Abstract
Today, the importance of restoring natural forest disturbance regimes and habitat structures for biodiversity is widely recognized. We evaluated the immediate effects of two restoration methods on wood-inhabiting (saproxylic) beetles in boreal forest voluntary set-asides. We used a before-after control-impact experimental set-up in 15 set-asides; each assigned to one of three treatments: (1) restoration burning, (2) gap cutting and (3) no-treatment reference stands. Before treatment, abundance, species richness and assemblage composition of trapped beetles did not differ significantly among treatments. Burning resulted in a significant change in assemblage composition and increased species richness and abundance compared to reference stands. As predicted, saproxylic species known to be fire favoured increased dramatically after burning. The immediate response shows that, initially, fire favoured species are attracted from the surrounding landscape and not produced on site. Gap cutting increased the abundance of cambium consumers but had no significant effect on total species richness or assemblage composition of saproxylic beetles. The stronger effect of burning compared to gap cutting on saproxylic assemblages is probably due to the very specific conditions created by fires that attracts many disturbance-dependent species, but that at the same time disfavour some disturbance-sensitive species. By contrast, gap cutting maintained assemblage composition, increased abundances and is likely to increase species richness in the years to follow, due to elevated level of dead wood. The restoration methods applied in this study may prove particularly useful, partly because of positive effect on saproxylic beetles, but also due to the cost-efficiency of the measures; the voluntary set-asides were already established and the restoration costs fully covered by revenue from the extracted timber.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Global extraction of forest resources has led to changes in ecosystem structures and processes, losses of biodiversity and declines in ecosystem services (FAO 2010). In the boreal forest, intensive management for timber production has caused declines in biodiversity and decrease in habitat quality for a large number of specialized species (Kuuluvainen 2009; Paillet et al. 2010). A likely explanation is that modern forestry practice (large scale clear-cutting) and fire protection create even-aged stands with reduced tree species diversity and shorter rotation periods compared to naturally dynamic stands. As a result, stand variability is reduced as well as the availability of dead wood substrates, known to be important for biodiversity. These changes have reduced the amount of potential habitat in managed forests and are considered a key factor underlying species declines in degraded boreal forest ecosystems (Buddle et al. 2006; Hjältén et al. 2012; Jonsson et al. 2005; Siitonen 2001; Stenbacka et al. 2010). As a consequence, a commitment to restore at least 15% of degraded forest ecosystems was established at the Aichi Convention on Biological Diversity 2010 (CBD 2010) and echoed in the EU Biodiversity Strategy for 2020 (EU 2011). As very little undisturbed forest habitats remain in many regions of the world, we have reached a situation where we no longer can rely on passive conservation measures, i.e. setting aside conservation areas under a free development philosophy (Aronson and Alexander 2013; MEA 2005). Instead, to achieve conservation goals, we need methods for restoration of hitherto managed forest, as well as for active management of forest reserves. Thus, today ecological restoration is recognized as a global priority and restoration efforts have increased exponentially (Jacobs et al. 2015; Sayer et al. 2004; Stanturf 2015).
However, conducting forest restoration is far from straightforward. Restoration theory is generally based on the assumption that it is appropriate to mimic natural processes (Lindenmayer et al. 2006). Fires and storms are among the most important abiotic disturbances in natural boreal forests (Saint-Germain et al. 2008; Siitonen 2001; Zackrisson 1977). Fire used to be the predominant large-scale disturbance in boreal forests, but in many areas such as Scandinavia fire frequency has dropped dramatically during the past century due to effective fire suppression measures (Zackrisson 1977). For example, less than 0.02% of the forest area burns each year in Sweden compared with approximately 1% before 1900 AD (Granström 2001; Zackrisson 1977). Many boreal species are adapted to fire (Granström and Schimmel 1993) and some species breed almost exclusively in burned forest (Buddle et al. 2006; Wikars 1997). Many of these species have a very good dispersal ability and can detect and colonize burned stands from a far (Wikars 1997; Hyvärinen et al. 2005). However, due to the low fire frequency in many boreal areas it has been argued that some fire adapted species must be able to maintain viable populations in the unburned forest matrix if it is of sufficient high quality, e.g. contains sufficient amounts of dead wood (Saint-Germain et al. 2008). Still, several specialized species associated with fire have declined and many of them are red-listed in Sweden (Westling 2015).
However, non-stand replacing disturbances such as gap dynamics have historically also been important for providing variation in light and structures in substantial portions of the boreal forest (Kuuluvainen and Aakala 2011). Commercial managed forests are denser, have less variation in tree height and are less permeable to sunlight than natural forest. In Sweden, stand-level volumes of timber have increased with 40–80% since the 1950s (SLU 2012). At the same time, deciduous trees have been disfavoured, e.g. during thinning. This has led to an impoverished fauna of species associated with sun-exposed conditions and deciduous broad-leaved trees (Berg et al. 1994; Bernes 2011; Westling 2015). Thus, restoring natural fire dynamics as well as gap dynamics is likely to improve the situation for biodiversity in boreal forest landscapes.
