Abstract
Despite continued wild harvesting, there is limited data that provides estimates on the availability of African edible orchids as a resource, and that identifies the ecological drivers of their dynamics. This study was designed to investigate the relationship between distance from surface water and the diversity, frequency, and population density of chikanda orchids in a harvesting wetland in Mwinilunga, Zambia. Vegetation sampling was conducted using an interrupted belt transect design. Eight 50 m transects were systematically placed and positioned perpendicular (90o) to a stream within three separate orchid communities. Individual plant count was recorded within the 0–10 m, 10–20 m, 20–30 m, 30–40 m, and 40–50 m distance zones. Results showed a statistically significant decrease in the Shannon’s Diversity Index between the distance zones in all three communities (p < 0.05). Mean ranks for orchid taxa frequency and density across the distance zones were significantly different for communities 1 and 2 but not 3 and communities 1 and 3 but not 2, respectively. However, there was a medium to large effect size for distance on frequency and a small to large effect size for distance on density in all three communities. These results suggest that distance from surface water influences the frequency and density of orchid taxa occurring in wetland ecosystems. This implies that for conservation purposes, it may be important to focus on areas close to water when establishing orchid sites for the species investigated in this study.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Orchids belong to one of the largest and most diverse plant families, with 880 genera and approximately 28 000 species (Christenhusz and Byng 2016). Of these species, 1 373 are found in sub-Saharan Africa, with 637 species occurring exclusively in Southern Africa (Bakayoko et al. 2022). Orchids are considered vulnerable to ecosystem changes, regarding abiotic factors such as moisture content, light regime, and nutrient availability (Swarts and Dixon 2009). In addition, various land use systems also present a threat to their habitats and abundance. Occurring in diverse types of ecosystems, in Central and Southern Africa, orchids are particularly harvested from extensive wetland areas. Wetland ecosystems are part of the larger Miombo ecoregion that is estimated to cover a total area of about 2.7 million km2 across parts of Angola, Botswana, Burundi, the Democratic Republic of Congo, Malawi, Mozambique, Namibia, Tanzania, Zambia, and Zimbabwe. Wetlands primarily consist of swamps or marshes, floodplains and dambos, where water either sits all year round or floods annually (Timberlake 1998). Dambos, for the purpose of this paper, are defined as shallow waterlogged depressions in the headwaters of streams or rivers and have a significant role in agriculture and water networks (von der Heyden 2004).
Covering 10–15% of Zambia, dry dambos have extensive grass cover, and vary in shape and size, with poorly drained soils ranging from sandy loam to clay soils and organic-rich to peat soils (Chidumayo 1992; Mäckel 1974; Taylor et al. 1995; von der Heyden 2004). Wet dambos, on the other hand, represent about 1% of the total dambo area in Miombo woodland and are usually covered by extensive evergreen forest (Chidumayo 1992). Thus, ecological changes to dambos are primarily caused by changes in rainfall and occurrences of drought. However, increased use of dambos for socio-economic purposes adds challenges to minimising threats to these ecosystems and, therefore, to orchid populations. Additionally, these regions are ecologically sensitive, and their conditions are rapidly declining, where 57% are moderately degraded and 27% are severely degraded (Kotze and Wood 2021; Musasa and Marambanyika 2020). While dambos are rich in orchid species, their importance for the growth and sustainable utilisation of terrestrial wild-harvested orchids in Zambia is yet to become an important consideration with regard to policy aimed at their conservation and monitoring.
Zambia is home to 412 orchid species from 46 genera and 22 of these are endemic (Bakayoko et al. 2022). A wide range of edible orchid species are harvested from the wild for their social, cultural, and economic importance. The most marketable species are usually from three genera, Habenaria, Satyrium, and Disa (Hinsley et al. 2018; Veldman et al. 2018). Other harvested species include Eulophia, Brachycorythis, Platycoryne, Neobolusia, Roeperocharis, Cynorkis and Disperis (Challe et al. 2018; Challe and Price 2009; Challe and Struik 2008; Hamisy 2007; Namoto 2018).
Although the distribution of African orchids is widely known, information on the distribution, abundance and the density of individual species is inadequate. The aim of the current study was to document a point-in-time inventory of chikanda orchids and determine the relationship between distance from surface water source and the abundance of terrestrial orchid communities in a wild harvesting dambo in Mwinilunga District. We address the following questions: (1) What is the relationship between distance from surface water on orchid diversity, frequency, and density? (2) What is the state of chikanda orchid populations in a wild harvested wetland area in Mwinilunga, Zambia?
Materials and methods
Study area
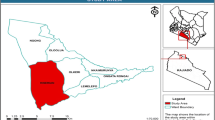
This study was conducted in the Mwinilunga district of North-Western Province, Zambia from the 11th to the 23rd of October 2021. Mwinilunga was chosen because orchid harvesting is relatively new in North-Western Zambia when compared to other more traditional harvesting sites in Northern Zambia. The annual average precipitation in Mwinilunga is 231 mm with annual minimum and maximum temperatures of 32.5°C and 10.8°C, respectively (Weather Atlas 2022). Mwinilunga has five major wetlands that are important for the harvesting of orchids. Chiwela dambo (11º27’50” S 24º37’58” E to 11º28’15” S 24º37’34” E), sized at approximately 90 ha, with an altitude of between 1 371 m and 1 374 m, was selected as the study site. Within this study site, three orchid communities were surveyed (Fig. 1).
Chiwela dambo lies within the Miombo ecosystem and is characterised as an open grassland-dominated (grasses, rushes, and sedges) wetland occupying a shallow, seasonally waterlogged depression at the headwater of Chiwela stream, while surrounded by Miombo woodland (Roberts 1988). At the time of the survey, 20 species (except orchids) belonging to 9 families were observed and documented to be characteristic of Chiwela dambo at that time of the year: Agathisanthemum globosum, Arthropteris orientalis (= A. albopunctata), Bulbostylis filamentosa, Cenchrus sp. (= Pennisetum sp.), Cyperus macrostachyos (= Cyperus tremulus), Digitaria gazensis, Eragrostis sp., Gladiolus atropurpureus, Gladiolus erectiflorus, Hyparrhenia diplandra, Mesanthemum sp., Myrica serrata, Nidorella auriculata, Nidorella welwitschii (= Conyza welwitschii), Pyrotis gilgiana (= Dissotis gilgiana), Rosettea falcipila (= Dissotis falcipila), Scleria greigiifolia, Scleria porphyrocarpa, Urochloa brizantha (= Brachiaria brizantha), and Urochloa clavipila (= Brachiaria clavipila).
