Abstract
Landscape fragmentation impacts freshwater habitats and their quality, affecting aquatic insect assemblages. Adjacent terrestrial areas are important secondary habitats where amphibiotic insects mature, feed, find mates, and move to locate aquatic breeding habitats. Using a factorial design with 27 small and large ponds within small and large natural patches in an exotic tree plantation-fragmented landscape of South Africa, and odonates as model organisms, we investigated (1) how pond size/natural terrestrial patch size interaction affects odonate diversity patterns versus habitat quality variables, and (2) determined whether anisopterans and zygopterans respond differently to landscape fragmentation. Species richness was similar among ponds. However, odonate abundance was highest in large ponds regardless of natural terrestrial patch size. Zygopteran functional richness and diversity was driven by pond and natural patch size, suggesting that zygopterans are sensitive to landscape fragmentation. In contrast, anisopterans were more resilient to fragmentation and more likely to select suitable habitats following water chemistry and vegetation characteristics. Overall, large ponds were occupied by different odonate assemblages compared to small ponds, and occupancy was strongly associated with mobility traits. These findings emphasize that ponds in both small and large natural terrestrial patches have conservation value. A pondscape that represents various pond sizes is important for maintaining regional odonate diversity. Pond conservation needs to be considered in the wider terrestrial context, which host a range of important secondary habitats. Adjacent natural terrestrial habitats also connect nearby aquatic habitats, which enable insects to move across the landscape in response to natural and artificial drivers.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Transformation and fragmentation of natural landscapes, along with climate change, have among the most severe impacts on freshwater habitats (Maynou et al. 2017; Riad et al. 2020). Historic structure and quality of aquatic habitats are important for maintaining freshwater diversity (Nagy et al. 2019) and habitat loss and fragmentation lead to freshwater diversity decline and population loss (Cardoso et al. 2020; Lima et al. 2022). Habitat quality decreases when aquatic and marginal vegetation structure and water physiochemical properties are altered (Heino 2009; Hill et al. 2019). Landscape transformation also often leaves only narrow natural vegetation buffer areas around ponds, which interferes with habitat colonization dynamics of species that occupy these aquatic habitats (Harabiš 2017). These changes adversely affect both amphibiotic and aquatic insects (Thorp and Rogers 2014).
Odonata (suborders Anisoptera and Zygoptera) are amphibiotic insects, and aquatic habitat quality directly influences odonate occupancy (e.g., Kietzka et al. 2017; Briggs et al. 2019a), with breeding being limited to high quality freshwater habitats in most species. Yet, they cross the ecosystem boundary from aquatic to terrestrial as they mature (Knight et al. 2005). Odonate occupancy in terrestrial areas also increases during the dry season when surface water resources become scarce (Harabiš 2017; Harabis and Dolny 2011). Being highly mobile as adults, most odonate species occupy wide terrestrial areas surrounding freshwater habitats (Kalkman et al. 2007), with anisopterans overall having larger overall habitat ranges than zygopterans (Conrad et al. 1999).
Co-occurring species variably rely on secondary terrestrial areas that facilitate daily movements (i.e., frequent movement between water and the adjacent terrestrial matrix), and seasonal movements (i.e., movement between suitable habitats in a terrestrial matrix) (Raebel et al. 2012). Odonate movement in the longer-term is also determined by water permanency, with species that occupy unpredictable seasonal ponds and wetlands relying more on quality terrestrial spaces compared to species which occupy more permanent habitats such as lakes and rivers (Hof et al. 2006; Deacon et al. 2020).
An odonate’s ability to move among ponds and establish new populations is not exclusively dependent on their movement ability. It is also partly driven by their phylogenetic and behavioural limitations (McCauley 2006; Outomuro and Johansson 2019). Furthermore, successfully reaching previously unoccupied habitats does not necessarily translate into successful establishment, as the new habitat must also support the ecological and behavioral needs of odonates. For instance, patrolling odonates more regularly move between aquatic and terrestrial habitats as opposed to those species that spend most of their time perched close to the water’s edge (Bried and Ervin 2006). Patrolling odonate species with wide territories thus need larger areas to establish breeding populations. This means that functional connectivity among suitable habitats relies on combinations between flight performance, flight behaviour, competitor/mate density, and habitat preferences in odonates (Chin and Taylor 2009; French and McCauley 2019).
Given the great variation in habitat preference, mobility and behavioral traits among the two odonate suborders, as well as among the species within them, odonates show promise as indicators of both freshwater habitat and terrestrial matrix quality (Sahlén 2006; Nagy et al. 2019). Furthermore, it is recognized that the ‘pondscape’ (i.e., landscape configuration, spatial abundance of ponds, and connectivity among them) may be as important as hydrography, vegetation characteristics, or water chemistry in maintaining historic population levels and assemblage diversity (Kadoya et al. 2008; Hassall et al. 2016).
Here, we focus on odonates that occupy permanent and seasonal fishless ponds in the coastal sub-tropical region of KwaZulu-Natal, South Africa, where much of the landscape is transformed and fragmented by plantation forestry. Our overall aim was to determine the importance of the pondscape (i.e., the ponds relative to the terrestrial context) in driving odonate species richness, abundance, assemblage composition and functional diversity patterns in a fragmented landscape. Specifically, we (1) determined whether pond size/natural terrestrial patch size interaction affects overall odonate species richness, abundance, assemblage composition, and functional diversity relative to pond characteristics, and (2) determined whether anisopterans and zygopterans respond differently to landscape context. Although local pond characteristics are important for odonates (Briggs et al. 2019a), we hypothesized that local pond characteristics and size of natural terrestrial patches in which ponds are located share equal weight in determining odonate occupancy in the transformed landscape investigated here, with large ponds in large natural patches expected to have the highest number of species. Due to the difference in area requirements between the two suborders of Odonata (Conrad et al. 1999), we also hypothesized that anisopterans predominantly occupy ponds within large natural patches of terrestrial vegetation, while zygopterans preferentially occupy ponds within smaller natural patches.
