gaviria L (2011) Vinasse: characterization and treatments. Waste Manage Res 29:1235–1250. https://doi.org/10.1177/0734242X10387313
España-Gamboa E, Vicent T, Font X, Mijangos-Cortés J, Canto-Canché B, Alzate L (2015) Phenol and color removal in hydrous ethanol vinasse in an air-pulsed bioreactor using Trametes versicolor. J Biochem Technol 6(3):982–986
España-Gamboa E, Vicent T, Font X, Dominguez-Maldonado J, Canto-Canché B, Alzate-Gaviria L (2017) Pretreatment of vinasse from the sugar refinery industry under non-sterile conditions by Trametes versicolor in a fluidized bed bioreactor and its effect when coupled to an UASB reactor. J Biol Eng. https://doi.org/10.1186/s13036-016-0042-3
España-Gamboa E, Chablé-Villacis R, Alzate-Gaviria L, Dominguez-Maldonado J, Leal-Baustista RM, Soberanis-Monforte G, Tapia-Tussell R (2021) Native fungal strains from Yucatan, an option for treatment of biomethanated vinasse. Revista Mexicana De Ingeniería Química 20(2):607–620. https://doi.org/10.24275/rmiq/IA2063
Fernandes JMC, Sousa RMOF, Fraga I, Sampaio A, Amaral C, Bezerra RMF, Dias AA (2020) Fungal biodegradation and multi-level toxicity assessment of vinasse from distillation of winemaking by-products. Chemosphere 238:124572. https://doi.org/10.1016/j.chemosphere.2019.124572
Ferreira JA, Brancoli P, Agnihotri S, Bolton K, Taherzadeh MJA (2018) Review of integration strategies of lignocelluloses and other wastes in 1st generation bioethanol processes. Process Biochem 75:173–186. https://doi.org/10.1016/j.procbio.2018.09.006
Fito J, Tefera N, Van Hulle SWH (2019) Sugarcane biorefineries wastewater: bioremediation technologies for environmental sustainability. Chem Biol Technol Agric 6:6. https://doi.org/10.1186/s40538-019-0144-5
Food and Drug Administration (FDA) (1998) Bacterial analytical manual, revision A, 8th edn. AOAC International, Washington, DC
Francisco JP, Folegatti MV, Silva LBD, Silva JBG, Diotto AV (2016) Variations in the chemical composition of the solution extracted from a Latosol under fertigation with vinasse. Rev Ciencia Agron. https://doi.org/10.5935/1806-6690.20160027
Freitas PV, da Silva DR, Beluomini MA, da Silva JL, Stradiotto NR (2018) Determination of phenolic acids in sugarcane vinasse. J Anal Methods Chem. https://doi.org/10.1155/2018/4869487
Fuess LT, Garcia ML (2014) Implications of stillage land disposal: a critical review on the impacts of fertigation. J Environ Manage 145:210–229. https://doi.org/10.1016/j.jenvman.2014.07.003
Fuess LT, Zaiat M (2018) Economics of anaerobic digestion for processing sugarcane vinasse: applying sensitivity analysis to increase process profitability in diversified biogas applications. Process Saf Environ Prot 115:27–37. https://doi.org/10.1016/j.psep.2017.08.007
Gupta A, Jana AK (2018) Effects of wheat straw solid contents in fermentation media on utilization of soluble/insoluble nutrient, fungal growth and laccase production. 3 Biotech 8:1–13. https://doi.org/10.1007/s13205-017-1054-5
Han J, Cao Z, Qiu W, Gao W, Hu J, Xing B (2017) Probing the specificity of polyurethane foam as a ‘solid-phase extractant’: Extractability-governing molecular attributes of lipophilic phenolic compounds. Talanta 172:186–198. https://doi.org/10.1016/j.talanta.2017.05.020
Hoarau J, Caro Y, Grondin I, Petit T (2018) Sugarcane vinasse processing: toward a status shift from waste to valuable resource. A review. J Water Process Eng 24:11–25. https://doi.org/10.1016/j.jwpe.2018.05.003
Janssen R, Rutz DD (2011) Sustainability of biofuels in Latin America: risks and opportunities. Energy Policy 39:5717–5725. https://doi.org/10.1016/j.enpol.2011.01.047
Jeon J-R, Chang Y-S (2013) Laccase-mediated oxidation of small organics: bifunctional roles for versatile applications. Trends Biotechnol 31(6):335–341. https://doi.org/10.1016/j.tibtech.2013.04.002
Koul B, Ahmad W, Singh J (2021) Chapter 30—mycoremediation: a novel approach for sustainable development. In: Kumar A, Kumar SV, Singh P, Kumar MV (eds) Woodhead publishing series in food science, technology and nutrition, microbe mediated remediation of environmental contaminants. Woodhead Publishing, Sawston, pp 409–420
Kumar P (2020) Effect of different agro-waste substrates on yield performance of oyster mushroom (Pleurotus sajor-caju). J Krishi Vigyan 8(2):70. https://doi.org/10.5958/2349-4433.2020.00014.8