Study species
The following descriptions of the studied species were obtained from the Royal Botanic Gardens KEW Plants of the World and The Journal Storage (JSTOR) plant databases (Fig. 2).
Chikanda orchids and tubers. a) Disa hircicornis; b) Satyrium trinerve; c) Brachycorythis cf. friesii; d) Disa hircicornis dissected tuber; e) Platycoryne guingangae; f) Eulophia angolensis; g) Satyrium buchananii; h) Satyrium trinerve dissected tuber; Photographs a-c and f-g by Graham Reed and d, e, h by Chimwemwe Tembo
Brachycorythis friesii is a slender, erect 8–27 cm tall herb. It occurs in shallow soil, seepage areas and open woodland areas between 1200 and 1650 m in altitude. The flowers are almost white, pale to deep pink or purple with darker marks on lip, blooming between December and February.
Disa hircicornis is a slender to stout glabrous herb between 30 and 76 cm tall. It occurs in damp grassy areas and marshes between 1500 and 1800 m in altitude. The inflorescence is between 2.5 and 20 cm long and consists of a dense cylindrical spike of 20–70 variable shades of purple flowers with dark markings. The tubers can be elongated-elliptical to elongated-ovoid and it flowers between December and February (Namoto 2018).
Platycoryne guingangae is 30–50 cm tall with ovoid tubers the size of about 10 × 8 mm. It has orange flowers that are erect with a short inflorescence and primarily grows in permanently wet dambo or sandy grassland between the altitude range of 1350–1500 m. It flowers between October and December (Williamson 1977).
Satyrium buchananii is a strong, glabrous herb of between 30 and 70 cm which occurs in dambos and wet montane grasslands between the altitude range of 1 300 m and 2 300 m. The flowers are fragrant, white, rarely spotted mauve, blooming between October and February. Tubers are tomentose, ellipsoid, ovoid or elongate ovoid with a size of about 1.5-4 × 1.2 cm.
Satyrium trinerve is between 30 and 108 cm in height. It is widely distributed and naturally occurs in grasslands, dambos and Brachystegia woodlands between the altitude range of 550 m to 2 300 m. The flowers are white and sometimes yellow, and the sepals and petals are joined together in the basal. Tubers are up to 4.5 cm long, ellipsoid to fusiform and woolly. Nutritionally, they are high in protein, calcium, phosphorus, and iron (Malaisse and Parent 1985). Flowering time is usually from December to March.
Vegetation sampling
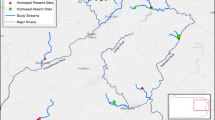
We subjectively identified a total of three dense populations of multiple orchid species i.e., Community 1 (C1), Community 2 (C2) and Community 3 (C3), within 200 m x 50 m sampling areas (Fig. 1). Sampling for C2 started at an average of 200 m from the stream due to the inaccessibility of the stream and a lack of orchid plants. However, sampling for C1 and C3 was possible 5 m from the stream. Both the C2 and C3 sites were waterlogged due to their proximity to the water source but C3 had a characteristic hydromorphic, peaty upper soil horizon that C1 and C2 did not have.
An interrupted belt transect experimental design was used. Eight 50 m transects (plots/replications) were systematically established 25 m apart and positioned perpendicular (90o) to the stream within each orchid community. A total of 16 quadrants were sampled per distance zone. On alternate sides of the transect, 2 × 2 m quadrants (sub-plots/sample sites) made from polyvinyl chloride pipes were placed. A coin was used to determine which side of the transect the quadrant would be placed. The quadrants were placed at 5 m intervals along the transect, 2 m from the edge of each distance zone. Distance from the water source was classified into five groups: 0–10 m, 10–20 m, 20–30 m, 30–40 m and 40–50 m where each identified orchid species was counted per individual plant.
Data analysis
Orchid diversity indices, and subsequent diversity t-tests, among the three orchid communities were determined using Paleontological Statistics (PAST) Version 4.03. Shannon’s diversity index (H’) was calculated to estimate the diversity of the species within each distance zone for each orchid community sampled. Additionally, the Shannon evenness index (varies between 0 and 1-complete evenness) was determined to measure how evenly individuals are distributed across the sample while the Berger-Parker dominance (d) was provided to demonstrate the proportional importance of the most abundant type (Pielou 1966).
A Kruskal-Wallis H test was run using IBM SPSS Statistics Version 28 (190) to determine if there were differences in the total orchid relative frequency and population density between the varying distance classes from the water source, per orchid community (Kruskal and Wallis 1952). Furthermore, the Kruskal-Wallis H test was used to compare the total orchid and individual edible orchid tuber species mean population sizes across the three sampled orchid communities. The chi-square statistic (χ2) is used to show significance for Kruskal-Wallis H test for clarity on the use of the asymptotic significance rather than the exact significance. Pairwise comparisons with a Bonferroni correction for multiple comparisons were conducted for statistically significant results between the distance zones (Dunn 1964). The epsilon-squared estimate of effect size was also calculated to evaluate the strength of the association between distance zones and the parameters measured (Tomczak and Tomczak 2014). A Mann-Whitney U test was used as a follow up test to determine the differences between the zones when the Bonferroni comparison test was insufficient (Mann and Whitney 1947). All figures were generated using Excel version 2023 (Build 16227.20258). Additionally, relative frequency, population density and population size were calculated in Excel with the following formulae:
Results
Edible, wild harvested chikanda orchids made up most of the sampled orchids within the Chiwela dambo area. A total of six orchid species from five genera were present in the orchid communities at the time of sampling (Fig. 2). Most of the orchids surveyed were edible species, Satyrium trinerve, S. buchananii, Disa hircicornis and Brachycorythis friesii. Of these species, the local communities were only aware of Satyrium trinerve, S. buchananii, and Disa hircicornis as being edible so these are the only ones that were harvested. Platycoryne guingangae and Eulophia angolensis were the two non-edible species sampled.
Diversity measures for chikanda orchid communities
There was a statistically significant difference in the Shannon’s Diversity Index between the distance zones in all three communities (p < 0.05). There was a general decreasing trend in the Shannon Weiner’s diversity index in all three community profiles with a sharp increase in the 40–50 m zone for communities 1 and 2 (Fig. 3). While there was a general decreasing trend in species diversity as distance increased from the surface water source, the Shannon Weiner’s index increased sharply within the 40–50 m zone in C1 and C2. No orchid species occurred in distance zone 0–10 m for C1 (Fig. 3).
