Abstract
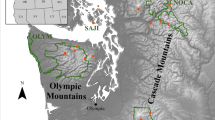
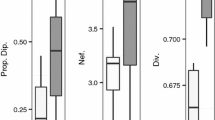
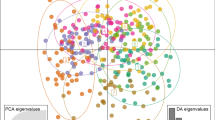
Maintenance of phenotypic and genotypic diversity within and across species and populations is critical for their capacity to survive and adapt to changing environments. Climate change potentially puts cryptic diversity and populations at increased risk, highlighting the importance of quantifying and understanding this diversity before it is lost. This study focuses on Bombus lapponicus sylvicola, a bumble bee species that has undergone recent taxonomic additions and revisions. We tested the null hypotheses that B. l. sylvicola over a 40,000 km2 geographic range and climatic gradient in the Canadian Rocky Mountains represented a single genetic population. Furthermore, we evaluated predictions for mechanisms behind genomic divergence among groups under this framework. We sampled bumble bees from 69 sites and used DNA from two different species (131 B. l. sylvicola and 435 individuals of the closely related B. melanopygus as an outgroup) to characterize 20,000 SNPs and measure relatedness and gene flow. We collected phenotypic data on color patterns and mapped population distribution based on environmental variables. We found evidence of two phenotypically and genetically distinct parapatric populations of B. l. sylvicola that appear to have diverged under conditions of gene flow and differential recombination. Our models suggest that these populations occupy distinct climatic regions, with a newly described cryptic population found in locations reaching a lower minimum temperature. This research presents evidence for the role of adaptative evolution in response to different climate conditions.







Similar content being viewed by others
Data availability
Color phenotype data and genetic data are available on Dryad.
References
Andrews S (2010) FastQC: a quality control tool for high throughput sequence data. https://www.bioinformatics.babraham.ac.uk/projects/fastqc/
Bellard C, Bertelsmeier C, Leadley P, Thuiller W, Courchamp F (2012) Impacts of climate change on the future of biodiversity. Ecol Lett 15:365–377
Bickford D, Lohman DJ, Sodhi NS, Ng PKL, Meier R, Winker K, Ingram KK, Das I (2007) Cryptic species as a window on diversity and conservation. Trends Ecol Evol 22:148–155
Bolger AM, Lohse M, Usadel B (2014) Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30:2114–2120. https://doi.org/10.1093/BIOINFORMATICS/BTU170
Burri R, Nater A, Kawakami T, Mugal CF, Olason PI, Smeds L, Suh A, Dutoit L, Bureš S, Garamszegi LZ et al (2015) Linked selection and recombination rate variation drive the evolution of the genomic landscape of differentiation across the speciation continuum of Ficedula flycatchers. Genome Res 25:1656–1665. https://doi.org/10.1101/GR.196485.115
Butlin RK (2005) Recombination and speciation. Mol Ecol 14:2621–2635. https://doi.org/10.1111/j.1365-294X.2005.02617.x
Butlin RK, Galindo J, Grahame JW (2008) Sympatric, parapatric or allopatric: the most important way to classify speciation? Philos Trans R Soc B Biol Sci 363:2997–3007. https://doi.org/10.1098/rstb.2008.0076
Cameron SA, Hines HM, Williams PH (2007) A comprehensive phylogeny of the bumble bees (Bombus). Biol J Linn Soc 91:161–188. https://doi.org/10.1111/j.1095-8312.2007.00784.x
Capella-Gutiérrez S, Silla-Martínez JM, Gabaldón T (2009) trimAl: a tool for automated alignment trimming in large-scale phylogenetic analyses. Bioinforma Appl 25:1972–1973. https://doi.org/10.1093/bioinformatics/btp348