Finding areas suitable for restoration is a challenge. Restoration of legally protected areas, e.g. nature reserves and national parks, is often controversial (Angelstam et al. 2011). However, voluntary set-asides established as part of forest certification requirements could provide a solution. In Sweden and Finland, the FSC standard stipulates that major landholders should set aside ≥5% of their productive forest area for conservation purposes (Anonymous 2010a, 2014). A majority of these voluntary set-asides are mature forests historically only subjected to selective felling and thus maintaining populations of some species associated with old-growth forest characteristics (Gustafsson et al. 2004; Hjältén et al. 2012; Stenbacka et al. 2010). FSC certification requirements also stipulate that forest companies shall burn the equivalent of ≥5% of the regeneration area on dry and mesic soils annually (Anonymous 2014). This also includes the possibility to burn standing forest for conservation purposes. Hence, voluntary set-asides may constitute a key resource for the implementation of restoration measures in boreal forest landscapes.
The aim of this study was to evaluate short-term effects of two different restorations methods (restoration burning and gap cutting) on saproxylic beetles, a group of insects severely threatened by modern forestry. We predict the following treatment specific effects on saproxylic beetle assemblages the first few months after the restoration treatment:
-
1.
Restoration burning increases species richness and abundance of fire dependent saproxylic species by attracting them from the surroundings. The richness and abundance of saproxylic species connected to late successional forest and forest continuity is expected to decrease in burned stands.
-
2.
Gap cutting increases species richness and abundance of saproxylic species, due to an increase of both light demanding species and species attracted to recently killed trees retained in 50% of the gaps, while still maintaining most extant late-successional species.
Materials and methods
Study area
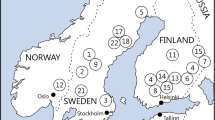
The study was conducted in northern Sweden, in an area (63°23′N to 64°30′N and 17°37′E to 21°20′E) belonging to the middle and northern boreal zones (Ahti et al. 1968). Fifteen forest stands, representative of the most common type of mature managed stands found in northern Sweden were selected for the study. The stands were selected from a larger number (approximately 60) of candidate stands, based on stand data provided by the forest company Holmen and visual inspection of all stands. To reduce between-stand variation, stand characteristics were standardized in selected stands and although there were variation in stand characteristics between individuals stands, similar variation was obtained within all treatment groups with respect to age (range 80–160 years), tree species composition [mix of pine (30–70%), spruce (30–60%), deciduous (5–20%)], field layer vegetation (Vaccinium myrtillus or V. myrtillus–V. vitis-idaea dominated) and stand volume (150–270 m3 ha−1) (Table 1). All stands included in the study are voluntary set-asides as a part of fulfilment of FSC certification requirements for one of Sweden’s forest major companies. Stand sizes varied between 3.5 and 21 ha. Norway spruce (Picea abies) and Scots pine (Pinus sylvestris) were the dominating tree species, whereas deciduous trees such as downy birch (Betula pubescens), silver birch (B. pendula), aspen (Populus tremula) and goat willow (Salix caprea) also occurred scattered throughout the stands.
Restoration treatments
Three treatments—restoration burning (‘Burn’), gap cutting (‘Gap’) and untreated reference (‘Ref’)—were, except for in one case, geographically stratified (stands in all treatments were evenly distributed in the geographical area used) and assigned to five forest stands each (Fig. 1). In the five forest stands that were assigned to burning, 5–30% of the trees at stand level were cut prior to burning and most of the cut trees were extracted as timber in the early spring of 2011. This logging was done to speed up the drying out of the forest floor and to cover the costs of restoration. Nevertheless, approximately 2–5 m3 ha−1 of the cut trees were left on site. Depending on local weather conditions at the different forest stands, burning was carried out between the 10th of June and the 13th of July 2011. Gap cutting was carried out during the winter/spring of 2011 before snowmelt. In each of the five gap cutting stands, standard harvesters were used to create small gaps (Ø = 20 m) in total covering approximately 19% of the stand area. Each gap was centred on one to three large retained trees; preferably goat willow, aspen, silver birch or downy birch, but when none of these species were present in the gap, Scots pine was retained instead. In 50% of the gaps, the trees were cut at the base and extracted as timber. In the 50% of gaps where trees were retained, all trees except for the centre trees were killed in four different ways and retained on-site; they were either cut, pushed over, ringbarked or truncated 3–4 m above ground to create a high stump. The timber extraction in 50% of the stand was done to ensure that the amount of fresh dead wood in the stand would not exceed the limits set by Swedish forestry legislation (5 m3 ha−1 of fresh dead wood from conifers). The remaining five forest stands were left as untreated references. The timer extracted at gap-cutting and thinning prior to burning, covered the cost for the restoration treatments (Olof Norgren personal communication).
Insect sampling
In each of the 15 set-asides, we deployed three flight interception traps of the Polish IBL2 model (for description see Stenbacka et al. 2010). The IBL2-traps were placed at 1.5–2 m height 30 m from the centre of each set aside with a between-trap angle of 120°. The year before treatment (2010), beetles were trapped between the 1st of June and the end of September. During the treatment year (2011), traps were placed on the burned stands and on the corresponding gap cut and reference stands 1–2 days after the fires, and were emptied in the end of September.