There was a statistically significant difference in the Shannon’s Diversity Index between the distance zones 30–40 m and 10–20 m (t(10.02) = 2.804, p < 0.05), and 20–30 m (t(18.58) = 4.785, p < 0.05), and 40–50 m (t(274.46) = 7.706, p < 0.05) in C1 (Table 1).
The diversity index in C2 was also statistically significantly different between zones 40–50 m and 0–10 m (t(324.32) = 2.657, p = 0.008), 10–20 m (t(402.25) = 3.674, p < 0.05), 20–30 m (t(356.71) = 5.13, p < 0.05), and 30–40 m (t(316.54) = 3.713, p < 0.05). Lastly, for C3, there were significant differences in orchid diversity between the distance zones 30–40 m and 0–10 m (t(92.22) = 2.812, p = 0.006), and 10–20 m (t(101.04) = 2.837, p = 0.005). In addition, there were differences between distance zones 40–50 m and 0–10 m (t(221.41) = 6.558, p < 0.05), 10–20 m (t(249.03) = 6.378, p < 0.05), and 20–30 m (t(270.95) = 4.764, p < 0.05).
For all three communities in Table 1, orchid taxa diversity followed the trend that an increase in dominance leads to a decrease in evenness and the Shannon Wiener’s index. For orchid community 3, the evenness decreased alongside the Shannon Wiener’s index while the Berger-Parker (d) dominance index increased with distance from the water source. Species evenness increased with distance from the water source in C2 while it decreased for C1 and C3.
Frequency measures and population density for total orchids sampled across distance per orchid community
Distributions of the total orchid relative frequency were not similar for all groups, as assessed by a visual inspection of a box plot. Pairwise comparisons were performed with a Bonferroni correction for multiple comparisons for statistically significant results between the distance classes. The distributions of the total orchid count were not similar for all distance classes per orchid community, as assessed by a visual inspection of a boxplot. Therefore, mean ranks are reported (Table 2).
Orchid taxa relative frequency
Relative frequency was used to measure the number of times orchid taxa were present within the quadrants sampled (Fig. 4). The distributions of the total orchid relative frequency were not similar for all distance classes per orchid community, as assessed by a visual inspection of a boxplot.
Specifically, the distributions of the orchid taxa’s mean relative frequency for C1 were statistically significantly different between the groups (χ2 (4) = 12.078, p = 0.017). The post hoc analysis revealed statistically significant differences in relative frequency mean ranks between the 0–10 m (mean rank = 29.5) and 30–40 m (mean rank = 49.28) (p < 0.022) distance classes but not between the 10–20 m (mean rank = 38.78) and 20–30 m (mean rank = 37.84) and 40–50 m (mean rank = 47.09) or any other group combination. The large effect size (E2R = 0.153) showed a strong relationship between distance and mean relative frequency.
The distributions of the orchid taxa’s mean relative frequency for C2 were statistically significantly different between the groups (χ2 (4) = 11.346, p = 0.023). The post hoc analysis revealed statistically significant differences in the mean ranks of relative frequency between the 20–30 m (mean rank = 27.22) and 40–50 m (mean rank = 51.66) (p = 0.017) but not between 0 and 10 m (mean rank = 43.84), 10–20 m (mean rank = 35.94), 30–40 m (mean rank = 43.84) or any other group combination. The large effect size (E2R = 0.144) showed a strong relationship between distance and mean relative frequency.
For C3, mean relative frequency remained constant between the 0–10 m, 10–20 m, and 20–30 m (mean rank = 35.16) distance zones then increased in the 30–40 m (mean rank = 48.59) distance zone. Finally, it decreases at the 40–50 m (mean rank = 48.44) distance zone. However, mean relative frequency ranks were not statistically significantly different between the distance zones (χ2(4) = 6.732, p = 0.151). The medium effect size (E2R = 0.085) showed a moderate relationship between distance and mean relative frequency.
Orchid taxa population density
The distributions of the total orchid population density were not similar for all distance classes per orchid community, as assessed by a visual inspection of a boxplot. The mean population densities for the chikanda orchid taxa assessed is visually represented in Fig. 5.
The distributions of the orchid taxa’s mean population density for C1 were statistically significantly different between the groups (χ2 (4) = 17.561, p = 0.002). The post hoc analysis revealed statistically significant differences in the mean ranks of population density between the 0–10 m (mean rank = 29.5) and 30–40 m (mean rank = 52.63) (p = 0.003), and 40–50 m (mean rank = 48.22) (p = 0.04) zones but not between 10 and 20 m (mean rank = 35.97), 20–30 m (mean rank = 36.19) distance zones or any other group combination. The large effect size (E2R = 0.222) showed a strong relationship between distance and mean population density.
For C2, the orchid taxa’s mean population density increased from the 0–10 m (mean rank = 39.5) distance class to 10–20 m (mean rank = 41.34), to 20–30 m (mean rank = 46.63), then decreased from 30 to 40 m (mean rank = 39.34), to 40–50 m (mean rank = 35.69). Population density mean ranks were not statistically significantly different between the distance classes (χ2(4) = 1.901, p = 0.754). The small effect size (E2R = 0.024) showed a weak relationship between distance and mean population density.
The distributions of the orchid taxa’s population density for C3 were statistically significantly different between the groups (χ2 (4) = 12.243, p = 0.016). The post hoc analysis revealed a statistically significant difference in the population density between the 10–20 m (mean rank = 50.34) and 30–40 m (mean rank = 26.34) (p = 0.034) distance zones. On the other hand, there was no statistically significant difference between zones 0–10 m (mean rank = 46.47), 20–30 m (mean rank = 45.84), 40–50 m (mean rank = 33.50), and any other group combination. The large effect size (E2R = 0.155) showed a strong relationship between distance and mean population density.
Frequency and density measures per species per distance zone
The distributions of the total orchid count were not similar for all distance classes per orchid community, as assessed by a visual inspection of a boxplot. Therefore, mean ranks are reported (Table 3).
Satyrium trinerve
The distributions of the mean relative frequency of S. trinerve in orchid Communities 2 and 3 were the same across the distance zones (Fig. 4). However, in C1, the distributions of mean relative frequency were statistically significantly different between the distance zones (χ2 (4) = 14.887, p = 0.005). The post hoc analysis showed a statistically significant difference in the population density between the 0–10 m (mean rank = 32.00) and 30–40 m (mean rank = 50.50) (p = 0.016) distance zones. On the other hand, there was no statistically significant difference between zones 10–20 m (mean rank = 35.00), 20–30 m (mean rank = 37.69), 40–50 m (mean rank = 47.31), and any other group combination. The large effect size (E2R = 0.188) showed a strong relationship between distance and mean relative frequency.