Catchen J, Hohenlohe PA, Bassham S, Amores A, Cresko WA (2013) Stacks: an analysis tool set for population genomics. Mol Ecol 22:3124–3140. https://doi.org/10.1111/MEC.12354
Chapin FSI, Sala OE, Burke IC, Grime P, Hooper DU, Lauenroth WK, Lombard A, Mooney HA, Mosier AR, Naeem S et al (1998) Ecosystem consequences of changing biodiversity: experimental evidence and a research agenda for the future. Bioscience 48:45–52
Chen S, Tang K, Wang X, Li F, Fu C, Liu Y, Faiz AH, Jiang X, Liu S (2022) Multi-locus phylogeny and species delimitations of the striped-back shrew group (Eulipotyphla: Soricidae): implications for cryptic diversity, taxonomy and multiple speciation patterns. Mol Phylogenet Evol 177:107619. https://doi.org/10.1016/J.YMPEV.2022.107619
Christmas MJ, Jones JC, Olsson A, Wallerman O, Bunikis I, Kierczak M, Peona V, Whitley KM, Larva T, Suh A et al (2021) Genetic barriers to historical gene flow between cryptic species of alpine bumblebees revealed by comparative population genomics. Mol Biol Evol 38:3126–3143. https://doi.org/10.1093/molbev/msab086
Clake DJ, Rogers SM, Galpern P (2022) Landscape complementation is a driver of bumble bee (Bombus sp.) abundance in the Canadian rocky mountains. Landsc Ecol 37:713–728. https://doi.org/10.1007/s10980-021-01389-2
Cortés AJ, López-Hernández F (2021) Harnessing crop wild diversity for climate change adaptation. Genes 12:783. https://doi.org/10.3390/genes12050783
Dray S, Dufour AB (2007) The ade4 package: implementing the duality diagram for ecologists. J Stat Softw 22:1–20. https://doi.org/10.18637/jss.v022.i04
Edgar RC (2004) MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 32:1792–1797. https://doi.org/10.1093/NAR/GKH340
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol 14:2611–2620. https://doi.org/10.1111/j.1365-294X.2005.02553.x
Feder JL, Egan SP, Nosil P (2012) The genomics of speciation-with-gene-flow. Trends Genet 28:342–350. https://doi.org/10.1016/J.TIG.2012.03.009
Fick SE, Hijmans RJ (2017) WorldClim 2: new 1-km spatial resolution climate surfaces for global land areas. Int J Climatol 37:4302–4315. https://doi.org/10.1002/joc.5086
Frichot E, François O (2015) LEA: an R package for landscape and ecological association studies. Methods Ecol Evol. https://doi.org/10.1111/2041-210X.12382
Garcia RA, Cabeza M, Rahbek C, Araújo MB (2014) Multiple dimensions of climate change and their implications for biodiversity. Science 344:1247579
Geraldes A, Basset P, Smith KL, Nachman MW (2011) Higher differentiation among subspecies of the house mouse (Mus musculus) in genomic regions with low recombination. Mol Ecol 20:4722–4736. https://doi.org/10.1111/J.1365-294X.2011.05285.X
Gernhard T (2008) The conditioned reconstructed process. J Theor Biol 253:769–778. https://doi.org/10.1016/j.jtbi.2008.04.005
Ghisbain G, Lozier JD, Rahman SR, Ezray BD, Tian L, Ulmer JM, Heraghty SD, Strange JP, Rasmont P, Hines HM (2020) Substantial genetic divergence and lack of recent gene flow support cryptic speciation in a colour polymorphic bumble bee (Bombus bifarius) species complex. Syst Entomol 45:635–652. https://doi.org/10.1111/SYEN.12419
Guisan A, Broennimann O, Buri A, Cianfrani C, D’Amen M, Di Cola V, Fernandes R, Gray SM, Mateo RG, Pinto E et al (2019) Climate change impacts on mountain biodiversity. In: Hannah L, Wilson EO, Lovejoy TE (eds) Biodiversity and climate change transforming the biosphere. Yale University Press, pp 221–236