The beetles were counted and identified to species level [with the exception of Acrotrichis spp. (Fam. Ptiliidae)] by experts. We classified beetle species as saproxylic according to the definition of Speight (1989), (Stokland et al. 2012) and we also classified beetles according to feeding habits using the database for saproxylic beetles (Anonymous 2007), with the addition of species confined to the northern part of Sweden (Hilszczański, J., Pettersson, R. and Lundberg, S. pers. comm.). Red-list status was based on the Swedish red list (Westling 2015). The classification of fire favoured and fire dependent species follows Wikars (2006). Nomenclature and taxonomy of the beetles follows Dyntaxa (2015).
Statistical analyses
We used generalized linear mixed models (GLMM) with Poisson distributed errors to analyse the effect of restoration treatment on the abundance and species richness of all saproxylic beetles, as well as separately for the following subgroups: cambivores, fungivores, predators, wood borers, fire favoured or fire dependent species and red-listed species. Treatment type, time relative to restoration (before or after restoration) and the interaction between these two factors were analysed as fixed factors, while the identity of each trap nested within the experimental stand was included as a random factor. In addition, we used observation level random effects (OLRE) to account for overdispersion when the ratio between residual deviance and degrees of freedom exceeded 1.4. When statistically significant effects (α = 0.05) were detected in the GLMM-analysis, we further explored the differences between treatment pairs with post hoc Tukey tests. We used the rarefy function of the vegan package to create rarefied species richness values for each stand. Since the numbers of collected beetles were too few (min 14) to be analysed on trap level, we pooled the data to stand level. We used linear mixed effects models with normally distributed errors to study differences in rarefied species richness between treatments. Analogues to the GLMM—analyses we set rarefied species richness as response variable with treatment type, time relative to restoration (before or after restoration) and the interaction between these two as fixed factors. Stand identity was set as a random factor. Rarefied species richness was analysed for the group containing all saproxylic beetles, the remaining groups contained to few beetles to be analysed in a meaningful way. We used ManyGLM, which is a model-based analysis of multivariate abundance data (Wang et al. 2012; Warton et al. 2012) to analyse differences in the composition of saproxylic species assemblages between treatments. As it is currently not possible to include random factors in the ManyGLM procedure, differences in the composition of species assemblages among treatments were analysed separately for the two sampling occasions (i.e. before and after restoration). When significant effects were detected, we created non-orthogonal contrasts to test for differences in the composition of species assemblages for all treatment pairs. For the treatment pairs that showed significant differences in assemblage composition, we used the univariate procedure implemented in ManyGLM to examine which speciest contributed significantly to the differences in assemblage composition. We used two-dimensional NMDS-plots based on Bray–Curtis dissimilarities for non-transformed data to visualize differences in assemblage composition between treatments. We found no deviant patterns in the distribution of residuals that indicated poor fit in any of the GLMMs or the ManyGLMs analyses conducted. We used the following packages in the statistical software R (R-Core-Team 2015) for the statistical analyses: lme4 (Bates et al. 2015) for the GLMM-analysis; multcomp (Hothorn et al. 2008) for the pairwise Tukey testing; mvabund (Wang et al. 2012) for the manyGLMs, vegan (Oksanen et al. 2015) for the NMDS ordination and RVAideMemoire to control for overdispersion.
Results
In total, we collected 15,100 saproxylic beetles belonging to 328 species (Online Appendix 1). The most common species were Cryptophagus lapponicus and Pityogenes chalcographus, representing 15.7 and 10.3% of the catch, respectively. The most species rich functional group were fungivores with 143 species followed by predators with 86 species, cambivores with 48 species and wood borers with 28 species.
The abundance and species richness of saproxylic beetles did not differ among treatments groups prior to the restoration treatment. The only exception being species richness of red-listed species that were lower in stands assigned for burning than in reference stands. By contrast, after treatment the overall abundance as well as the abundance of all studied functional groups, except for wood borers and red-listed species, was higher in burned stands compared to reference and gap-cut stands (Table 2; Fig. 2). In addition, cambivores were more abundant in gap-cut stands than in reference stands. Similar, although less pronounced, patterns were found for species richness, with higher richness for all saproxylic beetles, cambivores, fungivores, predators and fire favoured beetles in burned than in reference stands and a higher richness of all saproxylic beetles, cambivores, predators, wood boorers and fire favoured in burned compared to gap-cut stands (Fig. 3; Table 2). Species richness of cambivores and fire favoured beetles differed between gap-cut and reference stands after treatment. The abundances of saproxylic beetles were generally lower after (2011) compared to before (2010) restoration with the exception of burned stands, where abundances were generally higher after restoration (2011) (Fig. 2). Analyses of overall rarefied species richness revealed a reduced rarefied species richness in burned stands (see supplementary material).