Similarly, the distribution of the mean population density of S. trinerve in orchid C2 and C3 were the same across the distance zones. However, in C1, the distributions of mean population density were statistically significantly different between the distance zones (χ2 (4) = 18.448, p = 0.001). The post hoc analysis showed a statistically significant difference in the population density between the 30–40 m (mean rank = 52.84) and 0–10 m (mean rank = 32.00) (p = 0.004), 10–20 m (mean rank = 34.47) (p = 0.018), and 20–30 m (mean rank = 36.34) (p = 0.05) distance zones. On the other hand, there was no statistically significant difference between the zone 40–50 m (mean rank = 46.84), and any other group combination. The large effect size (E2R = 0.234) showed a very strong relationship between distance and mean relative frequency.
Satyrium buchananii
Satyrium buchananii only occurred in C3. The distributions of its mean relative frequency were the same across the distance zones (Fig. 4c). There were no statistically significant differences between the distance zones (χ2 (4) = 2.157, p = 0.707). The small effect size (E2R = 0.027) showed a weak relationship between distance and relative frequency. For S. buchananii’s mean population density, its distribution was the same across the distance zones. There were no statistically significant differences in mean population density between the distance zones (χ2 (4) = 3.727, p = 0.444). The small effect size (E2R = 0.047) showed a weak relationship between distance and mean population density.
Disa hircicornis
The distributions of the mean relative frequency of D. hircicornis was the same across the distance zones in all three orchid communities (Fig. 4). There were no statistically significant differences between the distance zones (p > 0.05). However, the large effect size (E2R = 0.108) showed a strong relationship between distance and mean relative frequency within C1 where D. hircicornis occurred mostly in the last distance zone.
The distributions of the mean population density of D. hircicornis were the same across the distance zones in all three orchid communities. There were no statistically significant differences between the distance zones (p > 0.05). However, the large effect size (E2R = 0.108) for C1 showed a strong relationship between distance and mean population density where D. hircicornis occurred most in the last distance zone.
Brachycorythis friesii
Brachycorythis friesii only occurred in C2 and C3. The distributions of the mean relative frequency of B. friesii, in both communities, were the same across the distance zones (Fig. 4b,c). There were no statistically significant differences in mean relative frequency between the distance zones (p > 0.05). However, the medium effect size (E2R = 0.077) showed a moderate relationship between distance and mean relative frequency within C2.
The distributions of the mean population density of B. friesii, in both communities, were the same across the distance zones. There were no statistically significant differences in mean relative frequency between the distance zones (p > 0.05). However, the medium effect size (E2R = 0.078) showed a moderate relationship between distance and population density within C2.
Platycoryne guingangae
Platycoryne guingangae only occurred in C3. The distributions of the mean relative frequency of P. guingangae were not the same across the distance zones (Fig. 4c). There were statistically significant differences in mean relative frequency between the distance zones (χ2 (4) = 16.14, p = 0.003). The post hoc analysis showed a statistically significant difference in the mean relative frequency between the 40–50 m (mean rank = 26.06) and 0–10 m (mean rank = 47.91) (p = 0.003), 10–20 m (mean rank = 51.00) (p = 0.001), and 20–30 m (mean rank = 44.59) (p = 0.013) distance zones. Additionally, there were statistically significant differences between the distance zones 30–40 m (mean rank = 32.94) and 0–10 m (p = 0.044), and 10–20 m (p = 0.015). However, the large effect size (E2R = 0.204) showed a strong relationship between distance and mean relative frequency within C3.
Similarly, the distributions of the mean population density of P. guingangae were not the same across the distance zones. There were statistically significant differences in mean population density between the distance zones χ2 (4) = 30.45, p < 0.001. The post hoc analysis showed a statistically significant difference in the mean relative frequency between the 40–50 m (mean rank = 24.28) and 0–10 m (mean rank = 57.09) (p < 0.001), and 10–20 m (mean rank = 52.88) (p = 0.008) distance zones. Additionally, there were statistically significant differences between the distance zones 30–40 m (mean rank = 27.84) and 0–10 m (p = 0.001), and 10–20 m (p = 0.008). Additionally, the large effect size (E2R = 0.385) showed a very strong relationship between distance and mean population density of P. guingangae.
Eulophia angolensis
The distributions of the mean relative frequency of E. angolensis, were the same across the distance zones in all three communities (Fig. 4). There were no statistically significant differences in mean relative frequency between the distance zones (p > 0.05). However, the medium effect size for C1 (E2R = 0.077) and large effect size (E2R = 0.117) for C2 showed a medium and a strong relationship between distance and mean relative frequency of E. angolensis, respectively.
The distributions of the mean population density of E. angolensis in C1 and C2, were the same across the distance zones. There were no statistically significant differences in mean population density between the distance zones (p > 0.05). However, the medium effect size (E2R = 0.078) showed a moderate relationship between distance and population density within C1. On the other hand, there were statistically significant differences in mean population density between the distance zones (χ2 (4) = 10.89, p = 0.028) in C3. The post hoc analysis showed a statistically significant difference in the mean population density between the 20–30 m (mean rank = 35.00) and 0–10 m (mean rank = 40.19) (p < 0.021) but not between any other distance combination. The large effect size (E2R = 0.138) showed a strong relationship between distance and mean population density of B. friesii within C3.
Population size differences
We tested for differences in the population sizes between the orchid communities and as well as within the edible species because of these orchids’ socio-economic importance in Zambia.
Total orchid population size differences across the three communities
A Kruskal-Wallis test showed that the distribution of the total orchid population size was not the same across the three orchid communities. The distribution of the total orchid mean population sizes was statistically significantly different across the orchid communities (χ2 (3) = 10.868, p = 0.004). A post hoc analysis showed a statistically significant difference in the total orchid mean population size between the C1 and C2 (p = 0.003) with a large effect size (E2R = 0.375) showing a strong relationship between the orchid community and mean population size.
Edible orchid population size differences across the three communities
Since S. buchananii only occurred in orchid C3, no test was conducted on it. However, for S. trinerve, D. hircicornis and B. friesii, there were statistically significantly different mean population sizes between the orchid communities (p < 0.05).