Market Watch (2019) Laccase market 2020 industry research, share, trend, global industry size, price, future analysis, regional outlook to 2026 research report [WWW Document]. Accessed from https://www.marketwatch.com/press-release/laccase-market-2020-industry-research-share-trend-global-industry-size-price-future-analysis-regional-outlook-to-2026-research-report-2020-11-18?tesla=y
Marzullo L, Cannio R, Giardina P, Santini MT, Sannia G (1995) Veratryl alcohol oxidase from Pleurotus ostreatus participates in lignin biodegradation and prevents polymerization of laccase-oxidized substrates. J Biol Chem 270:3823–3827. https://doi.org/10.1074/jbc.270.8.3823
Meghana M, Shastri Y (2020) Sustainable valorization of sugar industry waste: status, opportunities, and challenges. Bioresour Technol 303:122929. https://doi.org/10.1016/j.biortech.2020.122929
Molina-Cortés A, Sánchez-Motta T, Tobar-Tosse F, Quimbaya M (2020) Spectrophotometric estimation of total phenolic content and antioxidant capacity of molasses and vinasses generated from the sugarcane industry. Waste Biomass Valor 11(7):3453–3463. https://doi.org/10.1007/s12649-019-00690-1
Moraes BS, Zaiat M, Bonomi A (2015) Anaerobic digestion of vinasse from sugarcane ethanol production in Brazil: challenges and perspectives. Renew Sustain Energy Rev 44:888–903
Nyanhongo GS, Gűbitz G, Sukyai P, Leitner C, Haltrich D, Ludwig R (2007) Oxidoreductases from Trametes spp. in biotechnology: a wealth of catalytic activity. Food Technol Biotechnol 45(3):250–268
Ofosu-Budu GK, Hogarh JN, Fobil JN, Quaye A, Danso SKA, Carboo D (2010) Harmonizing procedures for the evaluation of compost maturity in two compost types in Ghana. Resour Conserv Recycl 54(3):205–209. https://doi.org/10.1016/j.resconrec.2009.08.001
Organisation for Economic Co-operation and Development (OECD)/Food and Agriculture Organization (FAO) (2019) Agricultural Outlook 2019–2028. OECD Publishing Paris/Food and Agriculture Organization of the United Nations, Rome
Osma JF, Toca-Herrera JL, Rodríguez-Couto S (2011) Cost analysis in laccase production. J Environ Manage 92:2907–2912. https://doi.org/10.1016/j.jenvman.2011.06.052
Pandey A (2003) Solid-state fermentation. Biochem Eng J 13:81–84. https://doi.org/10.1016/S1369-703X(02)00121-3
Patel A, Patel V, Patel R, Trivedi U, Patel K (2020) Fungal laccases: versatile green catalyst for bioremediation of organopollutants. Emerging technologies in environmental bioremediation. Elsevier, Amsrerdam, pp 85–129
Peña Balderrama JG, Khatiwada D, Gardumi F, Alfstad T, Ulloa Jimenez S, Howells MI (2020) Integrated analysis of land-use, energy and water systems for ethanol production from sugarcane in Bolivia. DiVA 2:1503850. https://doi.org/10.21203/rs.3.rs-97263/v2
Pinheiro VE, Michelin M, Vici AC, de Almeida PZ, Polizeli MLTM (2020) Trametes versicolor laccase production using agricultural wastes: a comparative study in Erlenmeyer flasks, bioreactor and tray. Bioprocess Biosyst Eng 43:507–514. https://doi.org/10.1007/s00449-019-02245-z
Prado R, Caione G, Campos CNS (2013) Filter cake and vinasse as fertilizers contributing to conservation agriculture. Appl Environ Soil Sci 2013:1–8. https://doi.org/10.1155/2013/581984
Rabelo SC, da Costa AC, Vaz Rossel CE (2015) Industrial waste recovery. Sugarcane. Agricultural production, bioenergy and ethanol. Elsevier, Amsterdam, pp 365–381
Rodrigues Reis CE, Hu B (2017) Vinasse from sugarcane ethanol production: better treatment or better utilization? Front Energy Res. https://doi.org/10.3389/fenrg.2017.00007
Rodríguez-Couto S (2019) Fungal laccase: a versatile enzyme for biotechnological applications. Springer, Cham, pp 429–457
Romanholo Ferreira LF, Aguiar MM, Messias TG, Pompeu GB, Queijeiro Lopez AM, Silva DP, Monteiro RT (2011) Evaluation of sugar-cane vinasse treated with Pleurotus sajor-caju utilizing aquatic organisms as toxicological indicators. Ecotoxicol Environ Saf 74:132–137. https://doi.org/10.1016/j.ecoenv.2010.08.042
Romanholo Ferreira LF, Torres NH, de Armas RD, Fernandes CD, Vilar DS, Aguiar MM, Pompeu GB, Monteiro RTR, Iqbal HMN, Bilal M, Bharagava RN (2020) Fungal lignin-modifying enzymes induced by vinasse mycodegradation and its relationship with oxidative stress. Biocatal Agric Biotechnol. https://doi.org/10.1016/j.bcab.2020.101691