Methods
Study area
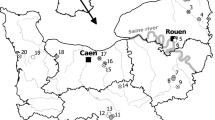
The study was conducted in northeast KwaZulu-Natal, a summer rainfall area falling in the Maputaland-Pondoland-Albany Biodiversity Hotspot (Myers et al. 2000). Natural vegetation is dominated by Maputaland Coastal Belt and Maputaland Wooded Grassland vegetation types (Mucina et al. 2006). Sample ponds were selected on two neighbouring Eucalyptus spp. plantation estates (owned and managed by SiyaQhubeka Forestry) on the eastern portion of the study area, and in iSimangaliso Wetland Park, a World Heritage Site on the western portion of the study area (Fig. 1). Plantation estate ponds occur in networks of large conservation corridors of remnant natural patches among plantation compartments, with varying sizes and degrees of connectivity.
Spatial layout of sampled odonate ponds in northeast KwaZulu-Natal, South Africa. Pale yellow indicates natural terrestrial patches and bright yellow indicates Eucalyptus plantation forestry compartments. Blue indicates small ponds in small patches, green indicates small ponds in large patches, red indicates large ponds in small patches and gray indicates large ponds in large patches
Ponds were fishless and used by large mammals roaming the study area, as the boundary between the plantation estates and the conservation area is unfenced. Ponds in the area are greatly influenced by El Niño Southern Oscillation events in the longer term, and their sizes and permanency are highly variable from one year to the next due to seasonal summer rainfall. These permanent and temporary ponds are obligate habitats for at least 29 Odonata species in the greater iSimangaliso Wetland Park region, and supplementary habitats for roughly 45 species, collectively supporting ~ 86% of all odonates in the region (Hart et al. 2014; Deacon et al. 2021).
Study design
We selected 27 sample ponds ranging in size and landscape context (Fig. 1). Ponds with a surface area smaller than 1999 m2 were classified as a ‘small pond’ while those with a surface area larger than 2000 m2 were classified as a ‘large pond’. As pond margins are highly dynamic in the study area, we considered pond size as the area covered by surface water at the time of sampling. The size of the natural patch of grassland and thicket vegetation in which each pond occurred was classified, and natural patch size was considered as the uninterrupted surface area covered by grassland and thicket vegetation, and excluding those areas covered by plantation trees or indigenous forest, where shading of pond habitats reduce odonate occupancy (Osborn and Samways 1996). Natural patches with a size smaller than 0.39 km2 were classified as a ‘small patch’, while natural patches with a size larger than 0.4 km2 were classified as a ‘large patch’.
Following this classification, eight ponds were classified as ‘small pond in small patch’, six ponds were classified as ‘small pond in large patch’, four ponds were classified as ‘large pond in small patch’, and nine ponds were classified as ‘large pond in large patch’ (Online Resource 1). The connectedness of each pond relative to surrounding terrestrial matrix was also determined. If a pond was totally surrounded by plantations, it was considered ‘disconnected’, but if a pond had shared grassland/thicket with a neighboring pond (i.e., ponds connected through a conservation corridor), it was considered ‘connected’. Spatial information was extracted using QGIS version 3.26 (Quantum GIS Development Team 2022), using a 10 m resolution Sentinel2 spatial dataset, captured on the 9th of February 2022 (Copernicus Sentinel2 Data 2022).
Field sampling for odonates was conducted from the 7th to the 27th of February 2022, during the peak unimodal flight season in the region. Each of the 27 sample ponds was visited in a random order on cloudless, windless days, between 09h30 and 16h00. As many odonate individuals, especially males, remain faithful to their territories (Jooste et al. 2020), each pond was sampled once to avoid double counts of individuals over a limited sampling period. At each sample pond, two observers walked along opposite pond margins for 45 min (regardless of pond size to equalize sampling effort), recording adult odonate species and abundance, considering both males and females. For small ponds where observers were closer than 30 m from one another, each record was vocally announced to ensure that the same individuals was not counted twice. If an individual could not be identified in the field, it was caught with an insect net, and identified ex situ.
Water pH, conductivity (µS/cm), temperature (ºC) and dissolved oxygen concentration (mg/L) were recorded at five randomly selected points along pond margins, using a Hanna HI98129 and a Hanna HI9142 portable water probe. A measure of turbidity was excluded from the study, as ponds in the areas are frequently used by roaming large mammals, and turbidity fluctuates dramatically over short periods. To estimate vegetation characteristics, five quadrats of 4 m2 were randomly selected along pond margins. Quadrats were equally aligned over aquatic and terrestrial space to represent fuzzy pond margins. Within quadrats, we recorded % grasses cover, % reeds cover, % herbaceous cover, and % bare ground. All obtained values relating to pond chemistry and vegetation characteristics were averaged for each study pond.
Data analysis
A sampling site-based species accumulation curve for observed odonate species richness was constructed using the ‘vegan’ package for R version 3.6 to evaluate sampling adequacy (Oksanen et al. 2020; R Core Team 2020). To supplement the species accumulation curve, we calculated the Chao2, Jackknife2 and ICE species estimator metrics using the ‘fossil’ package for R (Vavrek 2011). The species accumulation curve for observed odonate species richness reached a near asymptote and observed species richness neared the values obtained for the three calculated species estimator metrics (Online Resource 2). Consequently, we used raw species richness values as response variable in subsequent univariate analyses.