A post hoc analysis showed a statistically significant difference in the mean population size between the C1 and C2 (p = 0.006) for S. trinerve, with a large effect size (E2R = 0.331) showing a strong relationship between the orchid community and population size for this species. For D. hircicornis, there was a statistically significant difference between C1 and C2 (p = 0.02) and between C2 and C3 (p = 0.001), with a large effect size (E2R = 0.424) showing a very strong relationship between the orchid community and population size for D. hircicornis. Lastly, the post hoc analysis for B. friesii showed a statistically significant difference in the population size between C1 and C2 (p = 0.051) with a large effect size (E2R = 0.216) showing a strong relationship between the orchid community and population size for B. friesii.
Discussion
To address the limited information available on the abundance of Convention on International Trade in Endangered Species (CITES)-listed African edible orchid species, this study aimed to document a point-in-time inventory of chikanda orchids within a wild harvesting wetland in Zambia. Three orchid communities were identified within a dambo site and the relationship between the distance from the surface water source and orchid diversity, frequency and density were investigated. The results demonstrated that there were significant differences in orchid taxa diversity, frequency, and population density with distance from surface water within the wetland area.
Effect of distance on orchid diversity
In this study, the orchid species diversity decreased as distance from the surface water increased, in all three communities. The decreasing trend of the orchid species diversity may be because diversity is low when a few species contain a large majority of the individuals observed. In our case, those species are S. trinerve, P. guingangae and D. hircicornis.
Tsheboeng (2018) and Tsheboeng et al. (2017) also found that riparian plant community diversity varied between the different sampled sites as well as along the distance class gradient. Variation in species diversity in these cases was attributed to the intermediate disturbance hypothesis and the depth of the water table within a wetland ecosystem. In the present study, for communities 1 and 2, species diversity increases at the 40–50 m zone, which may indeed be due to this zone having an intermediate disturbance level where the species occurring have a coexistence relationship (Mccormick and Jacquemyn 2014). The increase in species diversity within this zone may be because of the presence and co-existence of both competitive and opportunistic species. With chikanda orchids being strongly associated with their specialised orchid mycorrhizal fungi (OMF), it is possible that OMF becomes an important contributor to the coexistence within this zone (Bone 2017; Chapagain et al. 2021; Djordjević et al. 2022; Frost 1996; Mccormick and Jacquemyn 2014; Rasmussen et al. 2015). A study by Jasinge et al. (2018) showed how orchid populations increased along with their accompanying OMF, particularly after burning had occurred. Notably, some of the species identified in this study are reportedly stimulated by burning, a frequent practice for local communities utilising dambos (Jasinge et al. 2018; Nyamadzawo et al. 2014).
The general decrease in species diversity along the distance zone gradient among all three orchid communities may also be attributed to not just the obvious water table depth but also the thickness of the vegetative cover. However, out of five vegetation types in the Nyika National Park, Namoto (2018) observed the highest number of edible orchids in a Montane wetland area, followed by a Montane grassland area. In that study, D. hircicornis was restricted to the wetland area while S. trinerve and S. buchananii occurred in both wetland and grassland areas. Therefore, the time of the survey, three months before harvesting starts, may have had an influence on species diversity as few orchid species had sprouted.
Effect of distance on relative frequency
Out of the six species sampled, only S. trinerve and P. guingangae seemed to have significant differences in their mean relative frequency across the distance zones. The general trend of the mean relative frequency for S. trinerve in C1 was increasing while remaining constant in C2 and decreasing in C3. However, it was only in C1 where the mean relative frequencies of S. trinerve had significant differences between the distance zones, with a strong association between distance and where it occurred. It should be noted that S. trinerve, D. hircicornis and E. angolensis occurred in all three communities. Platycoryne guingangae was restricted to the marshlands of C3 and showed a statistically significant decreasing trend in mean relative density across the distance zones (p < 0.05) with a strong association between mean relative frequency and distance. Similarly, S. buchananii was restricted to the marshlands of C3 but there were no significant differences in its mean relative frequency across the distance zones. A study by (Djordjević et al. 2022) demonstrated the importance of the bedrock type within a wetland vegetation in separating niches of orchid taxa. It may be that P. guingangae and S. buchananii are restricted to the marshland because they favour a particular geological substrate present in C3 but absent in C1 and C2. Additionally, in C3, the ground cover close to the water source consisted of a peaty upper soil horizon which then changes into a grassland vegetation at a distance away from the water. Peat moss occurs when plant material does not fully decay in the acidic and anaerobic conditions created when there is exposure to high moisture content for extended periods of time as is the case with a dambo. It can, therefore, be assumed that these conditions are most favourable for the orchids occurring in this community.
While there were no significant differences in the mean relative frequency of D. hircicornis and E. angolensis, there was a strong association between the mean relative frequencies of these species and the distance zone. A range of factors may contribute to the occurrence of the varied species within their community profiles including accompanying vegetation, soil, and nutrient content, associated mycorrhiza, and climatic conditions (Mccormick and Jacquemyn 2014).
Effect of distance on population density
Population density was used to measure how the studied orchids responded to their local conditions with higher densities indicating good conditions. While for C1, orchid taxa density increased as distance from the water source increased, the opposite was true for C3, where density decreased as distance from the water source increased. The dynamic occurring in C3 may be because of the vegetative composition change with distance from the water source. On the other hand, the distribution of the mean population density of the orchid taxa across the distance zones in C2 was the same so there were no statistically significant differences. Hence, orchid C2 and C3 had the most favourable conditions for orchid growth because they have the highest orchid taxa densities.
Of the edible orchids, Satyrium trinerve was the most abundant species and there were statistically significant differences in its mean population density in C1. The mean population density for the orchid taxa ranged from 0.93 plants per square meter to 3.3 plants per meter square. Satyrium trinerve is known to be abundant and prevalent in sub-Saharan Africa and is currently classified as Least Concern (LC) on the IUCN (International Union for Conservation of Nature) Red list and the Red list of South African plants (Barker et al. 2018). With an average population density of 1.743 plants per square meter, the Least Concern classification aligns with the results of this study in that Satyrium was present and in abundance, in all orchid communities. The estimated population densities for S. buchananii (0.1 plants per square meter) more closely match those conducted on the Lebanon endemic Orchis galilaea that belongs to the genus targeted for salep harvesting (Machaka-Houri et al. 2012). In their study, Machaka-Houri et al. (2012) estimated a density of 0.08 individuals per square meter. But S. buchananii has not been evaluated for its conservation status. Interestingly, it is one of the more desired species for trade and consumption (Veldman et al. 2018). Additionally, the conservation status of B. friesii and P. guingangae has not yet been assessed.