Abundance of trapped saproxylic beetles of different groups: i all species, ii cambivores, iii fungivores, iv predators, v wood borers, vi fire favoured species, and vii red-listed species. White pre-treatment and grey post-treatment. Different letters indicate significant differences in abundance among treatments post-restoration (there were no significant differences in abundance among treatments before restoration). Stars indicate differences in abundance between pre- and post-restoration within treatment groups and error bars show SE
Species richness of trapped saproxylic beetles of different groups: i all species, ii cambivores, iii fungivores, iv predators, v wood borers, vi fire favoured and fire dependent species, and vii red-listed species. White pre-treatment and grey post-treatment. Different case letters indicate significant differences in species richness among treatments post-restoration (there were no significant differences in species richness among treatments before to restoration). Stars indicate differences in species richness between pre- and post-restoration within treatment groups and error bars show SE
The assemblages composition did not differ among treatment groups prior to the restoration treatment (Fig. 4; Table 3) but after treatment assemblage composition differed significantly between burned and reference stands as well as between burned and gap-cut stands (Fig. 4; Table 3). By contrast, no significant difference in assemblage composition was found between gap-cut and reference stands. Eighteen species contributed significantly to the difference in assemblage composition between burned stands and both of the other two stand types (Table 4). In all cases, these species were more abundant in burned stands. Nine of these species, belonging to different functional groups, cambivores, fungivores and predators, are known to be favoured by forest fire and two are described as fire dependent, meaning that they depend on fire to maintain viable populations. An additional fourteen species were significantly more abundant in burned than in reference stands and five were more abundant in burned than gap-cut stands. Four species were more abundant in gap-cut than burned stands and seven species were more abundant in reference stands than in burned stands. In these to latter cases, none of the species were cambivores or wood borers, they were all fungivores or predators.
NMDS visualisation of species compositions in the experimental forest stands pre- and post-restoration. Stress values are 2010 = 0.1514717; 2011 = 0.07823413. Each symbol represents the beetles trapped in one experimental forest stand, where squares control stands, triangles artificial gap cuttings and circles burned stand
We collected 173 individuals belonging to 26 species figuring on the Swedish red list. Most red-listed species occurred in low numbers and we found no statistically significant effects of treatment on the species richness or abundance of red-listed species (Table 2; Figs. 2, 3).
Discussion
Fire effects
We found strong support for our first hypothesis, i.e. burning increased the species richness and abundance of fire favoured and fire dependent species, probably by attracting them from the surrounding landscape. However, fire also increased abundance and species richness of saproxylic beetles in general, the only exception being wood borers. However, it should be noted that not all species increased in abundance in burned stands. In fact, 125 of the species were collected in higher numbers in burned than in reference stands whereas 89 species were collected in lower numbers in burned stands compared to reference stands (Online Appendix 1). This suggested that the higher species richness found in burned stands was not simply due to an increased activity of all beetle species in burned stands (see also Hyvärinen et al. 2005). Furthermore, most of the increase in overall abundance was explained by a few species becoming superabundant in burned stands compared to reference stands, (e.g., Pitogenes chalcographus, 1444 vs 7; Polygraphus poligraphus 431 vs 5; Atomaria longicornis 657 vs 9; and Corticaria ferruginea 427 vs 2 individuals in burned vs reference stands) as also shown in other studies (Hyvärinen 2005). This also explains the reduced rarefied species richness in burned stands as these superabundant species will press the rarefaction curve downwards, which means that for a standardized number of individuals sampled fewer species will be detected in burned than in reference stands. However, this does not mean that the actual number of species collected in burned stands was reduced compared to reference stands, more species is still collected in these stands but also more individuals.
We collected beetles the same years as the burning was conducted and the strong immediate response indicates that beetle actually were attracted from the surrounding managed forest landscape and not produced on-site. This supports the view that that some, albeit not all, fire adapted species must be able to maintain viable populations in the unburned forest matrix if it is of sufficient high quality (Saint-Germain et al. 2008) as fire frequency is extremely low in many landscapes, including the landscapes used in this study, due to efficient fire protection programmes. Further support for this is that several of the fire dependent and fire favoured species collected in this study have also been found in unburned forest reserves with no recent history of fire but with quite high volumes of dead wood (e.g. Hyvärinen et al. 2005). For example, Sphaeriestes stockmanni Orthotomicus suturalis, Corticaria ferruginea, Phloeostiba lapponica and Caenoscelis ferruginea also occurs in unburned stands with high volumes of dead wood, although most of these species are more common in burned stands (Hyvärinen et al. 2005; Hjältén et al. 2012). Some fire favoured also occurs in clear cuts given that sufficient dead wood is made available (Wikars 2002; Hjältén et al. 2012). This being said we should also acknowledge that some species, e.g. Henoticus serratus, seemingly only occurs of burned areas (Hyvärinen et al. 2005; Wikars 2002), suggesting that for some species fire might be essential for maintaining viable population and fire is certainly beneficial for all these species.
The effect of on-site production of saproxylic beetles is more likely to occur in the years after burning (Hyvärinen et al. 2006). This suggests that burned areas can maintain high population of fire favoured species over considerable time as weakened trees will continue to die for many years after a fire, thus creating new fresh dead wood substrates that can be colonized (Boucher et al. 2012). However, long-term studies are needed to verify this. Still one should be aware that the beetle response to burning partly depends on the level of tree retention (Hyvärinen et al. 2009). Retention levels in our burned stands always exceeded 65% of the standing crop (average retention 76%) and a large proportion of these trees were killed or damaged by the fire, which may have contributed to the strong attraction of saproxylic beetles.