We calculated functional richness (i.e., the distribution of traits in the biological assemblage) and functional diversity (i.e., the distribution and abundance of species in trait space, simultaneously; represented by Rao’s Quadratic Entropy), using the ‘FD’ package for R (Laliberté and Legendre 2010). Functional richness and diversity were calculated for each study pond, and separately for the overall odonate assemblage, the anisopteran assemblage, and the zygopteran assemblage. In the case of Zygoptera, ten sample ponds were excluded, as no zygopterans were recorded from these ponds. In all cases, a Cailliez correction was applied to calculate each metric, and both functional richness and diversity were standardized to values ranging between 0 and 1. We considered ten traits from four broad categories: (1) habitat preference traits (relative number of biotopes occupied by adults, habitat preference (lentic vs. lentic/lotic), latitudinal range extent, elevation range extent, and Dragonfly Biotic Index score (DBI; Samways and Simaika 2016), (2) mobility traits (average body length, average fore wing length, and wing-to-body length ratio), (3) behavioural traits (categorized as ‘darter’, ‘flutterer’, ‘glider’, ‘hawker’, and ‘percher’), and (4) phenology traits (number of months active as adults (including overwintering). All odonate trait information (except DBI scores) were extracted from a previous continental-scale study (Deacon et al. 2020).
Eleven predictor variables were considered initially to test the relationships between environmental variables and species richness, abundance, functional richness, functional diversity and assemblage composition across all surveyed ponds. These variables were: water temperature, pH, conductivity, dissolved oxygen concentration, vegetation height, % grass cover, % reed cover, % herbaceous cover, % bare ground, pond category (large pond in large patch vs. small pond in large patch vs. large pond in small patch vs. small pond in small patch), and connectivity (binary; connected to nearby ponds vs. disconnected from nearby ponds).
Each response variable was tested for normality, and Mantel tests were performed to test all response variables for spatial autocorrelation. None of the investigated response variables were spatially autocorrelated, and no random spatial terms were considered in the subsequent modelling procedure. For the six normally distributed response variables (Odonata species richness, Anisoptera species richness, Anisoptera functional richness, Odonata functional diversity, Anisoptera functional diversity, and Zygoptera functional diversity), we used linear models (LM) to test the relationships between these variables and the environment. For the six non-normally response variables, we used generalized linear models (GLM) with Poisson distributions (in the case of Odonata species abundance, Anisoptera species abundance, and Zygoptera species richness and abundance), and Gamma distributions (in the case of Odonata functional richness and Zygoptera functional richness) to test for significant effects.
We performed model averaging to test the significant effects of predictor variables on each response variable (Bartoń 2020). Prior to model averaging, multi-collinearity (Variance Inflation Factor, VIF) was determined for all global models, using the ‘car’ package for R (Fox and Weisberg 2019). Only predictor variables with a VIF < 3 were considered during the final modelling procedure to reduce the statistical effects of strong correlations among predictor variables. Connectivity (strongly correlated with pond category; large ponds in large natural patches were mostly connected, while small ponds in small natural patches were mostly disconnected) and dissolved oxygen concentration (strongly correlated with water temperature) were the only variables to consistently have a VIF > 3 and was excluded from the model averaging procedure for all modelling scenarios. All global models built with the predictor variables with a VIF < 3 for each modelling scenario were tested for overdispersion, and candidate models were ranked from lowest to highest Akaike’s Information Criterion (AICc) and ΔAICc values, using the ‘MuMIn’ package for R (Bartoń 2020). Only the subsets of models where ΔAICc < 4 were considered for model averaging, and a predictor was considered as significant when the upper and lower 2.5% confidence intervals did not include 0, and if the relative variable importance under each modelling scenario was > 50% (i.e., the variable contributed significantly to explaining total variance under the modelling scenario; Bartoń 2020).
To determine odonate assemblage composition variation among study ponds, we performed a model-based analysis of multivariate abundance data in ‘mvabund’ for R (Wang et al. 2020). Only variables with a VIF < 3 (i.e., all environmental variables except pond connectivity and dissolved oxygen concentration) were considered for the multivariate component of the study. To supplement model-based analyses of odonate assemblage variation among pond categories, we used fourth corner analysis to analyze the direct relationships between pond category and the set of traits used throughout the study, implemented through ‘mvabund’. Fourth corner analysis was conducted for all odonates combined, supplying the modelling scenario with an abundance matrix, predictor variable matrix (containing only pond categories), and a trait matrix (containing all behavioural, phenological, habitat preference, and mobility traits). To account for species with low abundances, we applied a LASSO penalty to set association coefficients to zero when their statistical effects were small in the fourth corner model. To visually represent associations between traits and predictor variables, we constructed a fourth corner plot using the ‘lattice’ package for R (Sarkar 2008).
Results
Among 27 study ponds, we recorded ten lentic species and 21 lentic/lotic species across 20 Odonata genera (Online Resource 3). Twenty-five species were anisopterans, and six species were zygopterans. Palpopleura lucia was the most abundant species among anisopterans, with 328 individuals recorded, and Ceriagrion glabrum was the most abundant species among zygopterans, with 268 individuals recorded. Lestes plagiatus, Trithemis annulata, T. dorsalis, and Urothemis assignata had the lowest abundances overall, each with one individual recorded among all studied ponds.
Overall species richness increased with decreasing herbaceous cover along pond margins (Table 1). Overall abundance showed an increase with decreased water pH, water temperature, and herbaceous cover, while also showing an increase with increasing overall vegetation cover along pond margins. Pond size and patch size ranked among the top predictor variables, and odonate abundance was highest in large ponds in small patches compared to large ponds in large patches (z = 4.93, p < 0.001), small ponds in large patches (z = -9.46, p < 0.001), and small ponds in small patches (z =-8.91, p < 0.001; Fig. 2a). Large ponds in large patches had higher odonate abundance compared to small ponds in large patches (z = -8.67, p < 0.001) and small ponds in small patches (z = -6.18, p < 0.001). Small ponds in both small and large patches had similar odonate abundances. For odonate functional traits, highest functional richness was associated with increased water pH, while highest functional diversity was associated with decreased water pH levels (Table 2).