Hijmans RJ (2022) Raster: geographic data analysis and modeling. R package version 3.5–15. https://CRAN.R-project.org/package=raster
Jackson JM, Pimsler ML, Oyen KJ, Koch-Uhuad JB, Herndon JD, Strange JP, Dillon ME, Lozier JD (2018) Distance, elevation and environment as drivers of diversity and divergence in bumble bees across latitude and altitude. Mol Ecol 27:2926–2942. https://doi.org/10.1111/mec.14735
Johnson KH, Vogt KA, Clark HJ, Schmitz OJ, Vogt DJ (1996) Resilience and stability of ecosystems. Trends Ecol Evol 11:372–377
Jombart T (2008) Adegenet: a R package for the multivariate analysis of genetic markers. Bioinformatics 24:1403–1405. https://doi.org/10.1093/bioinformatics/btn129
Jombart T, Ahmed I (2011) Adegenet 1.3-1: new tools for the analysis of genome-wide SNP data. Bioinformatics 27:3070–3071. https://doi.org/10.1093/bioinformatics/btr521
Knaus BJ, Grünwald NJ (2017) vcfr: a package to manipulate and visualize variant call format data in R. Mol Ecol Resour 17:44–53. https://doi.org/10.1111/1755-0998.12549
Li H (2011) A statistical framework for SNP calling, mutation discovery, association mapping and population genetical parameter estimation from sequencing data. Bioinformatics 27:2987–2993. https://doi.org/10.1093/bioinformatics/btr509
Li H, Durbin R (2009) Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25:1754–1760. https://doi.org/10.1093/BIOINFORMATICS/BTP324
Li H, Handsaker B, Wysoker A, Fennell T, Ruan J, Homer N, Marth G, Abecasis G, Durbin R (2009) The sequence alignment/map format and SAMtools. Bioinformatics 25:2078–2079. https://doi.org/10.1093/bioinformatics/btp352
Loreau M (2000) Biodiversity and ecosystem functioning: recent theoretical advances. Oikos 91:3–17. https://doi.org/10.1034/j.1600-0706.2000.910101.x
Manichaikul A, Mychaleckyj JC, Rich SS, Daly K, Sale M, Chen W (2010) Robust relationship inference in genome-wide association studies. Bioinformatics 26:2867–2873. https://doi.org/10.1093/bioinformatics/btq559
Marske KA, Rahbek C, Nogués-Bravo D (2013) Phylogeography: spanning the ecology-evolution continuum. Ecography 36:1169–1181. https://doi.org/10.1111/j.1600-0587.2013.00244.x
Martinet B, Lecocq T, Brasero N, Gerard M, Urbanova K, Valterova I, Gjershaug JO, Michez D, Rasmont P (2019) Integrative taxonomy of an arctic bumblebee species complex highlights a new cryptic species (Apidae: Bombus). Zool J Linn Soc 187:599–621. https://doi.org/10.1093/zoolinnean/zlz041
Nosil P (2012) The speciation continuum: what factors affect how far speciation proceeds? Ecological speciation. Oxford University Press, pp 192–213
Nosil P, Feder JL (2012) Genomic divergence during speciation: causes and consequences. Philos Trans R Soc B Biol Sci 367:332–342. https://doi.org/10.1098/rstb.2011.0263
Obura DO, Declerck F, Verburg PH, Gupta J, Abrams JF, Bai X, Bunn S, Ebi KL, Gifford L, Gordon C et al (2022) Achieving a nature- and people-positive future. One Earth. https://doi.org/10.1016/j.oneear.2022.11.013
Oliver TH, Isaac NJB, August TA, Woodcock BA, Roy DB, Bullock JM (2015) Declining resilience of ecosystem functions under biodiversity loss. Nat Commun. https://doi.org/10.1038/ncomms10122
Ollerton J (2017) Pollinator diversity: distribution, ecological function and conservation. Annu Rev Ecol Evol Syst 48:353–376. https://doi.org/10.1146/annurev-ecolsys-110316-022919
Owen RE, Whidden TL, Plowright RC (2010) Genetic and morphometric evidence for the conspecific status of the bumble bees, Bombus melanopygus and Bombus edwardsii. J Insect Sci 10:1–18. https://doi.org/10.1673/031.010.10901