The significant changes in assemblage composition, which is consistent with earlier studies of fire effects on saproxylic communities (Boucher et al. 2012; Hekkala et al. 2014; Hyvärinen et al. 2009; Johansson et al. 2011) was not only explained by higher abundances of fire favoured and fire dependent species but also by higher abundance of many other species, belonging to different functional groups, in burned stands. As predicted we found an increased abundance and species richness of cambivores and fire favoured beetles, most likely explained by the increased availability of dead cambium (Saint-Germain et al. 2004, 2008; Wikars 1997). The cambivores P. chalcographus, and P. poligraphus were both more than 200 times more abundant in burned than in reference stands (Online Appendix 1).
The abundance of fungivores and predators also increased significantly in burned stands, the latter most likely a result of predators tracking increased prey availability (Azeria et al. 2012; Johansson et al. 2007) and predators are often attracted to the same volatiles as their prey (Hulcr et al. 2006; Schroeder 2003). Many common fungivorous beetles has been found to increase following natural fire (Johansson et al. 2011) and we found a similar pattern in our prescribed burns, the catch of the abundant fungivores Corticaria ferruginea, Henoticus serratus and Corticaria rubripes was 400, 100 and 9 times higher, respectively, in burned stands than in the other forest types (Online Appendix 1).
As predicted, some saproxylic beetle species associated with old-growth forest characteristics were disfavoured by fire. Out of the seven species that had significantly lower densities in burned than in reference stands, a majority can be classified as associated with mature forests with long forest continuity. For example, the abundance of Lordithon speciosus, Quedius plagiatus and Stenichnus bicolor was found to be 3–6 times higher in mature forest than in clear-cuts or young forests (Hjältén et al. 2012). This suggests that fire has detrimental effects on some saproxylic beetles and that this should be considered in restoration planning.
We did not find any significant positive effect of fire on the species richness of red-listed beetle species. The reason for this is not clear and other studies have reported positive effects of fire on red-listed species (Hekkala et al. 2014; Kouki et al. 2012). However, the inherently low abundance of red-listed species increases the influence of stochasticity in the data and makes it more difficult to identify patterns. Other possible explanations for this lack of response are that intensively forest management in this landscape has resulted in an impoverished regional species pool of red-listed species, as shown in other studies (Kouki et al. 2012); alternatively, that rare low density red-listed species should be expected to exhibit a slower response to fire than more abundant early successional saproxylic species. Hekkala et al. (2014) reported that both the species richness and abundance of rare and red-listed species was higher the second year after fire compared to the first year after fire.
Effect of gap cutting
We found limited support for our second hypothesis: gap cutting did not generally lead to an immediate significant increase in abundance and species richness of saproxylic beetles. The only exception from this was the abundance and species richness of cambium consumers and fire favoured species (species richness only) that was higher in gap-cut stands than in reference stands. This provides partial support for our prediction of an increase of both light demanding species and species attracted to recently killed trees in gap cut stands. Cambium consumers such as Dryocoetes autographus and Pityogenes chalcographus are early colonizers, well equipped to localize and colonize fresh dead wood (Johansson et al. 2006; Toivanen et al. 2009; Wermelinger et al. 2002). The abundances of D. autographus, Polygraphus poligraphus and P. chalcographus were 6, 5 and 15 times higher in gap-cut than in reference stands, respectively (Online Appendix 1). The higher availability of fresh cambium from killed trees in gap-cut compared to reference stands is a likely explanation for the overall higher abundance of cambium consumers in gap-cut stands.
The overall saproxylic assemblage composition did not change significantly due to gap cutting, providing support to our prediction that most extant species adapted to the conditions in mature forest are maintained in gap-cut stands. We sampled beetles the same year as the restoration was conducted, whereas in many earlier studies reporting significant changes in assemblage composition sampling was conducted the year after treatment or later (Buddle et al. 2006; Hyvärinen et al. 2009; Toivanen and Kotiaho 2007). Thus, it is likely that thee assemblage composition will change in the next couple of years as gap cutting increased stand heterogeneity with respect to light and temperature. Thus, the population and community response to the increase in available dead wood in gap cut stands might take more than one summer. Secondary species such as many fungivores and wood borers are expected to increase in numbers after a couple of years as the dead wood decays (Lee et al. 2014; Toivanen and Kotiaho 2010). Others studies also found a lack of short term response to dead wood creation (Toivanen and Kotiaho 2007) or that the temporal dynamics of species assemblages are more pronounced in burned sites than in sites where dead wood have been produced without burning (Boucher et al. 2012). This stresses the need for long-term studies of restoration effects.