For Anisoptera, highest species richness was associated with decreasing herbaceous cover along pond margins (Table 1). Anisopteran abundance showed an increase with decreasing water pH, herbaceous cover and reeds cover. Anisopteran abundance was also highest in large ponds in small patches compared to large ponds in large patches (z = 2.76, p < 0.05), small ponds in large patches (z = -6.85, p < 0.001), and small ponds in small patches (z = -6.51, p < 0.001; Fig. 2b). Large ponds in large patches had higher anisopteran abundance compared to small ponds in large patches (z = -6.54, p < 0.001) and small ponds in small patches (z = -4.67, p < 0.001). Small ponds in small and large patches had equal anisopteran abundances. Although water pH, water temperature, and herbaceous cover along pond margins were important drivers of anisopteran functional richness, none of these predictor variables were significant (Table 2). No predictor variables were identified as important for anisopteran functional diversity.
For Zygoptera, pond category was the only variable selected in species richness models, although the effects were not significant. However, zygopteran abundance increased with decreasing water temperature, as well as with decreasing herbaceous and reeds cover along pond margins. Zygopteran abundance increased with an increase in overall vegetation cover (dominated by grasses) although decreased with increasing overall vegetation height. Zygopteran abundance was also highest in large ponds in small patches compared to large ponds in large patches (z = 2.65, p < 0.05) and small ponds in large patches (z = 5.12, p < 0.001; Fig. 2c). Large ponds in large patches had higher zygopteran abundance compared to small ponds in large patches (z = -4.72, p < 0.001). Small ponds in small patches had similar zygopteran abundance compared to large ponds in small and large patches. Zygopteran functional richness was higher in large ponds in small patches compared to small ponds in large patches (z = 2.54, p < 0.05; Fig. 2d). Small ponds in large patches had lower zygopteran functional richness compared to large ponds in large patches (z = 2.55, p < 0.05) and small ponds in small patches (z = -2.56, p < 0.05). Small ponds in small patches had equal zygopteran functional richness compared to large ponds in large and small patches. Although reed cover along pond margins was important for zygopteran functional richness, this relationship was not significant. Zygopteran functional diversity was lowest in small ponds in large patches compared to small ponds in small patches (z = 2.41, p < 0.05), and large ponds in both large (z = 2.36, p < 0.05) and small patches (z = 2.22, p < 0.05; Fig. 2e).
Out of the eleven pre-selected environmental predictors, changes in odonate assemblage composition across the 27 sample ponds was only driven by pond and patch size interaction (likelihood ratio test value (LRT) = 171.4, p < 0.01). Large ponds in large patches had an overall distinct odonate assemblage compared to small ponds in large patches (LRT = 67.3, p < 0.05) and small ponds in small patches (LRT = 81.4, p < 0.05), but shared assemblages with large ponds in small patches (Fig. 3). Large ponds in small patches shared assemblages with small ponds in large and small patches. The assemblages of small ponds in small patches could also not be distinguished from those of small ponds in large patches.
Fourth corner results indicated that pond category occupancy was significantly related to odonate traits (LRT = 74.78, p < 0.05), and was predominantly driven by mobility traits (Fig. 4). Odonate species with relatively short wings mostly occupied small and large ponds in small patches, while odonate species with long wings occupied small and large ponds in large patches. These trends were reversed for odonate body size, and species with large bodies predominantly occupied small and large ponds in small patches, while small-bodied odonates mostly occupied small and large ponds in large patches. Similarly, odonates with high wing-to-body length ratios (i.e., species with high relative dispersal ability) were associated with small and large ponds in small patches, while species with low wing-to-body length ratios (i.e., species with low relative dispersal ability) were associated with small and large ponds in large patches. Odonate traits related to behaviour, phenology, and habitat preference had overall weak relationships with pond category occupancy.
Fourth corner results indicating associations between pond category and traits of odonates in northeast KwaZulu-Natal, South Africa. Blue indicates negative associations, red indicates positive associations, and clear indicates no associations between traits and pond categories. Values indicated on the index bar are standardized coefficients of all trait-environment interactions obtained through GLM-Lasso modelling
Discussion
The various pond categories supported comparable odonate species richness levels, though their abundance was greatly affected by landscape fragmentation, as well as by water chemistry and vegetation characteristics. These findings lend partial support to our first hypothesis that landscape configuration and pond characteristics share equal weight in determining odonate diversity patterns. Overall odonate functional richness and functional diversity were not driven by pond category, but rather by water pH gradients. Pond category was also an important driver of odonate assemblage variation, while also closely related to mobility traits of odonates. At the suborder level, although the drivers of anisopteran functional richness and diversity were unclear, for zygopterans they were driven by pond category and vegetation characteristics. These findings partially support our second hypothesis that anisopterans have high affinity to large terrestrial areas, while zygopterans have high affinity to small terrestrial areas. However, our results suggest that zygopterans (with overall lower movement ability) are more sensitive to the effects of landscape fragmentation, while anisopterans (with overall higher movement ability) are more resilient and select suitable habitats based on water chemistry and vegetation characteristics, as was found for other freshwater habitat types (Remsburg et al. 2008; Kietzka et al. 2017).
Relative importance of pond spatial characteristics
Various pond categories supported similar numbers of Odonata species. Yet, pond category was an important driver of individual abundance, with large ponds supporting highest overall odonate abundance, as well as for the two suborders individually. Small ponds are important steppingstone habitats for highly mobile lentic species (Hassall 2014; Maynou et al. 2017), though large ponds may support species often absent from small ponds, while also maintaining larger population sizes (Oertli et al. 2002). This is related to large ponds being more detectable to dispersing individuals, remaining in the landscape for longer in some cases, and having more niche space available, so reducing inter- and intraspecific competition for resources (Kadoya et al. 2004; Ruggiero et al. 2008).