Paradis E, Schliep K (2019) ape 5.0: an environment for modern phylogenetics and evolutionary analyses in R. Bioinformatics 35:526–528. https://doi.org/10.1093/bioinformatics/bty633
Pearman PB, D’Amen M, Graham CH, Thuiller W, Zimmermann NE (2010) Within-taxon niche structure: Niche conservatism, divergence and predicted effects of climate change. Ecography 33:990–1003. https://doi.org/10.1111/J.1600-0587.2010.06443.X
Pimsler ML, Oyen KJ, Herndon JD, Jackson JM, Strange P, Dillon ME, Lozier LD (2020) Biogeographic parallels in thermal tolerance and gene expression variation under temperature stress in a widespread bumble bee. Sci Reports 101(10):1–11. https://doi.org/10.1038/S41598-020-73391-8
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959. https://doi.org/10.1093/GENETICS/155.2.945
R Core Team (2023) R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Riddle BR (2019) Genetic signatures of historical and contemporary responses to climate change. In: Hannah L, Wilson EO, Lovejoy TE (eds) Biodiversity and climate change: transforming the biosphere. Yale University Press, pp 66–76
Sadd BM, Barribeau SM, Bloch G, de Graaf DC, Dearden P, Elsik CG, Gadau J, Grimmelikhuijzen CJP, Hasselmann M, Lozier JD et al (2015) The genomes of two key bumblebee species with primitive eusocial organization. Genome Biol 16:1–31. https://doi.org/10.1186/s13059-015-0623-3
Schluter D, Pennell MW (2017) Speciation gradients and the distribution of biodiversity. Nature 546:48–55. https://doi.org/10.1038/NATURE22897
Schön I, Pieri V, Sherbakov DY, Martens K (2017) Cryptic diversity and speciation in endemic Cytherissa (Ostracoda, Crustacea) from Lake Baikal. Hydrobiologia 800:61–79. https://doi.org/10.1007/S10750-017-3259-3/FIGURES/3
Seehausen O, Butlin RK, Keller I, Wagner CE, Boughman JW, Hohenlohe PA, Peichel CL, Saetre GP et al (2014) Genomics and the origin of species. Nat Rev Genet 15:176–192. https://doi.org/10.1038/nrg3644
Sheffield CS, Oram R, Heron JM (2020) Bombus (Pyrobombus) johanseni Sladen, 1919, a valid North American bumble bee species, with a new synonymy and comparisons to other “red-banded” bumble bee species in North America (Hymenoptera, Apidae, Bombini). Zookeys 2020:59–81. https://doi.org/10.3897/zookeys.984.55816
Sladen FWL (1919) The wasps and bees collected by the Canadian Arctic Expedition, 1913–1918. Report of the Canadian Arctic Expedition 1913–18 3: 25–35
Sousa V, Hey J (2013) Understanding the origin of species with genome-scale data: modelling gene flow. Nat Rev Genet 14:404–414. https://doi.org/10.1038/nrg3446
Suchard MA, Lemey P, Baele G, Ayres DL, Drummond AJ, Rambaut A (2018) Bayesian phylogenetic and phylodynamic data integration using BEAST 1.10. Virus Evol 4:vey016. https://doi.org/10.1093/ve/vey016
Taylor SA, Curry RL, White TA, Ferretti V, Lovette I (2014) Spatiotemporally consistent genomic signatures of reproductive isolation in a moving hybrid zone. Evolution 68:3066–3081. https://doi.org/10.1111/EVO.12510
Tilman D, Reich PB, Knops JMH (2006) Biodiversity and ecosystem stability in a decade-long grassland experiment. Nature 441:629–632. https://doi.org/10.1038/nature04742
Trontelj P, Fišer C (2009) Cryptic species diversity should not be trivialised. Syst Biodivers 7:1–3. https://doi.org/10.1017/S1477200008002909