Fire versus gap cutting
As expected, the abundance, species richness and assemblage composition differed between gap-cut and burned stands (see also e.g. Boucher et al. 2012). Many species were significantly favoured by fire compared to gap cutting and reference stands but some species exhibited higher abundances in gap-cut and reference stands compared to burned stands. This can be explained by the fact that the increase in temperature and sun exposure in burned stands will disfavour species adapted to the often more damp and shady conditions in old-growth forest and this stresses the need to consider alternative restoration methods in boreal forest. Burning is therefore not a suitable method in situations when restoration is aimed to improve the situation for old-growth associated species. In these situations gap-cutting is likely to be a better option as this will increase dead wood availability and thus overall population density without inducing dramatic changes in assemblage composition. To consider gap cutting as a restoration method is also ecologically motivated by the fact that different kinds of both large and small scale disturbances historically has shaped these forest ecosystems and the species therein (Kuuluvainen and Aakala 2011).
Between year variation
The low overall density of saproxylic beetles in 2011 compared to 2010, as exemplified by a lower density in the reference stands, is probably a result of differences in sampling period between years, sampling started first of June in 2010 but between 12 June and 15 July in 2011 due to variation in burning date. As many species of saproxylic beetles have their activity peak early in the season a delay in the onset of sampling could lead to a reduction in the number of sampled individuals and thus also species. However, between-year differences in climatic conditions, a pattern that has been reported in earlier studies (Boucher et al. 2012; Toivanen and Kotiaho 2010) could also potentially explain between-year variation in this study. This stresses the importance of a proper experimental design including reference (i.e. untreated) stands and before-after studies.
Conclusions
Restoration of degraded forest habitats is essential for future biodiversity conservation and our study shows that burning clearly attracts fire dependent and fire favoured saproxylic beetles. However, many species are disfavoured by burning suggesting that alternative restoration measures also should be considered. Although we did not find any strong overall significant effect of gap cutting on saproxylic assemblages as a whole, we found positive effects on abundance of cambium consumers and fire favoured species. We predict that the increase of dead wood availability in these stands is likely to increase on-site production with time (Hjältén et al. 2012). However, more long-term studies are needed to verify this.
For efficient biodiversity management, restoration often needs to be repeated at regular intervals in a landscape (Hekkala et al. 2014). However, restoration of legally protected areas, e.g. nature reserves and national parks, is often controversial. Voluntary set-asides could play an important role in landscape management, as stipulated in the Swedish FSC-standards, they cover 5% of the certified productive forest area in Sweden (Anonymous 2010b). In addition, the partly novel way to conduct restoration used in this study rendered restorations to be cost-neutral as revenues from the thinning prior to burning and trees extracted during gap cutting covered restoration costs (Olov Norgren pers. comm.). Voluntary set-asides thus provide an excellent opportunity for implementing active and cost-efficient landscape management for biodiversity conservation, and can act as an important complement to the often passive conservation measures provided by formally protected areas such as nature reserves.
References
Ahti T, Hämet-Ahti L, Jalas J (1968) Vegetation zones and their sections in northwestern Europe. Ann Bot Fenn 5:169–211
Angelstam P, Andersson K, Axelsson R, Elbakidze M, Jonsson BG, Roberge J-M (2011) Protecting forest areas for biodiversity in Sweden 1991-2010: the policy implementation process and outcomes on the ground. Silva Fenn 45:1111–1133. doi:10.14214/sf.90
Anonymous (2007) The saproxylic database—biodiversity in dead wood. www.saproxylic.org/. Accessed 25 May 2015
Anonymous (2010) Finlands FSC-standard. https://fi.fsc.org/preview.finlands-fsc-standard.a-97.pdf
Anonymous (2010) Swedish forestry stewardship council. Swedish FSC standard for forest certification, English version. http://www.fsc-sverige.org/. Accessed 8 Sept 2015
Anonymous (2014) Swedish FSC standard for forest certification including SLIMF indicators. FSC Sweden
Aronson J, Alexander S (2013) Ecosystem restoration is now a global priority: time to roll up our sleeves. Restor Ecol 21:293–296. doi:10.1111/rec.12011
Azeria ET, Ibarzabal J, Hebert C (2012) Effects of habitat characteristics and interspecific interactions on co-occurrence patterns of saproxylic beetles breeding in tree boles after forest fire: null model analyses. Oecologia 168:1123–1135. doi:10.1007/s00442-011-2180-0