Interestingly however, large ponds in small natural patches had overall higher odonate abundance compared to large ponds in large natural patches. While ponds in small natural patches had less terrestrial habitat available, small natural patches surrounded by tall plantation trees likely provide shelter against strong weather events such as wind and unpredictable downpours, while still receiving the benefits of sunning for most of the day (Harabiš et al. 2013; Timofeev 2016). Due to the windbreak caused by surrounding Eucalyptus spp. plantation trees, these small habitat islands may also better retain warmth and humidity, providing attractive microclimates to the odonates which occupy these habitats (du Toit et al. 2017). This means that the individuals that occupy ponds in small natural patches may be able to remain active around their territories for longer periods of the day.
On the other hand, temperatures in coastal KwaZulu-Natal may often exceed the optimal temperature for odonate activity and rainfall is unpredictable, and some species (e.g., Trithemis spp.) may take shelter during the wettest or warmest part of the day (Damm et al. 2009). Being closely surrounded by tall trees may enable shade tolerant species to avoid overheating or excessively wet conditions, but without the expense of leaving their territories entirely (May 1991; but see the relative importance of vegetation below).
Further emphasizing the overall importance of pond size relative to natural patch size in a fragmented landscape, results also indicated that each pond category supported dissimilar diversity in odonate assemblages. While a variety of ponds are important from an assemblage structuring perspective, overall odonate and anisopteran functional richness and diversity here were not significantly related to pond categories. The overall lentic odonate assemblages in the region are dominated by anisopterans (Samways and Simaika 2016; Deacon et al. 2020), with these findings suggesting that pond variety has a greater influence on the distribution of species as opposed to the distribution of traits across the landscape (Le Gall et al. 2018). However, in the case of zygopterans, functional richness and diversity was highest in large ponds, especially those in small natural grassland patches, suggesting that zygopteran assemblages rely more on large ponds rather than large remnant patches to remain in the landscape (Raebel et al. 2012).
Among the range of odonate traits investigated here, physical traits related to dispersal ability were most important relative to pond category occupancy. Long-winged species predominantly occupied ponds in large patches, while short-winged species largely occupied ponds in small patches. This seems to be related to the area requirements of species in that short-winged species are better adapted to move around in small areas, and consequently have smaller area requirements to persist in any given habitat. Conversely, long-winged species often traverse the landscape and require larger activity areas to persist (Wakeling and Ellington 1997; Rundle et al. 2007).
However, we found that large odonates and those with greater wing-to-body length ratios (as a proxy for greater overall dispersal ability) were more common in small natural patches compared to small species and those with smaller wing-to-body length ratios. This suggests that species with greater overall dispersal ability are better able to discover ponds in small remnant patches, and perhaps move around more freely across fragmented landscapes (Jenkins et al. 2007). This means species with low dispersal ability seldom reach small remnant patches, and are at higher risk when present (Vanschoenwinkel et al. 2013). It is possible that the presence of large-bodied zygopterans with relatively low dispersal ability (e.g., Lestes pallidus and L. plagiatus), and small-bodied anisopterans with relatively high dispersal ability (e.g., Diplacodes lefebvrii, Brachythemis leucosticta, Palpopleura portia, and P. lucia) in our dataset could have amplified these results.
Relative importance of vegetation characteristics and other environmental variables
Relatively higher species richness and abundance levels were associated with higher overall vegetation cover, but lower herbaceous and reeds cover along pond margins. Although vegetation along pond margins is important for odonates, tall and dense vegetation causes shading for long and continuous periods of the day (Remsburg et al. 2008). With some exceptions, most odonates in the region are sun-loving and avoid shaded areas (Samways and Simaika 2016). Ponds with intermediate-height grass cover along margins attract them, as these ponds provide many microhabitats where dragonflies and damselflies can perch, hold territories and hunt, while remaining relatively open and overall free of shading close to pond edges (Briggs et al. 2019a).
Higher abundance of both suborders, as well as high overall functional diversity, were both associated with relatively acidic ponds. With Eucalyptus spp. leaf litter leading to slightly more acidic soils and water (Soumare et al. 2015), and odonates being overall sensitive to changes in water pH (da Rocha et al. 2016; Jooste et al. 2020), these findings suggest that regionally common species are better able to occupy acidic ponds among Eucalyptus plantations. This is further emphasized by high overall functional diversity being associated with ponds with close-to-neutral pH.
High anisopteran abundance here was also associated with relatively cooler ponds. Although slightly warmer water shortens developmental time for most lentic insect immature stages (Suhling et al. 2015), water temperatures above optimal conditions significantly affect immature stage development and leads to lower oxygen levels and high mortality rates (Castillo-Pérez et al. 2022). Adult odonates are able to assess water conditions, and relatively sensitive species may avoid sub-optimally warm aquatic habitats (Kietzka et al. 2017).
Implications for conservation
From a terrestrial perspective, an effective strategy to mitigate landscape fragmentation is to instigate networks of conservation corridors of remnant natural vegetation among plantation compartments (Samways and Pryke 2016). While insects were not originally conservation targets in the area, they have benefitted greatly from conservation methods aimed at large mammals, birds, and vegetation types (Pryke and Samways 2012; Pryke et al. 2015). From an aquatic perspective, plantation forestry operations are usually performed around large freshwater habitats, meaning that large and permanent ponds are often left intact throughout conservation corridor networks. This is related to freshwater habitats in general maintaining hydrological processes, while also being important water sources for vertebrates that roam the area (Samways and Pryke 2016). Small and highly dynamic ponds, on the other hand, are not well protected due to their unpredictable formation and low detectability during the dry season. This means that small ponds are at risk, particularly through infilling (due to obscurity during dry seasons) or oversight to leaving sufficient buffer areas of natural vegetation around small ponds.