On the other hand, D. hircicornis and E. angolensis are of Least Concern on the red list of South African plants. The smaller densities found in this study of both D. hircicornis and E. angolensis in comparison to S. trinerve suggests that S. trinerve likely outcompetes the other two, which is what we may expect to find in a situation where orchid species seem to co-occur (Mccormick and Jacquemyn 2014). Another possible reason, for the smaller density of D. hircicornis in particular, may be that it is more negatively impacted by the harvesting in comparison to S. trinerve therefore needing a longer regeneration period (Challe and Struik 2008). This assumption was tested by Chapagain et al. (2021) who determined that the density of different life cycles of the endangered medicinal orchid, Dactylorhiza hatagirea, significantly decreased through diminished reproductive outputs. Similar research into the impact of wild harvested epiphytic orchids has been conducted (Emeterio-Lara et al. 2021). With mostly unregulated harvesting occurring in Zambia and its neighbouring countries, orchid tuber species requiring a longer resting phase due to harvesting impacts and other non-anthropogenic reasons such as regeneration patterns, plant growth strategies, mycorrhizal association, or habitat conditions, may not have that opportunity (Chapagain et al. 2021; Ticktin 2015; Ticktin et al. 2023). However, the fact that some studies have shown that there can be no significant differences between the density of orchids between protected and non-protected areas suggests that more long term research is needed to understand the impact of wild harvesting on marketable edible orchid tuber populations (Assédé et al. 2018; Challe and Struik 2008; Charitonidou et al. 2019).
Larger population sizes ranging from 6.8 to 218.8 plants per hectare have been simulated for wild harvested salep orchid populations (Charitonidou et al. 2019). This underlines the need for more rigorous orchid population assessment from diverse harvesting sites over prolonged periods of time to better understand orchid density responses to ecological factors and human stresses. However, Machaka-Houri et al. (2012) caution against using population density as an indicator of species vitality because they also found that it was negatively correlated to reproduction ability. None of the other species sampled are listed on the IUCN Red list but all orchid species are listed on CITES.
Edible orchid population size
Our findings revealed that there was a strong relationship between the sampled orchid communities and the mean population size. For this study, it was important to estimate the population sizes of the edible orchids to provide some baseline figures regarding what to expect at the time of sampling. Therefore, the population sizes of the edible orchids were as follows: S. trinerve at 17 430 individuals/ha, D. hircicornis at 2 240 individuals/ha, S. buchananii at 1 000 individuals/ha, and B. friesii at 140 individuals/ha. In a similar study conducted within the Nyika National Park in Malawi, the highest population sizes determined for the same species were as follows: S. trinerve at 533 individuals/ha, D. hircicornis at 920 individuals/ha, S. buchananii at 1 093 individuals/ha, and B. friesii at 460 individuals/ha (Namoto 2018). Notably, a better estimate would be obtained given at least one measurement of orchid taxa per month for the six to eight annual orchid harvesting months. The current study took advantage of the pre-harvesting season to collect some baseline data. That being said, Croix and Croix (1983) reported only seeing one individual flowering plant of Brachycorythis pubescens, a sister species to B. friesii, annually. In addition, Brachycorythis conica subsp. transvaalensis is listed as Critically Endangered A3c on the Red List of South African plants, with only 117 plants surviving in South Africa (Peter et al. 2017). This suggests that the genus Brachycorythis may not naturally occur in large population sizes but rather in small ones.
Both S. trinerve and S. buchananii are desired species for use in the preparation of the chikanda orchid cake (Veldman et al. 2017, 2018). Interestingly, the harvesting community of interest mostly harvested S. trinerve but since S. buchananii can be mistaken for S. trinerve, it is highly likely that S. buchananii is harvested indiscriminately alongside S. trinerve. In addition, S. buchananii may be more at risk considering its restriction to the marshland and the fact that its density is 17,5 times less than that of S. trinerve. Consequently, based on the assessment of the edible orchid populations within this study, we make a conservative statement that S. buchananii and B. friesii are more at risk with regard to their conservation status than S. trinerve and D. hircicornis, and may have a decreasing population trend if measured over time. Ultimately, the purpose of calculating the population size is to estimate how many edible orchids contribute to the biodiversity economy. With the following assumptions: (i) the species are evenly distributed, (ii) one orchid plant produces only one tuber annually, and (iii) all the tubers per plant are harvested during one growing season, we estimate that based on the data collected in this study, 1.9 million tubers would be harvested annually, at the bare minimum (Davenport and Ndangalasi 2003). This figure does not account for the multiple species growing at varying life cycle stages. The number is highly likely, more.
Conclusion
This study provided current information on the association between distance from the water source and the diversity, frequency, and population density of chikanda orchids residing within a Miombo wetland ecosystem. Firstly, there was a statistically significant difference in the species diversity in all three communities with a general decreasing trend in all three sampled orchid community profiles. Secondly, there was a significant effect of distance from the water source on the relative frequency of the orchid taxa present in at least two of the three sampled orchid communities. Thirdly, there was a significant effect of distance from the water source on the population density of orchid taxa in at least two of the three sampled orchid communities. Finally, the population sizes were estimated for the sampled edible orchid species. These results suggest a dynamic relationship between moisture and chikanda orchid taxa occurring in wetland ecosystems. Consequently, moisture cannot be used as an indicator of the hydrologic status of the wetland area without other factors being considered. More importantly, this is the first reported study providing a point-in-time estimate of chikanda orchid population densities from a wetland ecosystem in Zambia. This information will be useful in providing some baseline data on orchid species abundance with the goal to effect the sustainable monitoring and conservation of chikanda orchids.