Bates D, Maechler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. Arxiv e-print; in press, J Stat Softw. http://arxiv.org/abs/1406.5823
Berg A, Ehnström B, Gustafsson L, Hallingbäck T, Jonsell M, Weslien J (1994) Threatened plant, animal, and fungus species in swedish forests—distribution and habitat associations. Conserv Biol 8:718–731
Bernes C (2011) Biodiversity in Sweden, vol Monitor 22. Swedish Environmental Protection Agency
Boucher J, Azeria ET, Ibarzabal J, Hebert C (2012) Saproxylic beetles in disturbed boreal forests: temporal dynamics, habitat associations, and community structure. Ecoscience 19:328–343. doi:10.2980/19-4-3503
Buddle CM, Langor DW, Pohl GR, Spence JR (2006) Arthropod responses to harvesting and wildfire: implications for emulation of natural disturbance in forest management. Biol Conserv 128:346–357
CBD (2010) Global biodiversity outlook 2. Secretariat of the convention on biological diversity, Montreal
Dyntaxa (2015) Swedish taxonomic database. www.Dyntaxa.Se. Accessed 15 Oct 2015
EU (2011) Our life insurance, our natural capital: an EU biodiversity strategy to 2020. Brussels, 3 May 2011
FAO (2010) Global forest resources assessment 2010, Rome
Granström A (2001) Fire management for biodiversity in the European boreal forest. Scand J For Res 16:62–69
Granström A, Schimmel J (1993) Heat-effects on seeds and rhizomes of a selection of boreal forest plants and potential reaction to fire. Oecologia 94:307–313. doi:10.1007/bf00317103
Gustafsson L, Appelgren L, Jonsson F, Nordin U, Persson A, Weslien JO (2004) High occurrence of red-listed bryophytes and lichens in mature managed forests in boreal Sweden. Basic Appl Ecol 5:123–129
Hekkala A-M, Paatalo M-L, Tarvainen O, Tolvanen A (2014) Restoration of young forests in eastern Finland: benefits for saproxylic beetles (coleoptera). Restor Ecol 22:151–159. doi:10.1111/rec.12050
Hjältén J et al (2012) Micro and macro-habitat associations in saproxylic beetles: implications for biodiversity management. PLoS ONE 7:e41100. doi:10.1371/journal.pone.0041100
Hothorn T, Bretz F, Westfall P (2008) Simultaneous inference in general parametric models. Biometrical Journal 50:346–363. doi:10.1002/bimj.200810425
Hulcr J, Ubik K, Vrkoc J (2006) The role of semiochemicals in tritrophic interactions between the spruce bark beetle Ips typographus, its predators and infested spruce. J Appl Entomol 130:275–283. doi:10.1111/j.1439-0418.2006.01069.x
Hyvärinen E, Kouki J, Martikainen P, Lappalainen H (2005) Short-term effects of controlled burning and green-tree retention on beetle (coleoptera) assemblages in managed boreal forests. For Ecol Manag 212:315–332
Hyvärinen E, Kouki J, Martikainen P (2006) Fire and green-tree retention in conservation of red-listed and rare deadwood-dependent beetles in finnish boreal forests. Conserv Biol 20:1710–1719. doi:10.1111/j.1523-1739.2006.00511.x
Hyvärinen E, Kouki J, Martikainen P (2009) Prescribed fires and retention trees help to conserve beetle diversity in managed boreal forests despite their transient negative effects on some beetle groups. Insect Conserv Divers 2:93–105. doi:10.1111/j.1752-4598.2009.00048.x
Jacobs DF et al (2015) Restoring forests: what constitutes success in the twenty-first century? New For 46:601–614. doi:10.1007/s11056-015-9513-5
Johansson T et al (2006) Conservation-oriented manipulations of coarse woody debris affect its value as habitat for spruce-infesting bark and ambrosia beetles (Coleoptera: Scolytinae) in northern Sweden. Can J For Res-Revue Canadienne De Recherche Forestiere 36:174–185. doi:10.1139/x05-235
Johansson T et al (2007) The effects of substrate manipulations and forest management on predators of saproxylic beetles. For Ecol Manag 242:518–529. doi:10.1016/j.foreco.2007.01.064
Johansson T, Andersson J, Hjältén J, Dynesius M, Ecke F (2011) Short-term responses of beetle assemblages to wildfire in a region with more than 100 years of fire suppression. Insect Conserv Divers 4:142–151. doi:10.1111/j.1752-4598.2010.00114.x
Jonsson BG, Kruys N, Ranius T (2005) Ecology of species living on dead wood—lessons for dead wood management. Silva Fenn 39:289–309
Kouki J, Hyvärinen E, Lappalainen H, Martikainen P, Similä M (2012) Landscape context affects the success of habitat restoration: large-scale colonization patterns of saproxylic and fire-associated species in boreal forests. Divers Distrib 18:348–355. doi:10.1111/j.1472-4642.2011.00839.x
Kuuluvainen T (2009) Forest management and biodiversity conservation based on natural ecosystem dynamics in northern Europe: the complexity challenge. Ambio 38:309–315
Kuuluvainen T, Aakala T (2011) Natural forest dynamics in boreal fennoscandia: a review and classification. Silva Fenn 45:823–841. doi:10.14214/sf.73
Lee S-I, Spence JR, Langor DW (2014) Succession of saproxylic beetles associated with decomposition of boreal white spruce logs. Agric For Entomol 16:391–405. doi:10.1111/afe.12069
Lindenmayer DB, Franklin JF, Fischer J (2006) General management principles and a checklist of strategies to guide forest biodiversity conservation. Biol Conserv 131:433–445