Our results emphasize previous findings that conservation of the pondscape is essential for maintaining regional odonate taxonomic and functional diversity patterns (e.g., Hill et al. 2018; Briggs et al. 2019b). Conserving whole pondscapes accounts for variation in local characteristics (e.g., physicochemical gradients among ponds, changes in vegetation characteristics among ponds), to which odonates (Janssen et al. 2018; Perron et al. 2021) and other aquatic insects (Oertli and Parris 2019) respond. However, pond conservation should be considered in the terrestrial context, with natural patch size and terrestrial matrix context also being important drivers of odonate occupancy. These are important considerations for management of landscape transformation, as amphibiotic insects such as odonates track suitable habitats across terrestrial space, especially in the case of wandering species and during pre-reproductive phases (Kadoya et al. 2008).
Both odonate suborders, and the species within each suborder, differ in their terrestrial area size requirements. Although mostly occupied by good dispersers, ponds in small remnant patches are important, as they contribute greatly to regional odonate diversity patterns. Despite ponds in small patches being somewhat isolated, they are also important habitats for those species seeking refuge against unfavorable conditions (Timofeev 2016). Ponds in small patches can be even more attractive to a range of odonate species where there is improved connectivity, and weaker dispersers have greater opportunity to move across the landscape. On the other hand, ponds in relatively large remnant patches may be subject to less artificial disturbance and are important habitats for odonate source populations (Oertli et al. 2002).
Pondscape variety is important, with pondscapes supporting a range of ponds in both small and large patches having high conservation value for regional odonate diversity (Briggs et al. 2019b). In fragmented landscapes, ponds across a dynamic range of sizes in varying sizes of remnant natural grasslands patches should be preserved to encourage full odonate assemblages. By ensuring that a range of pond/natural patch size combinations are represented in pondscape conservation at the landscape level, a range of environmental conditions and microclimates will also be preserved, so enriching regional odonate diversity despite the total landscape being transformed and/or fragmented by plantation forestry.
While our focus was on adult odonates, they are good sentinel organisms for conservation in a variety of landscape types, informing how co-occurring amphibiotic insect taxa are affected by large-scale transformation (Bried et al. 2007). We encourage future studies to incorporate a range of other amphibiotic insect taxa, and other regions where habitat fragmentation threatens biodiversity associated with freshwater habitats, to improve conservation efforts for maintaining regional aquatic and amphibiotic insect diversity patterns.
Data Availability
The datasets generated and/or analysed during the current study are available from the Figshare data repository at: https://figshare.com/s/ebe4b2f17443a943fed3.
References
Bartoń K (2020) MuMIn: Multi-Model Inference. R package version 1.43.17. https://CRAN.R-project.org/package=MuMIn
Bried JT, Ervin GN (2006) Abundance patterns of dragonflies along a wetland buffer. Wetlands 26:878
Bried JT, Herman BD, Ervin GN (2007) Umbrella potential of plants and dragonflies for wetland conservation: a quantitative case study using the umbrella index. J Appl Ecol 44:833–842
Briggs AJ, Pryke JS, Samways MJ, Conlong DE (2019a) Macrophytes promote aquatic insect conservation in artificial ponds. Aquat Conserv 29:1190–1201
Briggs AJ, Pryke JS, Samways MJ, Conlong DE (2019b) Complementarity among dragonflies across a pondscape in a rural landscape mosaic. Insect Conserv Divers 12:241–250
Cardoso P, Barton PS, Birkhofer K et al (2020) Scientists’ warning to humanity on insect extinctions. Biol Conserv 242:108426
Castillo-Pérez EU, Suárez-Tovar CM, González-Tokman D, Schondube JE, Córdoba-Aguilar A (2022) Insect thermal limits in warm and perturbed habitats: dragonflies and damselflies as study cases. J Therm Biol 103:103164
Chin KS, Taylor PD (2009) Interactive effects of distance and matrix on the movements of a peatland dragonfly. Ecography 32:715–722
Conrad KF, Willson KH, Harvey IF, Thomas CJ, Sherratt TN (1999) Dispersal characteristics of seven odonate species in an agricultural landscape. Ecography 22:524–531
Copernicus Sentinel2 Data (2022) Copernicus Open Access Hub. Processed by ESA. https://scihub.copernicus.eu. Accessed 12 August 2022
da Rocha FC, de Andrade EM, Lopes FB, de Paula Filho FJ, Costa Filho JH, da Silva MD (2016) Physical-chemical determinant properties of biological communities in continental semi-arid waters. Environ Monit and Assess 188:1–15
Damm S, Dijkstra KDB, Hadrys H (2009) Red drifters and dark residents: Africa’s changing environment reflected in the phylogeny and ecology of a Plio-Pleistocene dragonfly radiation (Odonata, Libellulidae, Trithemis). Conservation genetics, Speciation and Biogeography in African Dragonflies 167
Deacon C, Samways MJ, Pryke JS (2020) Determining drivers of dragonfly diversity patterns and the implications for conservation in South Africa. Biol Conserv 245:108548
Deacon C, Samways MJ, Pryke JS (2021) Relative importance of ecological versus biological traits in driving range sizes of african dragonflies. J Biogeogr 48:1309–1321