For future similar research, we recommend the use of remote sensing to estimate population densities or the use of drones to access wetland ecosystems even during the rainy seasons of the year. The use of drones would assist in the capturing of images that can then be used to estimate population densities. Additionally, models are a useful tool to estimate and predict population density, diversity and other important parameters that help monitor the conservation of species. We recommend the use of survival analysis on data collected over extended periods of time as a useful monitoring tool to track the changes over time. Survival analysis uses statistical analysis to estimate the expected duration of time until an event occurs, for instance orchid mortality. Although there are reports of a decrease in the number of edible orchids in Zambia, there is much evidence-based research needed to assess the magnitude of the perceived loss and measure the current edible orchid distributions, compositions, and populations.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Assédé ESP, Djagoun CAMS, Azihou FA, Gogan YSC, Kouton MD, Adomou AC, Geldenhuys CJ, Chirwa PW, Sinsin B (2018) Efficiency of conservation areas to protect orchid species in Benin, West Africa. South Afr J Bot 116:230–237. https://doi.org/10.1016/j.sajb.2018.02.405
Bakayoko A, Ouattara ND, Yao AC, Malan DF, Neuba DF-R, Tra BFH, Kouakou TH (2022) Diversity of Orchids from Continental Sub-saharan Africa. In: Mérillon J-M, Kodja H (eds) Orchids Phytochemistry, Biology and Horticulture: fundamentals and Applications. Springer International Publishing, Cham, pp 135–152. https://doi.org/10.1007/978-3-030-38392-3_38
Barker A, Henry L, Hargreaves S (2018) Satyrium trinerve. The IUCN Red list of threatened species 2018: e.T87635507A87635512. International Union for Conservation of Nature and Natural Resources
Bone R (2017) Edible wild orchid trade: sustaining livelihoods and biodiversity in Zambia. Darwin Initiative Main Project
Challe JFX, Price LL (2009) Endangered edible orchids and vulnerable gatherers in the context of HIV/AIDS in the Southern Highlands of Tanzania. J Ethnobiol Ethnomedicine 5:1–11. https://doi.org/10.1186/1746-4269-5-41
Challe JFX, Struik PC (2008) The impact on orchid species abundance of gathering their edible tubers by HIV/AIDS orphans: a case of three villages in the Southern Highlands of Tanzania. NJAS - Wagening J Life Sci 56:261–279. https://doi.org/10.1016/S1573-5214(08)80011-5
Challe JFX, Struik PC, Price LL (2018) Perspectives of children orphaned by HIV/AIDS on ecology and gathering of wild orchids in Tanzania. J Ethnobiol 38:223–243. https://doi.org/10.2993/0278-0771-38.2.223
Chapagain DJ, Meilby H, Baniya CB, Budha-Magar S, Ghimire SK (2021) Illegal harvesting and livestock grazing threaten the endangered orchid Dactylorhiza Hatagirea (D. Don) Soó in Nepalese Himalaya. Ecol Evol 11:6672–6687. https://doi.org/10.1002/ece3.7520
Charitonidou M, Stara K, Kougioumoutzis K, Halley JM (2019) Implications of salep collection for the conservation of the elder-flowered orchid (Dactylorhiza Sambucina) in Epirus. Greece J Biol Res Greece 26. https://doi.org/10.1186/s40709-019-0110-1
Chidumayo EN (1992) The utilisation and status of dambos in Southern Africa: a Zambian case study, in: Matiza, T., H.N., C. (Eds.), Wetlands Conservation Conference for Southern Africa: Proceedings of the SADCC Wetlands Conference. IUCN, Gaborone, Botswana, pp. 105–108
Christenhusz MJM, Byng JW (2016) The number of known plants species in the world and its annual increase. Phytotaxa 261:201. https://doi.org/10.11646/phytotaxa.261.3.1
Croix IFL, Croix TML (1983) Orchids of Bvumbwe Soc Malawi J 36:12–18. https://www.jstor.org/stable/29778499
Davenport TRB, Ndangalasi HJ (2003) An escalating trade in orchid tubers across Tanzania’s Southern highlands: assessment, dynamics and conservation implications. ORYX 37:55–61. https://doi.org/10.1017/S0030605303000127
Djordjević V, Aćić S, Kabaš E, Lazarević P, Tsiftsis S, Lakušić D (2022) The orchids of Wetland Vegetation in the Central Balkans. Diversity 15:26. https://doi.org/10.3390/d15010026
Dunn OJ (1964) Multiple comparisons using Rank sums. Technometrics 6:241–252. https://doi.org/10.4324/9781315797564-20
Emeterio-Lara A, García-Franco JG, Hernández-Apolinar M, Toledo-Hernández VH, Valencia-Díaz S, Flores-Palacios A (2021) Is pseudobulb harvest a sustainable management strategy in wild orchid populations? An experiment with Laelia Autumnalis. For Ecol Manag 491:119205. https://doi.org/10.1016/j.foreco.2021.119205
Frost P (1996) The ecology of miombo woodlands. The Miombo in Transition: Woodlands and Welfare in Africa. Centre for International Forestry Research, Bogor, Indonesia, pp 11–58
Hamisy WC (2007) Development of conservation strategies for the Wild Edible Orchid in Tanzania. Tropical Pesticides Research Institute (TPRI), National Plant Genetic Resources Centre, Arusha, Tanzania
Hinsley A, De Boer HJ, Fay MF, Gale SW, Gardiner LM, Gunasekara RS, Kumar P, Masters S, Metusala D, Roberts DL, Veldman S, Wong S, Phelps J (2018) A review of the trade in orchids and its implications for conservation. Bot J Linn Soc 186:435–455. https://doi.org/10.1093/botlinnean/box083
Jasinge NU, Huynh T, Lawrie AC (2018) Changes in orchid populations and endophytic fungi with rainfall and prescribed burning in Pterostylis revoluta in Victoria, Australia. Ann Bot 121:321–334. https://doi.org/10.1093/aob/mcx164
Kotze DC, Wood AP (2021) Assessing the Long-Term Ecological Sustainability of Dambo Cultivation in Southern Africa: Ten-Year Case Studies from Zambia and Malawi. Wetlands 41. https://doi.org/10.1007/s13157-021-01399-5
Kruskal WH, Wallis WA (1952) Use of ranks in One-Criterion Variance Analysis. J Am Stat Assoc 47:583–621. https://doi.org/10.1080/01621459.1952.10483441
Machaka-Houri N, Al-Zein MS, Westbury DB, Talhouk SN (2012) Reproductive success of the rare endemic Orchis galilaea (Orchidaceae) in Lebanon. Turk J Bot 36:677–682. https://doi.org/10.3906/bot-1104-4
Mäckel R (1974) Dambos: a study in morphodynamic activity on the plateau regions of Zambia. Catena 1:327–365. https://doi.org/10.1016/S0341-8162(73)80018-9