MEA (2005) Millennium ecosystem assessment: Ecosystems and human well-being: biodiversity synthesis
Oksanen J et al. (2015) Vegan: community ecology package. R package version 2.3-1
Paillet Y et al (2010) Biodiversity differences between managed and unmanaged forests: meta-analysis of species richness in europe. Conserv Biol 24:101–112. doi:10.1111/j.1523-1739.2009.01399.x
R-Core-Team (2015) R: a language and environment for statistical computing. R foundation for statistical computing, Vienna, Austria. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org/
Saint-Germain M, Drapeau P, Hebert C (2004) Xylophagous insect species composition and patterns of substratum use on fire-killed black spruce in central Quebec. Can J For Res-Revue Canadienne de Recherche Forestiere 34:677–685. doi:10.1139/x03-235
Saint-Germain M, Drapeau P, Buddle CM (2008) Persistence of pyrophilous insects in fire-driven boreal forests: population dynamics in burned and unburned habitats. Divers Distrib 14:713–720. doi:10.1111/j.1472-4642.2007.00452.x
Sayer J, Chokkalingam U, Poulsen J (2004) The restoration of forest biodiversity and ecological values. For Ecol Manag 201:3–11. doi:10.1016/j.foreco.2004.06.008
Schroeder LM (2003) Differences in responses to alpha-pinene and ethanol, and flight periods between the bark beetle predators Thanasimus femoralis and T. formicarius (col.: Cleridae). For Ecol Manag 177:301–311. doi:10.1016/s0378-1127(02)00441-3
Siitonen J (2001) Forest management, coarse woody debris and saproxylic organisms: Fennoscandian boreal forest as an example. Ecol Bull 49:11–41
SLU (2012) Forest statistics 2012. Official statistics of Sweden. Swedish University of Agricultural Sciences, Umeå
Speight MCD (1989) Saproxylic invertebrates and their conservation. Council of Europe, Strasbourg
Stanturf JA (2015) Future landscapes: opportunities and challenges. New For 46:615–644. doi:10.1007/s11056-015-9500-x
Stenbacka F, Hjältén J, Hilszczanski J, Dynesius M (2010) Saproxylic and non-saproxylic beetle assemblages in boreal spruce forests of different age and forestry intensity. Ecol Appl 20:2310–2321. doi:10.1890/09-0815.1
Stokland JN, Siitonen J, Jonsson BG (2012) Biodiversity in dead wood. Cambridge University Press, Cambridge
Toivanen T, Kotiaho JS (2007) Mimicking natural disturbances of boreal forests: the effects of controlled burning and creating dead wood on beetle diversity. Biodivers Conserv 16:3193–3211. doi:10.1007/s10531-007-9172-8
Toivanen T, Kotiaho JS (2010) The preferences of saproxylic beetle species for different dead wood types created in forest restoration treatments. Can J For Res-Revue Canadienne De Recherche Forestiere 40:445–464. doi:10.1139/x09-205
Toivanen T, Liikanen V, Kotiaho JS (2009) Effects of forest restoration treatments on the abundance of bark beetles in norway spruce forests of southern Finland. For Ecol Manag 257:117–125. doi:10.1016/j.foreco.2008.08.025
Wang Y, Naumann U, Wright ST, Warton DI (2012) Mvabund: an r package for model-based analysis of multivariate abundance data. Methods Ecol Evol 3:471–474. doi:10.1111/j.2041-210X.2012.00190.x
Warton DI, Wright ST, Wang Y (2012) Distance-based multivariate analyses confound location and dispersion effects. Methods Ecol Evol 3:89–101. doi:10.1111/j.2041-210X.2011.00127.x
Wermelinger B, Duelli P, Obrist MK (2002) Dynamics of saproxylic beetles (coleoptera) in windthrow areas in alpine spruce forests. For Snow Landsc Res 77:133–148
Westling A (2015) Rödlistade arter i sverige 2015—the 2015 redlist of Swedish species. Artdatabanken, Uppsala
Wikars L-O (1997) Effects of forest fire and the ecology of fire adapted insects. PhD thesis, University of Uppsala
Wikars L-O (2002) Dependence on fire in wood-living insects: an experiment with burned and unburned spruce and birch logs. J Insect Conserv 6:1–12
Wikars L-O (2006) Åtgärdsprogram för bevarande av brandinsekter i boreal skog, vol RAPPORT 5610. Naturvårdsverket
Zackrisson O (1977) Influence of forest fires on the north Swedish boreal forest. Oikos 29:22–32
Acknowledgements
The forest company Holmen provided forest stands and conducted the restoration treatments. Funding was provided by The Swedish Research Council for Environment, Agricultural Sciences and Spatial Planning (Formas), the Kempe foundation and the research program Future Forests. We thank Stig Lundberg and Jacek Hilszczański for identifying the beetles and Adrian Hjältén, Alexander Hjältén, Isak Lindmark Caroline Letzner, Josefine Letzner, Nina Stenbacka and Nils Ericson for help with the fieldwork.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Andreas Schuldt.
This article belongs to the Topical Collection: Forest and plantation biodiversity.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Hjältén, J., Hägglund, R., Löfroth, T. et al. Forest restoration by burning and gap cutting of voluntary set-asides yield distinct immediate effects on saproxylic beetles. Biodivers Conserv 26, 1623–1640 (2017). https://doi.org/10.1007/s10531-017-1321-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-017-1321-0