du Toit B, Malherbe GF, Kunneke A, Seifert T, Wessels CB (2017) Survival and long-term growth of eucalypts on semi-arid sites in a Mediterranean climate, South Africa. South for 79:235–249
Fox J, Weisberg S An R Companion to Applied Regression, Third Edition. Thousand Oaks CA:, Sage (2019) https://socialsciences.mcmaster.ca/jfox/Books/Companion/
French SK, McCauley SJ (2019) The movement responses of three libellulid dragonfly species to open and closed landscape cover. Insect Conserv Divers 12:437–447
Harabiš F (2017) Does the management of surrounding terrestrial habitats increase the tendency of odonates to leave aquatic habitats? Biodivers Conserv 26:2155–2167
Harabiš F, Dolny A (2011) The effect of ecological determinants on the dispersal abilities of central european dragonflies (Odonata). Odonatologica 40:17–26
Harabiš F, Tichanek F, Tropek R (2013) Dragonflies of freshwater pools in lignite spoil heaps: restoration management, habitat structure and conservation value. Ecol Eng 55:51–61
Hart LA, Bowker MB, Tarboton W, Downs CT (2014) Species composition, distribution and Habitat types of Odonata in the iSimangaliso Wetland Park, KwaZulu-Natal, South Africa and the Associated Conservation Implications. PLoS ONE 9:e92588
Hassall C (2014) The ecology and biodiversity of urban ponds. Wiley Interdiscip Rev Water 1:187–206
Hassall C, Hill M, Gledhill D (2016) The ecology and management of urban pondscapes. Urban Landscape Ecology. Routledge, pp 147–165
Heino J (2009) Biodiversity of aquatic insects: spatial gradients and environmental correlates of assemblage-level measures at large scales. Freshw Reviews 2:1–29
Hill MJ, Hassall C, Oertli B et al (2018) New policy directions for global pond conservation. Conserv Lett 11:e12447
Hill MJ, Heino J, White JC, Ryves DB, Wood PJ (2019) Environmental factors are primary determinants of different facets of pond macroinvertebrate alpha and beta diversity in a human-modified landscape. Biol Conserv 237:348–357
Hof C, Brändle M, Brandl R (2006) Lentic odonates have larger and more northern ranges than lotic species. J Biogeogr 33:63–70
Janssen A, Hunger H, Konold W, Pufal G, Staab M (2018) Simple pond restoration measures increase dragonfly (Insecta: Odonata) diversity. Biodivers Conserv 27:2311–2328
Jenkins DG, Brescacin CR, Duxbury CV et al (2007) Does size matter for dispersal distance? Glob Ecol and Biogeogr 16:415–425
Jooste ML, Samways MJ, Deacon C (2020) Fluctuating pond water levels and aquatic insect persistence in a drought-prone Mediterranean-type climate. Hydrobiologia 847:1315–1326
Kadoya T, Suda SI, Washitani I (2004) Dragonfly species richness on man-made ponds: effects of pond size and pond age on newly established assemblages. Ecol Res 19:461–467
Kadoya T, Suda SI, Tsubaki, Washitani I (2008) The sensitivity of dragonflies to landscape structure differs between life-history groups. Landsc Ecol 23:149–158
Kalkman VJ, Clausnitzer V, Dijkstra KDB, Orr AG, Paulson DR, Tol JV (2007) Global diversity of dragonflies (Odonata) in freshwater. Freshwater animal diversity assessment. Springer, Dordrecht, pp 351–363
Kietzka GJ, Pryke JS, Samways MJ (2017) Aerial adult dragonflies are highly sensitive to in-water conditions across an ancient landscape. Divers Distrib 23:14–26
Knight TM, McCoy MW, Chase JM, McCoy KA, Holt RD (2005) Trophic cascades across ecosystems. Nature 437:880–883
Laliberté E, Legendre P (2010) A distance-based framework for measuring functional diversity from multiple traits. Ecology 91:299–305
Le Gall M, Fournier M, Chaput-Bardy A, Husté A (2018) Determinant landscape‐scale factors on pond odonate assemblages. Freshw Biol 63:306–317
Lima M, Firmino VC, de Paiva CKS, Juen L, Brasil LS (2022) Land use changes disrupt streams and affect the functional feeding groups of aquatic insects in the Amazon. J Insect Conserv 26:136–148
May ML (1991) Thermal adaptations of dragonflies, revisited. Adv Odonatol 5:71–88
Maynou X, Martín R, Aranda D (2017) The role of small secondary biotopes in a highly fragmented landscape as habitat and connectivity providers for dragonflies (Insecta: Odonata). J Insect Conserv 21:517–530
McCauley SJ (2006) The effects of dispersal and recruitment limitation on community structure of odonates in artificial ponds. Ecography 29:585–595
Mucina L, Scott-Shaw CR, Michael C, Camp KGT, Matthews WS, Powrie LW, Hoare DB (2006) Indian Ocean coastal belt. In: Mucina L, Rutherford MC (eds) The vegetation of South Africa, Lesotho and Swaziland, vol 19. Strelitzia, South African National Biodiversity Institute, pp 569–583
Myers N, Mittermeier RA, Mittermeier CG, da Fonseca GA, Kent J (2000) Biodiversity hotspots for conservation priorities. Nature 403:853–858
Nagy HB, László Z, Szabó F, Szőcs L, Dévai G, Tóthmérész B (2019) Landscape-scale terrestrial factors are also vital in shaping Odonata assemblages of watercourses. Sci Rep 9:1–8
Oertli B, Parris KM (2019) Toward management of urban ponds for freshwater biodiversity. Ecosphere 10:e02810
Oertli B, Joye DA, Castella E, Juge R, Cambin D, Lachavanne JB (2002) Does size matter? The relationship between pond area and biodiversity. Biol Conserv 104:59–70
Oksanen J, Blanchet FG, Friendly M et al (2020) vegan: Community Ecology Package. R package version 2.5–7. https://CRAN.R-project.org/package=vegan
Osborn R, Samways MJ (1996) Determinants of adult dragonfly assemblage patterns at new ponds in South Africa. Odonatologica 25:49–58