Malaisse F, Parent G (1985) Edible wild vegetable products in the Zambezian Woodland area: a nutritional and ecological approach. Ecol Food Nutr 18:48–82. https://doi.org/10.1080/03670244.1985.9990913
Mann HB, Whitney DR (1947) On a test of whether one of two Random variables is stochastically larger than the other. Ann Math Stat 18:50–60. https://doi.org/10.1214/aoms/1177730491
Mccormick MK, Jacquemyn H (2014) What constrains the distribution of orchid populations? New Phytol 202:392–400. https://doi.org/10.1111/nph.12639
Musasa T, Marambanyika T (2020) Threats to sustainable utilization of wetland resources in Zimbabwe: a review. Wetl Ecol Manag 28:681–696. https://doi.org/10.1007/s11273-020-09732-1
Namoto M (2018) A survey report on edible orchid distributions, abundance and tuber morphological variations: Nyika National Park, Malawi. Forestry Research Institute of Malawi and Mzuzu University
Nyamadzawo G, Wuta M, Nyamangara J, Nyamugafata P, Tendayi T (2014) Burning, biomass removal and tillage effects on soil organic carbon and nutrients in seasonal wetlands (Dambos) of chiota smallholder farming area, Zimbabwe. Arch Agron Soil Sci 60:1411–1427. https://doi.org/10.1080/03650340.2014.892583
Peter C, Hankey A, Wodrich K, Mincher B, Venter N (2017) Last chance to see? The race to see the spectacular Albertina Sisulu orchid, Brachycorythis conica subsp. transvaalensis, a critically endangered South African terrestrial orchid., in: 22nd World Orchid Conference. San Diego County Orchid Society, Guayaquil. Ecuador. https://www.researchgate.net/publication/336798720_Peter_C_Hankey_A_Wodrich_K_Mincher_B_Venter_N_2017_LAST_CHANCE_TO_SEE_The_race_to_see_the_spectacular_Albertina_Sisulu_Orchid_Brachycorythis_conica_subsp_transvaalensis_a_critically_endangered_South_A
Pielou EC (1966) The measurement of diversity in different types of biological collections. J Theor Biol 13:131–144. https://doi.org/10.1016/0022-5193(66)90013-0
Rasmussen HN, Dixon KW, Jersáková J, Těšitelová T (2015) Germination and seedling establishment in orchids: a complex of requirements. Ann Bot 116:391–402. https://doi.org/10.1093/aob/mcv087
Roberts N (1988) Dambos in Development: management of a Fragile Ecological Resource. J Biogeogr 15:141–148. https://doi.org/10.2307/2845054
Swarts ND, Dixon KW (2009) Terrestrial orchid conservation in the age of extinction. Ann Bot 104:543–556. https://doi.org/10.1093/aob/mcp025
Taylor ARD, Howard GW, Begg GW (1995) Developing Wetland inventories in Southern Africa: a review. Vegetation 118:57–79. https://www.jstor.org/stable/20046595
Ticktin T (ed) (2015) The ecological sustainability of non-timber forest product harvest. Ecological sustainability for Non-timber Forest products. Routledge, p 22. https://doi.org/10.4324/9781315851587
Ticktin T, Charitonidou M, Douglas J, Halley JM, Hernández-Apolinar M, Liu H, Mondragón D, Pérez-García EA, Tremblay RL, Phelps J (2023) Wild orchids: a framework for identifying and improving sustainable harvest. Biol Conserv 277. https://doi.org/10.1016/j.biocon.2022.109816
Timberlake J (1998) Biodiversity of the Zambezi Basin Wetlands: review of preliminary assessment of avialable information, ZAMSOC/BFA. The Zambezi Society and The Biodiversity Foundation for Africa, Harare, Zimbabwe
Tomczak M, Tomczak E (2014) The need to report effect size estimates revisited. An overview of some recommended measures of effect size. Trends Sport Sci 1:19–25. http://tss.awf.poznan.pl/files/3_Trends_Vol21_2014__no1_20.pdf
Tsheboeng G (2018) Spatial variation of the influence of distance from surface water on riparian plant communities in the Okavango Delta. Botsw Ecol Process 7. https://doi.org/10.1186/s13717-018-0140-x
Tsheboeng G, Murray-Hudson M, Kashe K (2017) Response of riparian plant communities to distance from surface water in the Okavango Delta. Botsw Afr J Ecol 55:402–410. https://doi.org/10.1111/aje.12364
Veldman S, Gravendeel B, Otieno JN, Lammers Y, Duijm E, Nieman A, Bytebier B, Ngugi G, Martos F, van Andel TR, de Boer HJ (2017) High-throughput sequencing of African chikanda cake highlights conservation challenges in orchids. Biodivers Conserv 26:2029–2046. https://doi.org/10.1007/s10531-017-1343-7
Veldman S, Kim SJ, van Andel TR, Font MB, Bone RE, Bytebier B, Chuba D, Gravendeel B, Martos F, Mpatwa G, Ngugi G, Vinya R, Wightman N, Yokoya K, de Boer HJ (2018) Trade in Zambian edible orchids—DNA barcoding reveals the use of unexpected orchid taxa for chikanda. Genes 9 https://doi.org/10.3390/genes9120595
von der Heyden CJ (2004) The hydrology and hydrogeology of dambos: a review. Prog Phys Geogr 28:544–564. https://doi.org/10.1191/0309133304pp424oa
Weather Atlas (2022) Climate and monthly weather forecast Mwinilunga, Zambia [WWW Document]. Weather Atlas. https://www.weather-atlas.com/en/zambia/mwinilunga-climate (accessed 7.27.22)
Williamson G (1977) The orchids of South Central Africa, the orchids of South Central Africa. Dent, London
Acknowledgements
All authors acknowledge Mr. Geophat Mpatwa, Ms. Mokwani Kaluwe and Ba Justin for the tremendous effort and support during fieldwork.
Funding
This work was supported by the Postgraduate Scholarship Programme from Stellenbosch University to CT, and ZaHBRI (Zambezi Horticultural and Botanical Research Institute) that supported fieldwork transportation costs.
Open access funding provided by Stellenbosch University.
Author information
Authors and Affiliations
Contributions
CT and EEP contributed to the study conception and design. Material preparation, data collection and analysis were performed by CT. EEP, SS, RRM, KM, and TD supervised various aspects of the research. The first draft of the manuscript was written by CT and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Communicated by David Hawksworth.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tembo, C., Phiri, E.E., Syampungani, S. et al. A point-in-time inventory of chikanda orchids within a wild harvesting wetland area in Mwinilunga, Zambia: implications for conservation. Biodivers Conserv 33, 705–724 (2024). https://doi.org/10.1007/s10531-023-02768-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-023-02768-8