Uemura R, Motoyama H, Masson-Delmotte V, Jouzel J, Kawamura K, Goto-Azuma K, Fujita S, Kuramoto T, Hirabayashi M, Miyake T et al (2018) Asynchrony between Antarctic temperature and CO2 associated with obliquity over the past 720,000 years. Nat Commun 91(9):1–11. https://doi.org/10.1038/s41467-018-03328-3
Ungerer MC, Johnson LC, Herman MA (2008) Ecological genomics: understanding gene and genome function in the natural environment. Heredity 100:178–183
Venables WN, Ripley BD (2002) Modern applied statistics with S, 4th edn. Springer, New York
Vimeux F, Cuffey KM, Jouzel J (2002) New insights into southern Hemisphere temperature changes from Vostok ice cores using deuterium excess correction. Earth Planet Sci Lett 203:829–843. https://doi.org/10.1016/S0012-821X(02)00950-0
Vodă R, Dapporto L, Dincă V, Vila R (2015) Cryptic matters: overlooked species generate most butterfly beta-diversity. Ecography 38:405–409. https://doi.org/10.1111/ECOG.00762
Weir BS, Cockerham CC (1984) Estimating F-statistics for the analysis of population structure. Evolution (NY) 38:1358–1370. https://doi.org/10.2307/2408641
Williams P, Thorp R, Richardson L, Colla S (2014) Bumble bees of North America. Princeton University Press
Wolf JBW, Ellegren H (2017) Making sense of genomic islands of differentiation in light of speciation. Nat Rev Genet 18:87–100. https://doi.org/10.1038/nrg.2016.133
Yule GU (1925) A mathematicaltheory of evolution, based on the conclusions of Dr. J. C. Willis, F. R. S. Phil Trans R Soc Lond B 213:21–87. https://doi.org/10.1098/rstb.1925.0002
Zhou Z, Guo H, Han L, Chai J, Che X, Shi F (2019) Singleton molecular species delimitation based on COI-5P barcode sequences revealed high cryptic/undescribed diversity for Chinese katydids (Orthoptera: Tettigoniidae). BMC Evol Biol 19:1–19. https://doi.org/10.1186/s12862-019-1404-5
Acknowledgements
The authors would like to thank Hailey Bloom, Jessy Bokvist, David Clake, Brenna Stanford, and Luke Storey for helping to carry out the field work in this study, and Parks Canada for their support in facilitating field sampling. Thanks to Hailey Bloom, Emma Dunlop, and Michael Gavin for assisting with species identification, and to Rebecca Innes for helping to collect phenotypic data. This research occurred on the traditional lands of the Siksikaitsitapi (Blackfoot Confederacy) Siksika, Kainai and Piikani First Nations; the Iyarhe (Stoney) Nakoda First Nation; the Tsuut’ina First Nation, the Métis Nation of Alberta; and the Ktunaxa First Nations.
Funding
This work was funded by Natural Sciences and Engineering Research Council (NSERC) Discovery Grants to Paul Galpern and Sean Rogers, and an Alberta Conservation Association (ACA) Grant in Biodiversity to Danielle Clake. We are grateful to the Digital Research Alliance of Canada for supplying computing resources.
Author information
Authors and Affiliations
Contributions
All authors contributed to research design. DC performed data collection, data analysis and wrote the original draft of the manuscript. PG and SR provided resources, supervision and review and editing of writing.
Corresponding author
Ethics declarations
Competing interests
The authors have no relevant competing interests to disclose.
Additional information
Communicated by Nigel Stork.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Clake, D.J., Rogers, S.M. & Galpern, P. Cryptic genotypic and phenotypic diversity in parapatric bumble bee populations associated with minimum cold temperatures. Biodivers Conserv 33, 485–507 (2024). https://doi.org/10.1007/s10531-023-02753-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-023-02753-1