Rulli MM, Villegas LB, Colin LL (2020) Treatment of sugarcane vinasse using an autochthonous fungus from the northwest of Argentina and its potential application in fertigation practices. J Environ Chem Eng. https://doi.org/10.1016/j.jece.2020.104371
Saetang J, Babel S (2010) Effect of glucose on enzyme activity and color removal by trametes versicolor for high strength landfill leachate. Water Sci Technol 62(11):2519–2526. https://doi.org/10.2166/wst.2010.552
Shakerian F, Zhao J, Li S-P (2020) Recent development in the application of immobilized oxidative enzymes for bioremediation of hazardous micropollutants—a review. Chemosphere. https://doi.org/10.1016/j.chemosphere.2019.124716
Shindo H, Huang PM (1984) Catalytic effects of manganese (IV), iron (III), aluminum, and silicon oxides on the formation of phenolic polymers. Soil Sci Soc Am J 48(4):927–934
Silva AFR, Brasil YL, Koch K, Amaral MCS (2021) Resource recovery from sugarcane vinasse by anaerobic digestion—a review. J Environ Manage. https://doi.org/10.1016/j.jenvman.2021.113137
Singh H (2006) Mycoremediation: fungal Bioremediation. Wiley, New Jersey
Sultan M (2017) Polyurethane for removal of organic dyes from textile wastewater. Environ Chem Lett 15:347–366. https://doi.org/10.1007/s10311-016-0597-8
Sun X, Bai R, Ya Y, Wang Q, Fan X, Yuan J, Cui L, Wang P (2013) Laccase-catalyzed oxidative polymerization of phenolic compounds. Appl Biochem Biotechnol 171:1673–1680. https://doi.org/10.1007/s12010-013-0463-0
Timberlake CF, Bridle P (1976) Interactions between anthocyanins, phenolic compounds, and acetaldehyde and their significance in red wines. Am J Enol Vitic 27:97–105
Unuofin JO, Falade AO, Aladekoyi OJ (2021) Applications of microbial laccases in bioremediation of environmental pollutants: potential issues, challenges, and prospects. Bioremediation for Environmental Sustainability. Elsevier, Amsterdam, pp 519–540
Vilar DS, Carvalho GO, Pupo MMS, Aguiar MM, Torres NH, Américo JHP, Cavalcanti EB, Eguiluz KIB, Salazar-Banda GR, Leite MS, Ferreira LFR (2018) Vinasse degradation using Pleurotus sajor-caju in a combined biological—electrochemical oxidation treatment. Sep Purif Technol 192:287–296. https://doi.org/10.1016/j.seppur.2017.10.017
Xu L, Sun K, Wang F, Zhao L, Hu J, Ma H, Ding Z (2020) Laccase production by Trametes versicolor in solid-state fermentation using tea residues as substrate and its application in dye decolorization. J Environ Manage. https://doi.org/10.1016/j.jenvman.2020.110904-
Yamakawa CK, Copa Rivera E, Kwon H, Herrera Agudelo WE, Saad MBW, Leal J (2019) Study of influence of yeast cells treatment on sugarcane ethanol fermentation: operating conditions and kinetics. Biochem Eng J 147:1–10. https://doi.org/10.1016/j.bej.2019.03.022
Zhong C, Zhao H, Cao H, Huang Q (2019) Polymerization of micropollutants in natural aquatic environments: a review. Sci Total Environ 693:133751. https://doi.org/10.1016/j.scitotenv.2019.133751
Zou J, Huang J, Yue D, Zhang H (2020) Roles of oxygen and Mn (IV) oxide in abiotic formation of humic substances by oxidative polymerization of polyphenol and amino acid. Chem Eng J 393:124734
Funding
This research was supported by Agencia Nacional de Promoción Científica y Técnica (ANPCyT) through projects PICT 2013-2016, PICT 0532-2016; PICT 01765-2018, PICT 3500-2018, Estación Experimental Agroindustrial Obispo Colombres (EEAOC), and Consejo de Investigaciones de la Universidad Nacional de Tucumán (CIUNT).
Author information
Authors and Affiliations
Contributions
PMA, Investigation (lead), Writing—original draft (lead), Formal analysis (lead), Writing—Review and Editing (equal); CGN-P, Conceptualization (supporting), Writing—original draft (supporting), Writing—Review and Editing (equal), Formal analysis (supporting); LICF, Funding acquisition, Project administration; HFP, Conceptualization (Lead), Formal analysis (equal), Supervision (lead), Writing—original draft (supporting), Writing—Review and Editing (equal).
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ahmed, P.M., Nieto-Peñalver, C.G., de Figueroa, L.I.C. et al. Vinasse odyssey: sugarcane vinasse remediation and laccase production by Trametes sp. immobilized in polyurethane foam. Biodegradation 33, 333–348 (2022). https://doi.org/10.1007/s10532-022-09985-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10532-022-09985-y