Outomuro D, Johansson F (2019) Wing morphology and migration status, but not body size, habitat or Rapoport’s rule predict range size in North-American dragonflies (Odonata: Libellulidae). Ecography 42:309–320
Perron MAC, Richmond IC, Pick FR (2021) Plants, water quality and land cover as drivers of Odonata assemblages in urban ponds. Sci Total Environ 773:145467
Pryke JS, Samways MJ (2012) Ecological networks act as extensions of protected areas for arthropod biodiversity conservation. J Appl Ecol 49:591–600
Pryke JS, Samways MJ, De Saedeleer K (2015) An ecological network is as good as a major protected area for conserving dragonflies. Biol Conserv 191:537–545
Quantum GIS, Development Team (2022) Quantum GIS geographic information system. Open source Geospatial Foundation project. http://qgis.osgeo.org
R Core Team (2020) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Raebel EM, Merckx T, Feber RE, Riordan P, Thompson DJ, Macdonald DW (2012) Multi-scale effects of farmland management on dragonfly and damselfly assemblages of farmland ponds. Agric Ecosyst Environ 161:80–87
Remsburg AJ, Olson AC, Samways MJ (2008) Shade alone reduces adult dragonfly (Odonata: Libellulidae) abundance. J Insect Behav 21:460–468
Riad P, Graefe S, Hussein H, Buerkert A (2020) Landscape transformation processes in two large and two small cities in Egypt and Jordan over the last five decades using remote sensing data. Landsc Urban Plan 197:103766
Ruggiero A, Céréghino R, Figuerola J, Marty P, Angélibert S (2008) Farm ponds make a contribution to the biodiversity of aquatic insects in a french agricultural landscape. C R Biol 331:298–308
Rundle SD, Bilton DT, Foggo A (2007) Chap. 10: by wind, wings or water: body size, dispersal and range size in aquatic invertebrates. In: Hildrew A, Raffaelli DG, Edmonds-Brown R (eds) Body size: the structure and function of aquatic ecosystems. Cambridge University Press, Cambridge, pp 186–209
Sahlén G (2006) Specialists vs. generalists in the Odonata, the importance of forest environments in the formation of diverse species pools. In: Cordero Rivera A (ed) Forests and Dragonflies. Pensoft, pp 153–180
Samways MJ, Pryke JS (2016) Large-scale ecological networks do work in an ecologically complex biodiversity hotspot. Ambio 45:161–172
Samways MJ, Simaika JP (2016) Manual of Freshwater Assessment for South Africa: Dragonfly Biotic Index. Suricata 2. South African National Biodiversity Institute, Pretoria
Sarkar D (2008) Lattice: Multivariate Data visualization with R. Springer, New York
Soumare A, Manga A, Fall S, Hafidi M, Ndoye I, Duponnois R (2015) Effect of Eucalyptus camaldulensis amendment on soil chemical properties, enzymatic activity, Acacia species growth and roots symbioses. Agrofor Syst 89:97–106
Suhling F, Suhling I, Richter O (2015) Temperature response of growth of larval dragonflies – an overview. Int J Odonatol 18:15–30
Thorp JH, Rogers DC (eds) (2014) Thorp and Covich’s freshwater invertebrates: ecology and general biology, vol 1. Elsevier
Timofeev AN (2016) The ecology and behavioral features of dragonflies (Insecta, Odonata) in the forest-steppe zone of Central Chernozem region. Russ J Ecol 47:501–507
Vanschoenwinkel B, Buschke F, Brendonck L (2013) Disturbance regime alters the impact of dispersal on alpha and beta diversity in a natural metacommunity. Ecology 94:2547–2557
Vavrek MJ (2011) Fossil: palaeoecological and palaeogeographical analysis tools. Palaeontologia Electronica 14:1T. http://palaeo-electronica.org/2011_1/238/index.html
Wakeling JM, Ellington CP (1997) Dragonfly flight III. Lift and power requirements. J Exp Biol 200:583–600
Wang Y, Naumann Y, Eddelbuettel D, Wilshire J, Warton D (2020) mvabund: Statistical Methods for Analysing Multivariate Abundance Data. R package version 4.1.6. https://CRAN.R-project.org/package=mvabund
Acknowledgements
This work was conducted under Ezemvelo KZN Wildlife permit number OP2085/2021. Access to study sites was granted by iSimangaliso Wetland Park and Mondi Group. We thank Robyn Symons for assistance in the field, and Mondi Group International and the Foundational Biodiversity Information Programme for funding the research.
Funding
Open access funding provided by Stellenbosch University. This work was supported by Mondi Group International, and the Foundational Biodiversity Information Programme (FBIP). Author CD received research support from FBIP, and authors MJS and JSP received support from Mondi Group International.
Open access funding provided by Stellenbosch University.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, analyses and preparation of the first draft were performed by Charl Deacon. Michael Samways and James Pryke commented on and edited previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Communicated by Nigel Stork.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Online Resource 1
: Lentic habitat types in northeast KwaZulu-Natal, South Africa. (a) small pond in small patch, (b) large pond in small patch, (c) small pond in large patch, and (d) large pond in large patch
Online Resource 2
: Species accumulation curve and species richness estimators for lentic odonates in northeast KwaZulu-Natal, South Africa
Online Resource 3
: Species list and abundances of odonates recorded from northeast KwaZulu- Natal
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Deacon, C., Samways, M.J. & Pryke, J.S. Interplay between pond size and matrix extent drives odonate diversity patterns in a fragmented landscape. Biodivers Conserv 32, 4767–4785 (2023). https://doi.org/10.1007/s10531-023-02726-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-023-02726-4